Abstract
Ruxolitinib (RUX) is a JAK1 & JAK2 inhibitor that resulted in rapid and durable reductions in splenomegaly and improved disease-related symptoms and quality of life in patients (pts) with myelofibrosis (MF) compared with either placebo (COMFORT-I) or best available therapy (BAT; COMFORT-II). In addition, RUX-treated pts had longer overall survival (OS) compared with placebo and BAT. We recently reported that, among 879 primary MF pts receiving conventional BAT, those harboring a mutation in any one of EZH2, ASXL1, IDH1/2 and SRSF2 constituted an IPSS- and DIPSS-plus prognostic score–independent “high molecular risk” (HMR+) category associated with shorter OS and greater risk of leukemia compared with pts with no mutations (“low molecular risk”; LMR) (Vannucchi AM, et al. Leukemia. 2013). The aim of this study was to analyze the impact of mutational status on spleen volume reduction, anemia development, and OS in pts receiving RUX in the COMFORT II trial.
In COMFORT-II, pts with primary or post–polycythemia vera/–essential thrombocythemia MF were randomized to receive RUX (n=146) or BAT (n=73). Mutations in 12 genes (JAK2, MPL, EZH2, ASXL1, TET2, IDH1/2, CBL, SRSF2, SOCS1, SOCS2, SOCS3, and SH2B3) were genotyped, in DNA derived from whole blood, at baseline in 166 pts (RUX n=120, BAT n=46). Analysis was performed by next-generation sequencing with the Ion Torrent PGM or Roche 454 platform. Sequencing data were analyzed with Nextgene software or Roche 454 Analysis software v2.6 with variant frequency cutoff adjusted to 5%. All mutations were confirmed at least twice. Missense, nonsense, and frameshift mutations only were considered; in the case of novel mutations, SNPs were excluded by database searching and by germline DNA genotyping when available. Development of anemia was defined as a drop of hemoglobin level by more than 1 g/dL from baseline to a value <10 g/dL within the first 48 weeks of treatment. Survival estimates were obtained with Kaplan-Meier method. The treatment effect and the prognostic value of the molecular variables with regard to OS were analyzed by Cox regression and adjusted for the IPSS category.
The frequency of mutations was: JAK2V617F 75.47%; MPLW515 7.74%; ASXL1 32.53%; TET2 10.69%; EZH2 7.24%; CBL 4.4%; SRSF2 3.01%; SH2B3 1.3%; IDH1-2 0.7%; SOCS1 0.65%; SOCS2 0.65%; SOCS3 0.0%, with no difference between RUX and BAT. Forty-six (38.3%) and 20 (43.5%) pts in the RUX and BAT groups, respectively, were classified as HMR+.
We first determined whether an HMR+ status impacted the achievement of a ≥35% spleen volume reduction (primary study endpoint). The percentage of RUX treated pts achieving ≥35% spleen volume reduction was 36.3% (16/44) and 33.8% (21/62) at 24 wk and 30.7% (12/39) and 36.3% (20/55) at 48 wk in the HMR+ and LMR categories, respectively. Mean spleen volume reduction was also similar: -29.0% and -23.5% in HMR+ vs -29.9% and -30.6% in LMR pts at 24 and 48 wk. None of the other mutations analyzed correlated with spleen volume reduction in pts receiving RUX.
We also found that an HMR+ status did not predict for the development of anemia associated with RUX administration: the percentage of anemic pts was 74% in the HMR+ group vs 72% in the LMR group. This was also independent of the presence of mutation in any one of the genes associated with JAK2/STAT signaling (JAK2, MPL, SH3B2, CBL, and SOCSs): anemic pts were 74% in mutated vs 72% in wild-type ones.
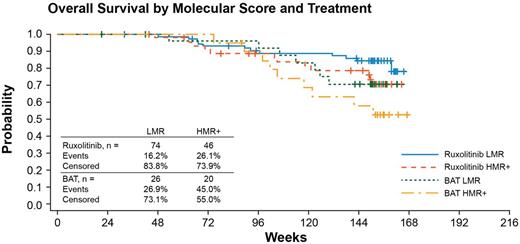
The survival estimate at 114 wk of follow-up in BAT pts was 0.58 and 0.71 in HMR+ and LMR pts, confirming the negative impact of the mutational risk category. In the RUX arm, the survival estimate was 0.79 and 0.85 for HMR+ and LMR pts, indicating a benefit of RUX treatment in both groups. In the multivariate Cox model, a risk of death with RUX compared with BAT was reduced by 43% (HR=0.57, 95% CI: 0.30-1.08) and LMR patients had a reduction in risk compared with HMR+ by 38% (HR=0.62, 95% CI: 0.33-1.16; Figure).
The prognostic value of detrimental mutations comprising an HMR+ category in MF was maintained within this subset of COMFORT II patients. The results suggest that the negative impact of HMR+ status on survival in MF pts in COMFORT-II is halved by treatment with RUX compared with BAT and that the effect of RUX is similar in both the HMR+ and LMR groups; in addition, HMR+ status did not affect the likelihood of obtaining a ≥35% spleen volume reduction nor the risk of developing anemia under treatment.
Stalbovskaya:Novartis Pharma AG: Employment, Equity Ownership. Squires:Novartis Pharma AG: Employment. Harrison:NOVARTIS: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Research Funding, Speakers Bureau; Sanofi: Honoraria, Membership on an entity’s Board of Directors or advisory committees, Speakers Bureau; YM Bioscience: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Celgene: Honoraria; Shire: Speakers Bureau; Sbio: Honoraria, Membership on an entity’s Board of Directors or advisory committees; Gilead: Honoraria, Membership on an entity’s Board of Directors or advisory committees. Vannucchi:NOVARTIS: Honoraria, Membership on an entity’s Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.