Abstract
Background: The systemic histiocytic disorders Erdheim-Chester disease (ECD) and Langerhans cell histiocytosis (LCH) are rare hematologic malignancies with heterogenous clinical courses and prognoses that share a common primary event: the pathologic accumulation and infiltration of cells thought to be of the monocyte/macrophage lineage in affected tissue. The recent discovery of BRAFV600E mutations in a high proportion (50-60%) of patients with LCH/ECD has provided pivotal insight into the cause and potential management of these disorders. The VE-BASKET study (ClinicalTrials.gov identifier, NCT01524978) was designed to explore the efficacy and safety of vemurafenib, a selective oral inhibitor of mutated BRAFV600 kinase, in non–melanoma, non–papillary thyroid cancers harboring BRAFV600 mutations. The preliminary efficacy data in adult patients with ECD/LCH are presented herein.
Methods: Simon 2-stage adaptive-design, open-label, multicenter, multinational, phase 2 study of vemurafenib in patients with cancer harboring BRAFV600 mutations. The primary objective was to evaluate the efficacy of vemurafenib in patients with BRAFV600 mutation–positive cancers at week 8 by investigator-assessed response rate using RECIST version 1.1. Tumor assessment was performed by computed tomography, magnetic resonance imaging, or physical examination every 8 weeks. Patients without RECIST v1.1 measurable disease were followed up by use of positron emission tomography; these response data will be presented separately. Data cutoff was March 18, 2014.
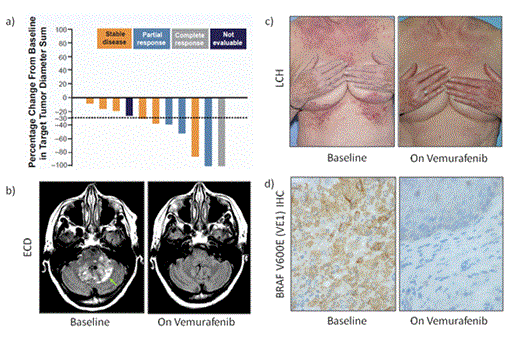
Results: Fifteen patients with ECD/LCH (6 male, 9 female) have been enrolled. Median age is 67 years (range, 35-83). Eleven of 15 patients (73%) received at least 1 prior therapy. Median duration of vemurafenib treatment was 94 days (range, 6-478). Maximal percentage change from baseline in target lesion diameter sum and best overall response are shown in Figure 1A. To date, in 11 patients assessed by RECIST v1.1, overall response rate was 36.4% (95% CI, 10.9-69.2) and clinical benefit rate was 90.9% (95% CI, 58.7-99.8). No patient experienced progression while taking vemurafenib. Best overall responses by RECIST 1.1 are as follows: 1 (9.1%) complete, 3 (27.3%) partial, 6 (54.6%) stable, 0 progressive, and 1 (9.1%) not evaluable. Three patients (20%) discontinued treatment because of adverse events (AEs; grade 3 gastric infection, grade 3 arthralgia, and grade 4 alanine transaminase level elevation); 12 patients are still receiving treatment. Thirteen patients (87%) had at least 1 grade 3 or 4 AE—most commonly skin squamous cell carcinoma (40%)—and actinic keratosis, keratoacanthoma, maculopapular rash, arthralgia, and dehydration (each 13%). No patient died during the study. Two representative patient vignettes are presented herein. Patient 1 is a 68-year-old woman with extensive ECD involving the brain, bones, and retroperitoneum. She previously received high-dose methotrexate and underwent stenting for disease-related renal artery stenosis that resulted in an emergency hypertensive event and renal failure, transiently requiring hemodialysis. At the time of enrollment, this patient had severe gait instability and dysarthria and was unable to perform activities of daily living (ADLs). After 8 weeks of vemurafenib therapy, she achieved partial response (Figure 1B), is ambulating with minimal assistance, and is independent with ADLs. She continues therapy. Patient 2 is a 77-year-old woman with LCH previously treated with vinblastine, methotrexate, an AKT inhibitor, and cladribine. At the time of enrollment she had debilitating cutaneous involvement that required long-term administration of narcotics and resulted in recurrent infection. After 4 weeks of vemurafenib therapy, the patient achieved complete response, including total resolution of skin lesions (Figure 1C). Subsequent biopsy also confirmed complete pathologic response and complete absence of the BRAFV600E mutant protein by immunohistochemistry using the VE1 antibody (Figure 1D). The patient continues therapy.
Conclusion: There are no approved therapies for adult patients with multisystem histiocytic disorders. The magnitude and durability of response to vemurafenib in patients with multisystemic BRAFV600-mutated ECD and LCH are encouraging. Updated results will be presented at the American Society of Hematology annual meeting.
Hyman:Chugai Pharma: Consultancy; Atara Biotherapeutics: Consultancy. Off Label Use: Vemurafenib is a BRAF inhibitor currently approved for treatment of patients with unresectable or metastatic melanoma with BRAF V600E mutation as detected by an FDA-approved test. The data included in this abstract is a subgroup analysis of VE-BASKET, a phase 2 study of vemurafenib in non-melanoma cancer patients harboring BRAFV600 mutations in order to explore the efficacy of vemurafenib in other cancer populations. Subbiah:MD Anderson Cancer Center: Employment. Blay:University Lyon I: Employment; Centre Leon Berard: Employment. Sirzen:F. Hoffmann-La Roche: Employment, Equity Ownership. Veronese:F. Hoffmann-La Roche: Employment. Laserre:F. Hoffmann-La Roche: Employment. Baselga:Roche: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.