Abstract
Ph-negative myeloproliferative neoplasms (MPNs) are characterized by many somatic mutations which have already been shown useful in the prognostic assessment of MPN patients [A.M. Vannucchi et al., Leukemia, 2013]. Moreover, aberrant microRNA (miRNA) expression seems to add to the molecular complexity of MPNs, as specific miRNA signatures capable of discriminating MPN cells from those of normal donors were previously reported [P. Guglielmelli et al., Exp Hematol, 2007]. In order to have a comprehensive picture of miRNA deregulation and its relationship with differential gene expression in primary myelofibrosis (PMF) cells, we obtained gene- (GEP) and miRNA expression profiles (miEP) of CD34+ cells from 31 healthy donors and 42 PMF patients using Affymetrix technology (HG-U219 and miRNA 2.0 arrays). Among 726 differentially expressed genes (DEG) we found that several putative cancer markers (WT1, ANGPT1) and several genes related to PMF progression, i.e. involved in megakaryocyte (MK) differentiation (NFE2, CD9), and fibrosis development (DLK1, LEPR1), were significantly more expressed in PMF samples than in the normal counterpart. Similarly, as regards the miEP, among 74 human differentially expressed miRNAs (DEM) in PMF compared to controls we found the upregulation of several miRNAs associated with hematological malignancies or known as oncomiRs (i.e. hsa-miR-155-5p [S. Jiang et al., Cancer Res, 2010], miRNAs belonging to the miR-17-92 cluster [L. Venturini et al., Blood, 2007]), and other aberrantly expressed miRNAs never described in hematopoiesis (i.e. hsa-miR-335-5p).
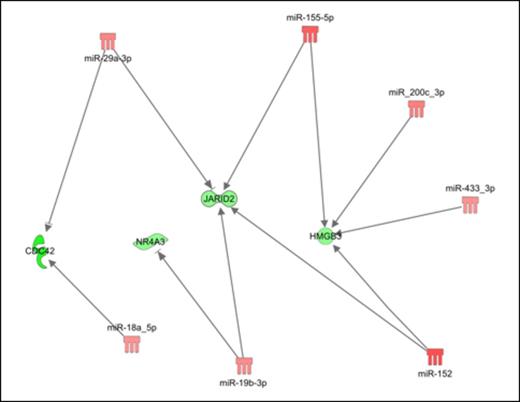
Then, in order to construct regulatory networks of the functional human miRNA-target interactions, we performed an integrative analysis (IA) with Ingenuity Pathway analysis software, which combines the miRNA expression profile with computational predicted targets and with the gene expression data. IA between DEG and DEM disclosed a high number of predicted targets with anti-correlated expression to the trend of their targeting miRNAs. Of note, IA identified an interaction network (see Figure) in which the upregulated oncomirs miR-155-5p [R.M. O'Connel et al., J Exp Med, 2008], miR29a-3p [Y.C. Han et al., J Exp Med, 2010] and miR-19b-3p [K.J. Mavrakis et al., Nat Cell Biol, 2010] could explain the downregulation of targets whose lower expression was already described as involved in myeloproliferative phenotypes, such as NR4A3, CDC42, HMGB3. Additionally, IA disclosed the chromatin remodeler JARID2, which is frequently deleted in leukemic transformation of chronic myeloid malignancies, as a shared target of several upregulated miRNAs in PMF samples (i.e. miR-155-5p, miR-152-3p). Noteworthy, these miRNA-mRNA interactions were functionally confirmed by 3' UTR luciferase reporter assays.
Next, in order to characterize the role of JARID2 in PMF pathogenesis, we performed RNAi-mediated gene silencing experiments on CD34+ cells of healthy donor. Interestingly, inhibition of JARID2 expression produces in silenced cells a significant increase of CD41 expression when compared with control (28.6±3.1% vs 15.3±1.8% at day 8, 52.6±7.6% vs 35.4±4.9% at day 12 of serum free liquid culture) and a remarkable increase in CFU-MK colonies (59.6±6.5% vs 39.8±5.9%). The values are reported as mean ± 2S.E.M from five independent experiments.
Moreover, morphological analysis after May-Grunwald-Giemsa staining showed that JARID2 silencing induces in normal CD34+ cells a considerable enrichment in MK precursors at different stages of maturation.
This study allowed the identification of different networks possibly involved in PMF onset, highlighting the potential contribution of miRNAs to PMF pathogenesis. Furthermore, for the first time, we demonstrated that the JARID2 downregulation in CD34+ cells might contribute to the abnormal megakaryopoiesis typical of PMF.
Rambaldi:Novartis: Honoraria; Sanofi: Honoraria; Italfarmaco: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

