Abstract
Abstract 2303
The genes and pathways that govern the functions and expansion of hematopoietic stem cells (HSC) remain to be elucidated. Pim (proviral insertion in murine lymphomas) protein kinases are a small family of constitutively active, highly conserved oncogenic serine/threonine kinases and have 3 members: Pim1, Pim2, and Pim3. Currently, little is known about the contribution of each Pim kinase in hematopoiesis. Pim1 was recently found to be important in the regulation of CXCR4 expression in HSCs (Grundler R et al, J. Exp Med 2009, 206:1957). However, the roles of Pim1 in HSC proliferation, self-renewal, and long-term repopulation are unclear. In the current study, we performed detailed hematological and bone marrow transplant studies to address these questions.
We generated Pim1 transgenic (Tx) mice bearing human PIM1 under the control of vav hematopoietic promoter (vav-hPIM1 Tx). Pim1−/−, Pim2−/−, Pim3−/− single knockout (KO) mice were also utilized. We quantified the number of hematopoietic stem/progenitor cells (HSPCs) in these mice using flow cytometry, colony forming units (CFUs) and cobblestone area forming cell colonies (CAFCs) assays. In vivo BrdU labeling was performed to determine the proliferation status of long-term HSCs in these mice. CXCR4 expression and the homing of HSCs to bone marrow and spleen were also analyzed. Additionally, we performed noncompetitive, competitive, and serial transplantation assays to examine the role of Pim1, Pim2 and Pim3 in hematological reconstitution in lethally irradiated recipient mice. Finally, a limiting dilution competitive bone marrow transplantation assay using purified long-term HSC cells (Lin− Sca-1+c-kit+ CD34−) was performed to calculate the frequency of active HSCs in vav-hPIM1 Tx and Pim1−/−mice.
Compared to wild type (WT) littermate controls, vav-hPIM Tx mice showed enhanced hematopoiesis as demonstrated by increased numbers of Lin− Sca-1+c-Kit+ (LSK) HSPCs and CAFCs, higher BrdU incorporation in long-term HSC population, and a better ability to reconstitute lethally irradiated mice. The number of HSPCs was increased by ∼2 fold in vav-hPIM1 Tx mice as estimated by quantification of LSK cells and by limiting dilution transplant assay.
Pim1−/− bone marrow cells had reduced CFU-GEMMs and reduced capacity to form CAFCs compared to WT bone marrow cells. Those defects were not observed in Pim2−/− or Pim3−/−mice.
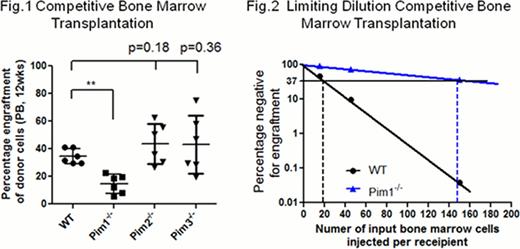
Bone marrow cells from Pim1−/−, but not Pim2−/− or Pim3−/−mice had defects in reconstituting lethally irradiated recipient mice as demonstrated in noncompetitive, competitive (Fig. 1, ** p<0.01) and serial transplantation assays.
Pim1−/− mice demonstrated reduced BrdU incorporation in LSK+CD34−CD135− long-term HSCs in vivo. Interestingly, in contrast to previous report, we did not observe significant defects in the homing of Pim1−/− bone marrow cells to recipient's bone marrow at 12 hrs and 24 hrs post bone marrow transplantation. A slight reduction (<20%) in homing to spleen was noted in recipient mice transplanted with Pim1−/− Lin−ScalI+cells at 12 hrs post transplantation.
Limiting dilution competitive bone marrow transplantation and Poisson statistical analysis (n = 54 mice) showed that the frequency of active LSK+CD34− HSCs was reduced by ∼10 fold in Pim1−/− mice (1 in 146 cells in Pim1−/−KO mice vs. 1 in 19 cells in WT control mice, p<0.01) (Fig. 2).
Our current studies demonstrate a novel role of Pim1 serine/threonine kinase in the regulation of self-renewal, proliferation, and long-term repopulation of HSCs. The functions of Pim1 in hematopoiesis do not overlap with Pim2 or Pim3.
We thank Richard Peppler at the Hollings Cancer Center Flow Cytometry Core for performing flow cytometry analysis. This work is supported by MUSC Hollings Cancer Center Startup Fund, Hollings Cancer Center ACS IRG, ASCO Conquer Cancer Foundation Career Development Award, NIH 1K08HL 103780–01A1, and NIH 3P30CA138313–01S3.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.