Abstract
Background: Oprozomib (OPZ) is an oral epoxyketone proteasome inhibitor that selectively and irreversibly binds to its target. Preliminary findings of OPZ in pts with hematologic malignancies (HM) have been reported previously (Savona, ASH 2012, 203; Kaufman, EHA 2013, P223; Ghobrial, ASH 2013, 3184). Updated safety and efficacy results from the subset of pts with MM enrolled in this ongoing phase 1b/2 study in pts with HM are presented.
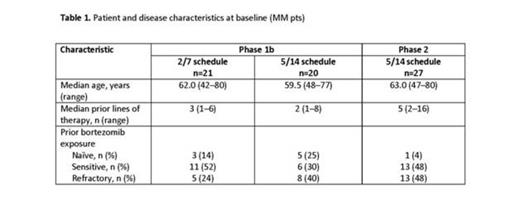
Methods: This open-label, phase 1b/2 study (NCT01416428) is enrolling adult pts with HM who have relapsed after receiving ≥1 line of therapy. The primary objectives (phase 1b portion) are to determine the maximum tolerated dose (MTD) and the safety and tolerability profile of OPZ. The primary objective (phase 2 portion) is to determine the best overall response rate (ORR; ≥PR). In the phase 1b portion, OPZ is being administered once daily on days 1, 2, 8, and 9 of a 14-day cycle (2/7 schedule) or on days 1–5 of a 14-day cycle (5/14 schedule). The starting dose was 150 mg/day (mg/d); doses were escalated in 30-mg increments up to 330 mg/d. In the phase 2 portion, pts are receiving OPZ (240 mg/d) on the 5/14 schedule (initial phase 2 schedule opened to enrollment). For this report, all enrolled patients with HM are included in the description of the MTD while only the subset of patients with WM is included in the safety and efficacy results.
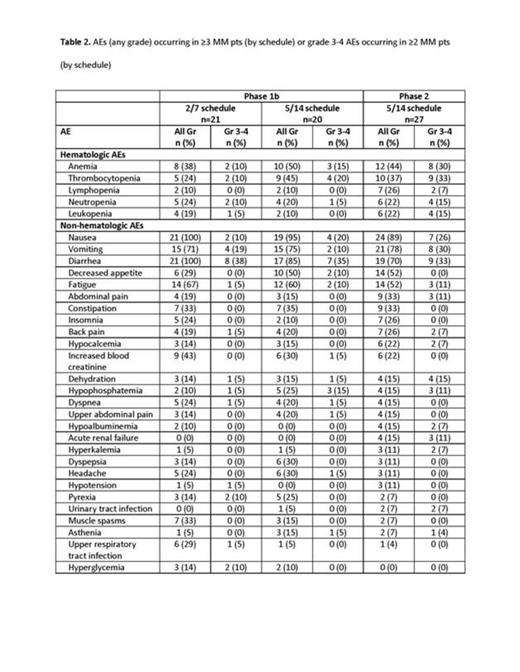
Results: As of July 21, 2014, 106 pts with HM (including 68 pts with MM) were enrolled and treated with the OPZ tablet. Enrollment and baseline demographic information for pts with MM are presented in Table 1. Median treatment duration (phase 1b) was 21.3 weeks (range, 0.3–62.1; 2/7 schedule) and 10.1 weeks (range, 0.3–81.1; 5/14 schedule); preliminary median treatment duration in the ongoing phase 2 portion was 5.4 weeks (range, 0.7–26.7). In all patients with HM, the MTD for the 2/7 schedule was 300 mg/d and 240 mg/d for the 5/14 schedule. Of 3 dose-limiting toxicities (DLTs) on the 2/7 schedule, all 3 DLTs (including hypotension [300 mg/d, n=1], grade 3 diarrhea and grade 4 thrombocytopenia [330 mg/d, n=1 each]) were observed in pts with MM. Of 4 DLTs on the 5/14 schedule, 2 DLTs (grade 3 renal failure [180 mg/d, n=1] and grade 3 tumor lysis syndrome [270 mg/d, n=1]) were observed in pts with MM. The most common adverse events (AEs) in patients with MM are shown in Table 2. Grade 4 AEs included 8 pts (11.8%) with thrombocytopenia, 4 pts (5.9%) with anemia, and 1 pt each (1.5%) with sepsis, leukopenia, decreased platelet count, hyperkalemia, and acute renal failure. Two pts died from upper gastrointestinal bleeding (5/14 schedule, 240 mg/d). One pt (5%) on the 2/7 schedule (phase 1b), 3 pts (15%) on the 5/14 schedule (phase 1b), and 8 pts (30%) on the 5/14 schedule (phase 2) discontinued treatment due to an AE; 6 pts (29%) on the 2/7 schedule (phase 1b), 7 pts (35%) on the 5/14 schedule (phase 1b), and 11 pts (41%) on the 5/14 schedule (phase 2) had their dosage reduced at least once due to an AE. Thirty-four pts in the phase 1b portion were eligible for response. In the phase 1b portion, the ORR in 15 pts on the 2/7 schedule (all were carfilzomib [CFZ]-naïve) was 33.3%; the clinical benefit rate (CBR) was 46.7% (3 very good partial response [VGPR], 2 partial response [PR], and 2 minimal response [MR]). Among 19 pts on the 5/14 schedule (phase 1b portion; all were CFZ-naïve), the ORR was 36.8%; the CBR was 42.1% (1 complete response, 2 VGPR, 4 PR, and 1 MR). In the 5/14 schedule (phase 1b), the ORR among bortezomib [BTZ]-refractory pts (n=7) was 14.3%. On the phase 2 portion (5/14 schedule), the ORR among CFZ-refractory pts (n=11) and CFZ-sensitive pts (n=12) was 27.3% (3 PR) and 33.3% (2 VGPR and 2 PR), respectively; the ORR among BTZ-refractory pts (n=12) was 25.0%.
Conclusions: The MTD of OPZ was 300 mg/d in the 2/7 schedule and 240 mg/d in the 5/14 schedule; these MTDs were determined from all patients with HM. The most common grade 3 AEs were diarrhea, nausea, and vomiting; grade 4 AEs were infrequent. Additional measures will be taken to improve gastrointestinal tolerability. Single-agent OPZ continues to have promising antitumor activity. Enrollment of pts with MM is continuing on the phase 2 study in both treatment schedules. Extended-release OPZ tablets will be introduced and assessed for safety, activity, and pharmacokinetics. Updated results will be presented at the meeting.
Vij:Celgene: Honoraria, Research Funding; Onyx: Honoraria, Research Funding; Sanofi: Honoraria; Jannsen: Honoraria; Novartis: Honoraria; Millennium: Honoraria; Array: Honoraria. Off Label Use: Carfilzomib as treatment in multiple myeloma and solid tumors. Savona:Incyte: Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Consultancy, Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees. Siegel:Celgene: Honoraria, Speakers Bureau; Onyx: Honoraria, Speakers Bureau; Millennium: Honoraria, Speakers Bureau. Kaufman:Millennium: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Onyx: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria; Spectrum: Consultancy, Honoraria; Merck: Research Funding. Ghobrial:BMS: Advisory Board Other; Celgene: Advisory Board, Advisory Board Other; Millennium: Advisory Board, Advisory Board Other; Onyx: Advisory Board, Advisory Board Other. Paner:Celgene Corporation: Honoraria. Jagannath:Millenium: Honoraria; Celgene: Honoraria; Onyx: Honoraria; Merck: Honoraria; Ortho: Membership on an entity's Board of Directors or advisory committees; im: Membership on an entity's Board of Directors or advisory committees; Medicom Worldwide: Membership on an entity's Board of Directors or advisory committees; Optum Health Worldwide: Membership on an entity's Board of Directors or advisory committees; PER group: Membership on an entity's Board of Directors or advisory committees. Jakubowiak:BMS: Advisory Board Other, Consultancy, Honoraria; Celgene: Advisory Board, Speaking, Advisory Board, Speaking Other, Consultancy, Honoraria; Janssen: Advisory Board, Speaking, Advisory Board, Speaking Other, Consultancy, Honoraria; Millennium: Advisory Board, Advisory Board Other, Consultancy, Honoraria; Novartis: Advisory Board Other, Honoraria; Onyx: Advisory Board, Speaking Other, Consultancy, Honoraria; Skyline Dx: Advisory Board Other, Honoraria. Mikhael:Onyx: Research Funding; Celgene: Research Funding; Sanofi: Research Funding; Novartis: Research Funding. Kapoor:Onyx: Membership on an entity's Board of Directors or advisory committees, Research Funding. Neuman:Onyx: Employment. Lee:Onyx Pharmaceuticals, an Amgen subsidiary: Employment.
Author notes
Asterisk with author names denotes non-ASH members.