Abstract
Abstract 928
B-cell receptor (BCR) signaling and cytoskeletal activation represent critical steps in the pathogenesis of Chronic Lymphocytic Leukemia (CLL). We previously demonstrated that Hematopoietic cell specific Lyn substrate 1 (HS1) i) is a potential prognostic marker as CLL patients whose leukemic cells carry hypo-phosphorylated (hypo-p) HS1 have a better prognosis than patients with the hyper-phosphorylated (hyper-p) form; ii) is a central interactor of several cytoskeletal components and iii) has a profound effect on the development and progression of CLL in the Eμ-TCL1 transgenic mouse suggesting that the hyper-phosphorylation of the molecule may be responsible of HS1 inactivation rather than its activation.
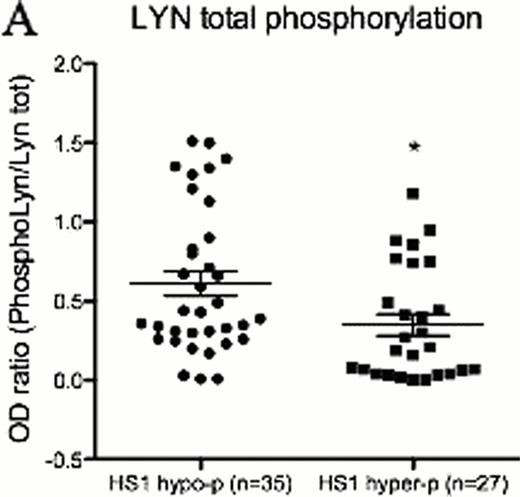
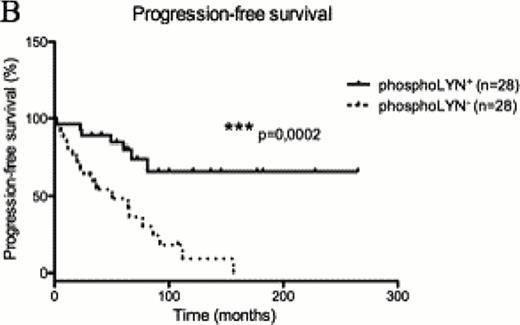
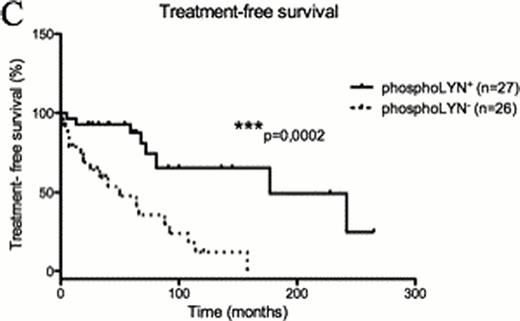
To better define the role of HS1 phosphorylation in CLL cell biology we dissected the BCR signaling pathway in cells with differential phosphorylation of the HS1 protein. Taking advantage of the CLL cell line MEC1 where we silenced the expression of HS1, we observed that the phosphorylation status of several BCR signalling molecules, including LYN kinase, is reduced in the absence of HS1. A similar pattern of modifications was confirmed in primary cells from 62 patients with different phosphorylation status of HS1 (Figure A n=35 HS1hypo-p vs n=27 HS1hyper-p, p=0,01), thereby identifying a distinct phospho-signature associated to HS1. LYN phosphorylation correlates with clinical parameters as phosphoLYN+ patients showed significantly increased (Figure B n=28 phosphoLYN+ vs n=28 phosphoLYN− p=0,0002) Progression-free survival and Treatment-free survival (Figure C n=27 phosphoLYN+ vs n=26 phosphoLYN− p=0,0002) as compared to phosphoLYN−patients.
We further investigated the relationship between HS1 and LYN phosphorylations and showed that LYN kinase activation- Tyrosine (Y)396 phosphorylation- is associated with the phosphorylation of the active site Y397 on HS1 protein (Spearman correlation n=40, p<0,0001). Patients may thus be categorized into two subgroups based on the phosphorylation status of the two molecules as “active-HS1” - HS1 hypo-p/phosphoLYN+ and “inactive-HS1” - HS1 hyper-p/phosphoLYN−-.
These different patterns correlated with distinct cytoskeletal functionality in terms of migration (active-HS1 n=12 vs inactive-HS1 n=9, p= 0,009), spontaneous adhesion (active-HS1 n=12 vs inactive-HS1 n=9, p=0,03), adhesion to an HS5 stromal cell layer (active-HS1 n=9 vs inactive-HS1 n=10 p=0,006) and F-actin polymerization (active-HS1 n=12 vs inactive-HS1 n=11, p=0,001). Targeting the LYN/HS1 axis in vitro with the tyrosine kinase inhibitor Dasatinib significantly affected primary CLL cell viability with a direct correlation with basal LYN phosphorylation (Sperman correlation n=32, p=0,04) and interfered with cytoskeleton activity, with a preference for patients carrying the “active-HS1” signature. Accordingly, 100nM Dasatinib markedly reduced migration of active-HS1 samples (active-HS1 n=10, p=0,003 vs inactive-HS1 n=7, p>0,05), with no significant effect on inactive-HS1 samples. F-actin polymerization (active-HS1 n=9 p=0,003, vs inactive-HS1 n=8 p=0,01) was reduced in both groups by Dasatinib treatment, while spontaneous (active-HS1 n=7, p=0,03 vs inactive-HS1 p>0,05) and HS5-mediated cell adhesion (active-HS1 n=8, p=0,03 vs inactive-HS1 n=8 p>0,05) were affected by the treatment only in the active-HS1 group.
In the adoptive transfer mouse model based on the EmTCL1 transgenic mouse, a 4-week Dasatinib treatment reduced the percentage of CD19+CD5+Igk+ CLL cells in the blood as soon as one week after treatment initiation and throughout the whole treatment period (p(Day6)=0,007, p(Day13)=0,005, p(Day20)=0,004, p(day of killing)=0,01). At sacrifice, a significant reduction in the absolute number of leukemic cells in the spleen (p=0,05), bone marrow (p=0,03) and lymph nodes (p=0,04) was observed in treated mice as compared to controls. Consistent with the biochemical action of Dasatinib, significant reduction in SRC-Y416 phosphorylation was observed after intracellular staining of leukemic B cells (p=0,04) at the end of treatment.
These observations suggest that the phosphorylations status of LYN and HS1 as a biological indicator of respectively distinct signaling pathway and cytoskeletal-related features that can be selectively targeted for therapeutic intervention in a sizable fraction of CLL patients.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.