Abstract
Rationale
Autologous stem cell transplantation for adult acutemyelogenousleukemia (AML) is a therapeutic option for good and intermediate risk patients. New compounds and new formulations in conjunction with pre andpost transplant targeted therapy may improve outcome.
A recent EBMT retrospective survey investigating on Busulfan administered IV (Nagler A et al:Haematologica. 2014 ) pre autologous stem cell transplantation (ASCT) suggested that the combination of Busulfan and High dose Melphalan (BUMEL) was associated with the best overall survival. To address this question, we compared this regimen with the historical Busulfanand Cyclophosphamide (BUCY) association.
Methods
Eligibilitycriteria consistedof all adult patients (age >18 years) with AMLautograftedin CR1, following a preparative regimen consisting of either BUCY or BUMEL, reported to the EBMT registry in the period from January 2005 to December 2013. The doses of Busulfan were either oral at 16 mg/kg over 4 days or IV at 12.8 mg/kg over 4 days. High dose Melphalan was 140mg/m². The data came from 231 centers 23 ofwhich usedboth regimens. The relationships between outcomes and patient, disease and transplant characteristics was evaluated in multivariate analyses using the Cox model.
Results
1120 patients received BUCY and 436 BUMEL prior to autografting in first remission. Patients receiving BUMEL were older (median 52 years versus 50 years, p=0.01) with less good risk diseases by cytogenetics (16% versus 28%, p=0.001). They were autografted with a longer interval from CR1 to autografting (120 days versus 104 days, p=0.001) and they were more frequently in a negative minimal residual disease (MRD) status by molecular biology (89% versus 82%, p= 0.03).
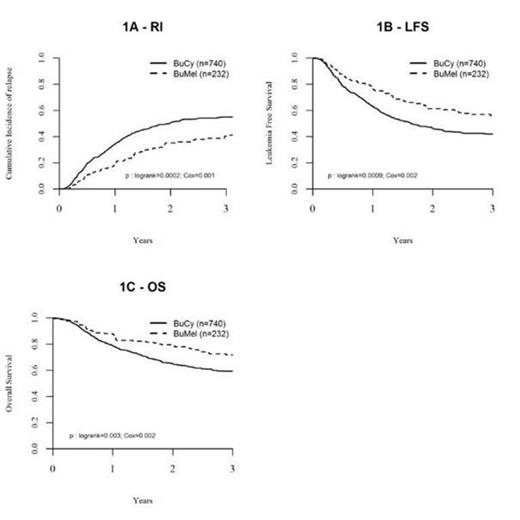
The nature of the pretransplant regimen interacted with the interval from diagnosis to autografting on both relapse incidence (p=0.005) and leukemia free survival (p= 0.02): the outcome post BUMEL was superior to BUCY only in patients transplanted within less than six months post diagnosis. In these patients, following BUMEL and BUCY respectively, the RI was 35%versus 50.6% (p< 10-3), the LFS 61.1% versus 46.2% (p< 10-3) and the OS 79.4% versus 64.8% (p= 0.003). There was no difference for NRM.
When considering only patients autografted at the time when MRD detection was negative, the LFS at two years was 75.8% for 134 patients receiving BUMEL and 58.6% in the 371 patients receiving BUCY, with no significant difference (p= 0.1). The major cause of death in the two groups was leukemia recurrence.
Conclusion
We conclude that the BUMEL combination pre ASCT in AML CR1 is superior to BUCY when the transplant is performed less than 6 months after diagnosis. These registry data should be confirmed in a well designed two arm study assessing the role of ASCT in good risk and possibly intermediate -1 risk adult patients with AML.
