Abstract
BACKGROUND:
Treatment may be safely deferred in asymptomatic patients with advanced stage, low tumor burden follicular lymphoma (FL) until onset of symptoms or organ failure without compromising overall survival (OS). A randomized trial comparing immediate rituximab vs. observation in this setting reported a Time to New Treatment benefit without a corresponding OS benefit (Ardeshna et al, Lancet Oncol 2014). This is a comparison of Time to 1st Treatment (TT1T, observation arm) vs. Time to 2nd Treatment (TT2T, rituximab arm) and thus biased to immediate intervention.
TT2T might be a less biased primary endpoint for trials evaluating immediate intervention vs. observation. Time to Treatment n (TT(n)T) is a function of cumulative time spent from diagnosis in periods of observation, treatment, and remission, and TT(n)T approaches OS as n approaches the maximum number of therapies received, thus we also hypothesize that TT2T might be a surrogate for OS.
We have therefore performed a comprehensive evaluation of standard endpoints (TT1T, OS, and EFS12), described TT2T as a novel endpoint for trials evaluating intervention vs. observation in asymptomatic patients with advanced stage, low tumor burden FL, and aimed to determine if TT2T is a reliable surrogate for OS.
METHODS:
We identified 246 consecutive patients at our institution, diagnosed from 1998 to 2007 with advanced stage, low tumor burden follicular lymphoma grade 1-3A, for whom physician intention at diagnosis was observation.
Median time to event was calculated using the Kaplan-Meier method for each event: TT1T, TT2T, and OS. Modified Kendall's tau was used to assess the correlation between TT1T, TT2T, and OS, accounting for censoring in these quantities. Tests of modified Kendall's tau against 0 (i.e. no correlation) were performed.
EFS12 was defined as non-death event within 12 months of initiation of first treatment, and median OS for EFS12 analysis was calculated from end of first treatment. Of 69 patients who received chemoimmunotherapy as first therapy following initial observation, the log-rank test was used to compare survival distributions by EFS12.
RESULTS:
Of 246 patients with advanced stage, follicular lymphoma grade 1-3A for whom physician intention at diagnosis was observation, there was a slight female predominance (1.16:1), 34.6% were above age 60 with a median age of 56.1 years (range 25-88), and 11.8% (29/246) developed histologic transformation prior to 1st/2nd therapy, including 7.7% (19/246) prior to 1st treatment.
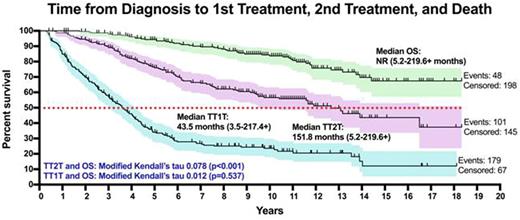
At a median follow-up of 10.9 years: median TT1T was 43.5 months (3.5-217.4+, 179 events), median TT2T was 151.8 months (range 5.2-219.6+, 101 events), and median OS was not reached (range 5.2-219.6+, 48 events). The modified Kendall's tau measuring correlation between TT1T and OS was 0.012 (p = 0.537), and was 0.078 (p < 0.001) between TT2T and OS, suggesting that TT2T is strongly correlated with OS while TT1T is not.
Failure to achieve EFS12 was observed in 10.3% (7/68) and associated with inferior OS (p=0.001). Of 7 patients who failed to achieve EFS12, 3 had histologic transformation.
CONCLUSIONS:
TT2T is a preferred primary endpoint for clinical trials evaluating intervention vs. observation. In patients with advanced stage, low tumor burden FL who are initially observed, the median time from diagnosis to 2nd treatment is 12.7 years. Future trials evaluating the role of immediate therapy in low tumor burden FL might restrict eligibility to high risk patients expected to have inferior TT2T. Efforts are ongoing to develop biomarkers (e.g. m7-FLIPI) that identify a population enriched with high risk patients. Our data also suggest that TT2T might be a better surrogate for OS than TT1T, and analyses are ongoing to validate this finding.
Hamlin:Molecular Templates: Research Funding; Seattle Genetics: Research Funding; Portola: Research Funding; Gilead: Membership on an entity's Board of Directors or advisory committees; Novartis: Research Funding; Xencor: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees. Horwitz:Spectrum: Consultancy, Research Funding; Bristol-Myers Squibb: Consultancy; Huya: Consultancy; Infinity: Consultancy, Research Funding; Kyowa Hakka Kirin: Consultancy, Research Funding; Takeda: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; Celgene: Consultancy; ADCT Therapeutics: Research Funding. Palomba:Pharmacyclics: Consultancy. Moskowitz:Merck: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol-Myers Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees, Research Funding. Noy:Pharmacyclics, LLC, an AbbVie Company: Other: travel, accommodations, expenses, Research Funding. Kumar:Celgene: Research Funding; Pharmacyclics: Research Funding; Adaptive Biotechnologies: Research Funding; Seattle Genetics: Research Funding; Celgene: Honoraria, Other: Scientific Advisory Board. Zelenetz:Amgen: Consultancy; Takeda Pharma: Consultancy; Novartis: Consultancy; Nanostring Tech: Consultancy; Portola Pharmaceuticals: Consultancy; Adaptive Biotechnology: Consultancy; Bristol Myers: Research Funding; Boehringer Ingelheim: Other: DMC Membership; Janssen: Consultancy, Research Funding; Hospira: Consultancy; Celgene: Consultancy; Gilead: Consultancy, Research Funding; GSK: Consultancy, Research Funding; Genentech/Roche: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.