Abstract
Relapse continues to be the major cause of failure after allogeneic hematopoietic cell transplantation (HCT) for myelodysplastic syndrome (MDS). Most patients who experience relapse do so within a short interval after HCT. However, late relapses occur and patients who relapse late may do so as a result of different pathogenic mechanisms.
From 1984 to 2007, 961 patients underwent a first HCT for MDS (including RAEB-T and CMML) at our intuition, of whom 215 relapsed after HCT. We analyzed results in the 44 patients whose relapse was diagnosed more than 1.5 years after HCT (median 2.62, range 1.83–20.53 years). Patients were 12–71 (median 45) years old and had been conditioned with either high- (91%) or reduced-intensity (9%) conditioning regimens. Donors were related (64%) or unrelated (36%); the graft source was marrow (55%), peripheral blood (43%), or cord blood (2%). The association of the patient, disease, and transplant parameters with time to relapse and survival after relapse was examined in univariate and multivariate analyses.
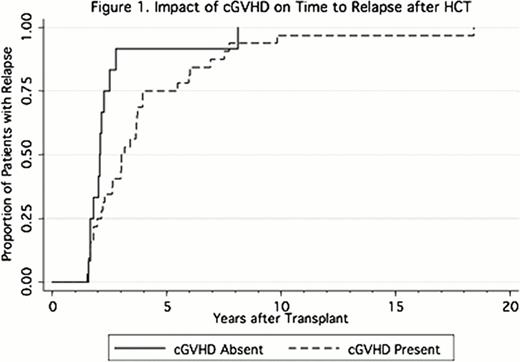
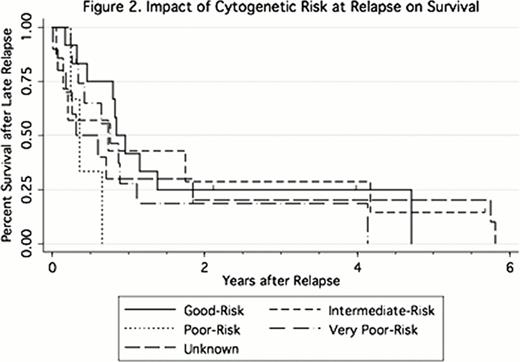
Five patients relapsed with extramedullary disease, 4 without evidence of bone marrow involvement. The presence of chronic graft-versus-host-disease (cGVHD; HR = 0.48 [0.27–0.94, p = 0.04], Figure 1.) was associated with a longer delay of relapse. Both the presence of cGVHD (HR = 0.34 [0.14–0.83, p = 0.18]) and bone marrow as a source of stem cells (HR = 0.36 [0.17–0.74, p = 0.06]) were associated with a longer time to relapse (when adjusted for age, disease duration, and cytogenetic risk). 5 patients were alive (1 of 4 in remission, 1 unknown) at the time of analysis. The median survival after relapse was 0.72 (range 0.01–5.81) years. Age > 50 years (HR = 0.41 [0.21–0.80, p = 0.001]) and time from HCT to relapse > 3 years (HR = 0.2 [0.09–0.46, p < 0.001]) were associated with better survival after relapse. Patients who relapsed at > 3 years (HR = 0.24 [0.09–0.63, p = 0.004]) and patients who received a second HCT (HR = 0.34 [0.12–0.94, p = 0.039]) had increased survival after relapse (adjusted for age, year, and cytogenetic risk at relapse). Cytogenetic risk at relapse was not significantly associated with survival after relapse (Figure 2.).
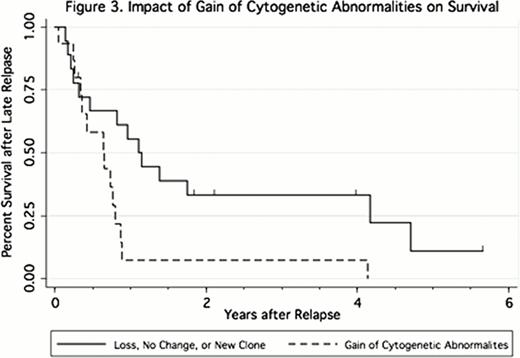
All patients had cytogenetics determined prior to HCT, 33 patients had evaluable cytogenetics at relapse, and 28 patients had a complete set (diagnosis, prior to HCT, and at relapse). The majority (88%) of the relapse karyotypes harbored at least one of the abnormalities present prior to HCT, suggesting a common clonal origin; however, 21 (64%) exhibited a change in the pattern of chromosomal abnormalities from pre-HCT to relapse. Of these, 15 gained new abnormalities (with or without loss of previously present abnormalities), and 2 showed exclusively a loss of lesions present prior to HCT. In 4 patients, all abnormalities present before HCT were absent at relapse, raising the possibility that the relapse represented the emergence of a second unrelated clone. Cytogenetic changes (gain, loss, no change, new clone) on relapse were not predictive of time to relapse. However, gain of new abnormalities was associated with inferior survival after relapse (HR = 2.76 [1.21–6.32, p = 0.016], Figure 3.) in a univariate analysis, and after adjusting for second HCT (HR = 2.56 [1.13–5.85, p = 0.025]).
Relapse more than eighteen months after HCT for MDS is an uncommon event. However, study of late relapses after HCT may provide useful insights into pathogenic mechanisms of relapse as disease control appears to have been obtained initially and maintained for a period of time, but was subsequently lost. Although relapse is thought to be the result of a malignant clone escaping the graft-versus-leukemia effect, the exact pathways leading to relapse after HCT and predictors of subsequent survival are poorly understood. The heavy impact of cGVHD on the time to relapse supports this hypothesis. Survival after relapse was predicted by disease-specific factors, in particular the gain of cytogenetic abnormalities as opposed to the overall cytogenetic risk, rather than graft-specific factors. This suggests that graft manipulation (i.e. donor lymphocyte infusion) would be a less effective strategy in this setting. Finally, our data suggest that in select patients, a second transplant may provide a survival benefit.
No relevant conflicts of interest to declare.

This icon denotes a clinically relevant abstract
Author notes
Asterisk with author names denotes non-ASH members.