| . | . | p . | HR . | 95% CI . | |
|---|---|---|---|---|---|
| NRM | BUMEL vs BUCY | 0.450 | 1.376 | 0.599 | 3.16 |
| Age atTx/ 10years | 0.001 | 1.064 | 1.025 | 1.10 | |
| Time from diagnosis to Transplant. | 0.900 | 0.999 | 0.984 | 1.01 | |
| Cytogenetics :Intermediatevs good | 0.610 | 0.709 | 0.191 | 2.63 | |
| Poor versus good | 0.710 | 0.708 | 0.112 | 4.46 | |
| Missing | 0.420 | 0.591 | 0.164 | 2.11 | |
| centre (frailty) | 0.360 | ||||
| RI | BUMEL vs BUCY | 0.001 | 0.602 | 0.445 | 0.816 |
| Age at Transplant / 10years | 0.005 | 1.012 | 1.003 | 1.020 | |
| Time from diagnosis to Transplant: | 0.720 | 1.000 | 0.996 | 1.004 | |
| Cytogenetics : Intermediatevs good | 0.510 | 1.145 | 0.764 | 1.714 | |
| Poor versus good | 0.031 | 1.799 | 1.055 | 3.067 | |
| Missingversus good | 0.480 | 1.150 | 0.779 | 1.699 | |
| centre (frailty) | 0.110 | ||||
| OS | BUMEL vs BUCY | 0.002 | 0.614 | 0.449 | 0.841 |
| Age at Transplant / 10years | <10-4 | 1.025 | 1.014 | 1.036 | |
| Time from diagnosis toransplant | 0.94 | 0.999 | 0.995 | 1.004 | |
| Intermediatevs good | 0.88 | 1.036 | 0.649 | 1.653 | |
| Poor versus good | 0.11 | 1.632 | 0.901 | 2.956 | |
| Missingversus good | 0.81 | 1.058 | 0.672 | 1.665 | |
| centre (frailty) | 0.29 | ||||
| LFS | BUMEL vs BUCY | 0.002 | 0.649 | 0.490 | 0.858 |
| Age at Transplant / 10years | <10-4 | 1.015 | 1.006 | 1.023 | |
| Time from diagnosis to Transplant | 0.740 | 1.000 | 0.996 | 1.004 | |
| Cytogenetics : Intermediatevs good | 0.590 | 1.110 | 0.755 | 1.630 | |
| Poor vs good | 0.046 | 1.679 | 1.008 | 2.796 | |
| Missingversus good | 0.590 | 1.106 | 0.763 | 1.604 | |
| centre (frailty) | 0.170 | ||||
| . | . | p . | HR . | 95% CI . | |
|---|---|---|---|---|---|
| NRM | BUMEL vs BUCY | 0.450 | 1.376 | 0.599 | 3.16 |
| Age atTx/ 10years | 0.001 | 1.064 | 1.025 | 1.10 | |
| Time from diagnosis to Transplant. | 0.900 | 0.999 | 0.984 | 1.01 | |
| Cytogenetics :Intermediatevs good | 0.610 | 0.709 | 0.191 | 2.63 | |
| Poor versus good | 0.710 | 0.708 | 0.112 | 4.46 | |
| Missing | 0.420 | 0.591 | 0.164 | 2.11 | |
| centre (frailty) | 0.360 | ||||
| RI | BUMEL vs BUCY | 0.001 | 0.602 | 0.445 | 0.816 |
| Age at Transplant / 10years | 0.005 | 1.012 | 1.003 | 1.020 | |
| Time from diagnosis to Transplant: | 0.720 | 1.000 | 0.996 | 1.004 | |
| Cytogenetics : Intermediatevs good | 0.510 | 1.145 | 0.764 | 1.714 | |
| Poor versus good | 0.031 | 1.799 | 1.055 | 3.067 | |
| Missingversus good | 0.480 | 1.150 | 0.779 | 1.699 | |
| centre (frailty) | 0.110 | ||||
| OS | BUMEL vs BUCY | 0.002 | 0.614 | 0.449 | 0.841 |
| Age at Transplant / 10years | <10-4 | 1.025 | 1.014 | 1.036 | |
| Time from diagnosis toransplant | 0.94 | 0.999 | 0.995 | 1.004 | |
| Intermediatevs good | 0.88 | 1.036 | 0.649 | 1.653 | |
| Poor versus good | 0.11 | 1.632 | 0.901 | 2.956 | |
| Missingversus good | 0.81 | 1.058 | 0.672 | 1.665 | |
| centre (frailty) | 0.29 | ||||
| LFS | BUMEL vs BUCY | 0.002 | 0.649 | 0.490 | 0.858 |
| Age at Transplant / 10years | <10-4 | 1.015 | 1.006 | 1.023 | |
| Time from diagnosis to Transplant | 0.740 | 1.000 | 0.996 | 1.004 | |
| Cytogenetics : Intermediatevs good | 0.590 | 1.110 | 0.755 | 1.630 | |
| Poor vs good | 0.046 | 1.679 | 1.008 | 2.796 | |
| Missingversus good | 0.590 | 1.106 | 0.763 | 1.604 | |
| centre (frailty) | 0.170 | ||||
Multivariate analyses of prognostic factors in patients autografted within 6 months
Leukemia free survival and overall survival of patients with AMLautograftedwithin 6 months from initial diagnosis, in first remission following high dose consolidation by BUMEL or BUCY in the period fromfromJanuary 2005 to December 2013 (EBMT registry).
Leukemia free survival and overall survival of patients with AMLautograftedwithin 6 months from initial diagnosis, in first remission following high dose consolidation by BUMEL or BUCY in the period fromfromJanuary 2005 to December 2013 (EBMT registry).
Nemet:Celgene: Honoraria; Pliva: Honoraria; Amgen: Honoraria; Sanofi: Honoraria; Pfizer: Honoraria; Janssen: Honoraria. Mohty:Janssen: Honoraria; Celgene: Honoraria. Nagler:Biokine LTD: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.