Abstract
Background: In vivo T-cell depletion with Alemtuzumab reduces the incidence of acute and chronic graft-versus-host disease (GvHD) and graft failure, yet it results in an increase of opportunistic infections and delayed immune recovery. In addition, Alemtuzumab has considerable anti-lymphoma activity in T-cell neoplasms.
We report on a pilot study analysing the safety and feasibility of Alemtuzumab as pre-conditioning prior to reduced intensity conditioning allogeneic stem cell transplantation (RIC-alloSCT) in patients diagnosed with T-cell non-Hodgkin lymphoma.
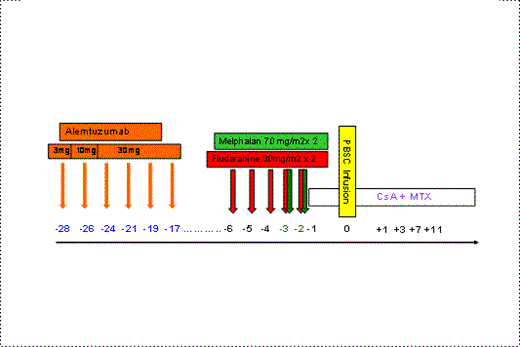
Methods: Six consecutive patients diagnosed with a T-cell lymphoid malignancy underwent RIC-alloSCT from a HLA-identical sibling (n=5) or a matched unrelated donor (n=1). Diagnosis were Angioimmunoblastic T-cell lymphoma (n=3), hepato-splenic gamma-delta T-cell lymphoma (n=1), T/NK cavum lymphoma (n=2) and Sèzary Syndrome (n=1). All received unmanipulated peripheral blood stem cells; At transplantation 2 patients were in CR, and 4 in PR. Median number of prior regimens was 2 (range 2-5). Conditioning regimen included Fludarabine (5 x 30mg/m2) and Melphalan (2 x 70mg/m2) in all cases. GvHD prophylaxis comprised Cyclosporine A and Methotrexate.
The pre-conditioning protocol consisted on 6 escalating doses of intravenous Alemtuzumab starting on day -28 (3 mg on day -28, 10 mg on day -26 followed by four doses of 30 mg on days -24 to -17) given 3 times a week (MWF) on an out-patient basis.
Results: Five patients received all six doses of Alemtuzumab as planned whereas one patient received only four doses for logistic reasons related to donor availability. Acetaminophen, Hydrocortisone and Dexchlorpheniramine were administered as pre-medication in all cases with no severe infusional reaction observed. There were no delays in proceeding to SCT conditioning and patients develop no infectious complications during the Alemtuzumab - alloSCT interval.
All patients engrafted with a median time to neutrophil engraftment of 23 days (range 14-27). Acute skin GvHD (grade I and III) was observed in 2 patients (33.3%), both obtaining a CR with topical and systemic corticosteroids respectively. Two patients developed mild ocular chronic GvHD (one after subsequent infusions of donor lymphocytes - DLI) but none has suffered from extensive chronic GvHD. No opportunistic infection was observed apart from CMV reactivation that developed in 3 out of 4 seropositive patients (no CMV disease was observed). All patients experienced delayed immune reconstitution (CD4 >200/ul) at a median of 321 days (range 229-518). All 4 patients in PR prior to alloSCT achieved a complete remission. Only one patient (diagnosed with Sèzary Syndrome) experienced a relapsed and responded to DLI. With a median follow-up of 42 months, all patients remain alive and in CR.
Conclusions: Our preliminary results suggest that administration of Alemtuzumab prior to reduced intensity conditioning is feasible and safe. In addition to providing T-cell depletion, Alemtuzumab may improve the response rate in T-cell lymphoid malignancies without increasing the risk of infectious complications. These results require further validation in larger phase II studies.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract