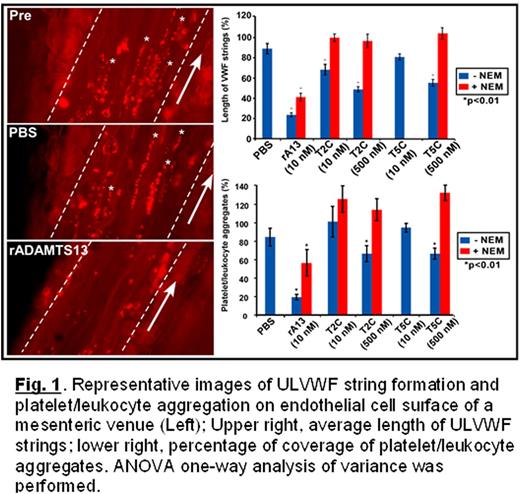
von Willebrand factor (VWF) undergoes self-association mediated by free cysteine residues between two apposed VWF multimers. This process occurs in solution or between soluble and ultra large (UL) VWF on endothelial cells or matrix under flow. ADAMTS13 is a key regulator for the ULVWF string formation and adhesive function, primarily through its proteolytic cleavage at the Tyr1605-Met1606 bond in the central A2 domain of VWF. The proteolytic cleavage of VWF by ADAMTS13 rapidly removes newly secreted ULVWF on activated endothelial cell surface, thereby preventing excessive platelet aggregation and thrombus formation under physiological conditions. Previous studies have shown that the proteolytic cleavage of both cell-bound and soluble ULVWF by ADAMTS13 depends on its amino terminal MDTCS domains. However, the role of more distal carboxyl-terminal domains is not fully established. Here, we show by N-ethylmaleimide (NEM) labeling and mass spectrometry that TSP1 2-8 repeats and CUB domains of human recombinant ADAMTS13 contain multiple free cysteine residues (-SH). We hypothesize that these free thiols may directly interact with those on the surface of VWF multimers, thereby preventing their lateral association, thereby inhibiting thrombus formation under flow. Using an intravital microscopic imaging technique we demonstrate that even in the absence of circulating ADAMTS13 (Adamts13-/-) the formation and elongation of ULVWF strings on endothelial cells of mesenteric venules after FeCl3 injury are highly dynamic over time (180 seconds), suggesting other proteases or reductases in blood are able cleave or reduce ULVWF strings under flow. However, the average length of ULVWF strings remains relatively constant. Recombinant ADAMTS13 (rA13) at physiological concentration (10 nM), when infused, rapidly cleaves or eliminates preformed ULVWF strings and platelet/leukocyte aggregates on endothelial cells. The efficacy of rA13 in reducing the average length of ULVWF strings and platelet/leukocyte aggregation decreases by ∼50% if rA13 is pretreated with NEM. Moreover, a C-terminal fragment of ADAMTS13 comprising 2-8 TSP1 repeats and two CUB domains (T2C) or containing 5-8 TSP1 repeats and two CUB domains (T5C), when infused, also significantly attenuates the formation and/or elongation of ULVWF strings/bundles and platelet/leukocyte aggregation on endothelial cells under the same conditions. The inhibitory activity of T2C and T5C is dose-dependent. At the concentration of 0.5 µM, the average length of the ULVWF strings and the surface coverage of platelets/leukocytes decrease by ∼50% and ∼40% (p<0.001), respectively. Also, treatment with NEM abolishes the inhibitory effect of T2C and T5C on the ULVWF string formation and platelet/leukocyte aggregation on activated endothelial cells surface (Fig. 1). In conclusion, our results demonstrate for the first time that the distal C-terminal fragments of ADAMTS13 lacking the protease domain are of direct anti-thrombotic activity, which is mediated through its disulfide-bond reduction mechanism. Furthermore, the carboxyl-terminal disulfide bond reducing activity may contribute to the overall anti-thrombotic activity of ADAMTS13 in vivo.
No relevant conflicts of interest to declare.