Abstract
Background
Myelofibrosis (MF) is a clonal myeloproliferative neoplasm (MPN) associated with a high degree of symptomatology, progressive cytopenias and potential to transform into acute myelogenous leukemia (AML). Thrombocytopenia amongst MF patients is a proven negative prognostic indicator and predictor of transformation to AML. Ruxolitinib is an effective JAK inhibitor for MF symptoms and splenomegaly, but is not indicated in patients with severe thrombocytopenia. Phase III trials of pacritinib have shown alleviation of the MF symptom burden amongst patients with thrombocytopenia (ASCO 2015 Mesa et. al.). In this study, we assessed the symptom burden of MF patients with significant thrombocytopenia who were naïve to pacritinib.
Methods
Data was assessed from a prospectively collected international database of MF patients in which demographics, disease features, and MF symptoms utilizing the myeloproliferative neoplasm symptom assessment form (MPN-SAF; Scherber et al, 2011). The MPN-SAF includes the patient's perceptions of common MPN-related symptoms and overall quality of life (QOL) on a 0 (absent) to 10 (worst imaginable) scale. Total symptom score (TSS) was computed based on symptom items using the published scoring algorithm on a 0 (all reported symptoms absent) to 100 (all reported symptoms worst imaginable) scale. MF risk scores were calculated using the DIPSS criteria (Gangat, 2011). Thrombocytopenia was defined as a platelet count <100 x 10(9)/L, anemia was defined as hemoglobin<10 g/dL and leukopenia was defined as a white blood cell count <4.0 x 10(9)/L. Associations between the MPN-SAF individual symptoms were investigated using Pearson correlations.
Results
Demographic and Disease Features:
A total of 418 patients with (n=89) and without (n=329) thrombocytopenia completed the MPN-SAF. Patients with thrombocytopenia were slightly younger (57.4 vs. 61.0, p=0.01) with longer disease durations (11.9 years vs. 8.8 years, p=0.0489) and had a higher prevalence of primary myelofibrosis (PMF; 82% vs. 66%, p=0.01). The presence of thrombocytopenia was associated with other laboratory abnormalities including anemia (60.7% vs. 25.3%, p<0.001) and leukopenia (34.8% vs. 52.3%, p<0.001), along with the need for red blood cell transfusions (34.8% vs. 14.6%, p<0.001). Patient cohorts did not differ by gender, DIPSS risk score, history of prior thrombosis or hemorrhage. In comparing patients with severe thrombocytopenia (<50 x 10(9)/L, n=43) to those with moderate thrombocytopenia (51-100 x 10(9)/L, n=46), severe thrombocytopenia patients were more likely to have anemia (74.4% vs. 47.8%, p=0.01) and require red blood cell transfusions (51.2% vs. 19.6%, p=0.002).
Symptoms
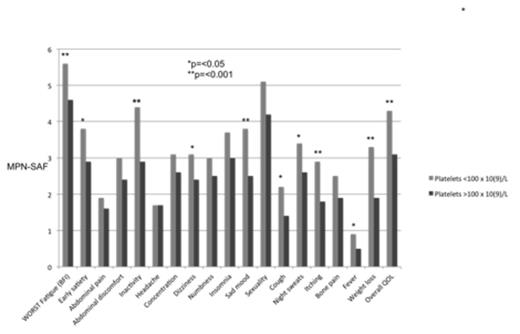
Scores for individual MPN-SAF items were assessed for each subgroup. Patients with thrombocytopenia had markedly higher total symptom scores than patients without thrombocytopenia (32.8 vs. 24.1, p<0.001). Individual scores were also higher for most items (fatigue, early satiety, inactivity, dizziness, sad mood, sexuality, cough, night sweats, itching, fever, weight loss, overall QOL)[Figure 1]. No significant differences in MPN SAF TSS or individual symptom scores were observed in comparing severe vs. moderate thrombocytopenia patients.
Discussion
MF patients with thrombocytopenia have distinctive clinical characteristics and face a significantly more severe symptom burden. Importantly, despite thrombocytopenia being a recognized risk factor for disease advancement, no correlations are noted between patient symptomatology and risk category. In addition, patients with severe thrombocytopenia do not differ symptomatically from patients with moderate thrombocytopenia despite having more severe anemia, leukopenia and transfusion requirements. This implies that the degree of symptomatology expressed by thrombocytopenic MF patients occurs independent from the exact platelet value.
Conclusion
The results of this study suggest that patients with thrombocytopenia will benefit from aggressive symptomatic control, potentially from targeted agents.
MF Symptoms in Patients With and Without Thrombocytopenia
MF Symptoms in Patients With and Without Thrombocytopenia
Kiladjian:Novartis: Consultancy; Novartis: Other: Travel grant; Research Funding paid to institution (Hôpital Saint-Louis et Université Paris Diderot); Incyte Corporation: Consultancy. Zweegman:Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding. Harrison:Shire: Speakers Bureau; CTI Biopharma: Consultancy, Honoraria, Speakers Bureau; Sanofi: Honoraria, Speakers Bureau; Gilead: Honoraria; Novartis: Honoraria, Research Funding, Speakers Bureau. Cervantes:Novartis: Consultancy, Speakers Bureau; Sanofi-Aventis: Consultancy; CTI-Baxter: Consultancy, Speakers Bureau. Barbui:Novartis: Speakers Bureau. Etienne:ARIAD: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Other: Congress Travel/Accomodations, Research Funding, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau. Roy:BMS: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Vannucchi:Shire: Speakers Bureau; Novartis Pharmaceuticals Corporation: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Baxalta: Membership on an entity's Board of Directors or advisory committees. Mesa:Novartis. Research- incyte, Gilead, cti, Genentech, promedior, NS Pharma: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.