Abstract
Introduction
With standard intensive induction regimens, up to 80% of Acute Myeloid leukemia (AML) patients can achieve complete remission (CR). Several evidences demonstrated that the persistence of detectable disease (MRD) assessed with highly sensitive techniques such as Multicolor-Flow-Cytometry (MFC) and PCR based molecular analysis, retains a prognostic value among patients achieving morphological remission (Walter RB, 2015; Araki D et al, 2016, Zhou Y et al, 2016).
The aim of the present study was to retrospectively evaluate the prognostic impact of MRD in a cohort of uniformly treated AML patients.
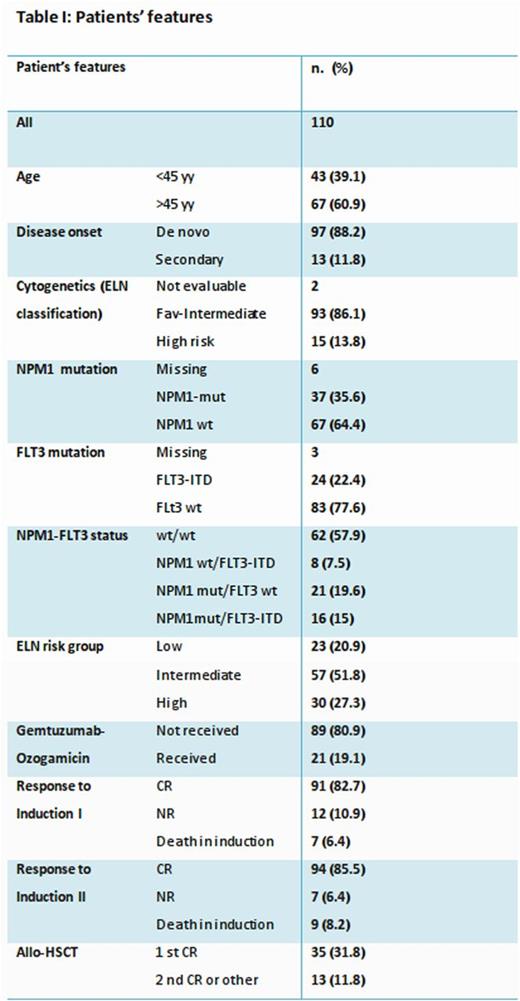
One hundred and ten consecutive AML patients who had been treated in our center between January 2004 and December 2014 were retrospectively analyzed. All patients had received a fludarabine-containing induction (FLAI-5) and received second cycle and further consolidation therapy according to our published strategy (Guolo F, 2016). Median age was 47 years (range 17-61). Median follow up was 59 months. Patients features are summarized in Table I.
MRD assessment was performed through 4-colour MFC analysis (MFC-MRD)and through WT1-gene expression analysis, as previously described (Guolo F, 2016). Three different MRD time-points (TP)were considered: TP1, after induction I; TP2, after induction II; TP3, after consolidation therapy for patients who did not undergo HSCT and at HSCT for patients who underwent HSCT. Relapse-free survival (RFS) was calculated from the time of diagnosis until last follow-up or documented leukemic relapse.
CR rate after 1st and 2ndinduction was 82.7 and 85.5%, respectively, whereas 30 and 60 days mortality was 6.4% and 8.2%, respectively.
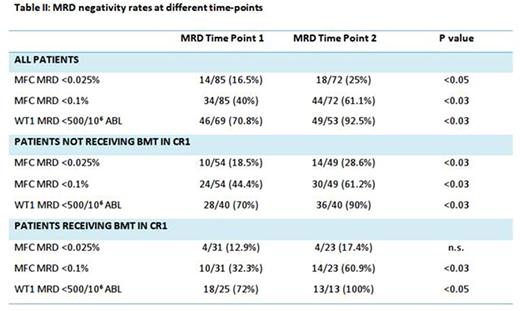
Overall, patients showed MRD reduction from TP1 to TP2. Detailed MRD negativity rates are provided in table II. MRD clearance probability was significantly influenced only by ELN risk group and Karyotype (p <0.05 for both).
In the whole cohort, 2 years RFS was 62.4% (median not reached). RFS duration was significantly influenced by secondary disease (p <0.001), NPM mutation (p <0.05), ELN risk group (p <0.001), WT1 expression level at diagnosis (p<0.03) and MFC MRD positivity, at any time-point with both <0.025% and <0.1% as cut-off for positivity (p<0.03 and 0.05, respectively, for MRD TP1 and p<0.05 and <0.005, respectively, for MRD TP2, Figure 1), WT1 MRD at TP1 (p<0.05). Multivariate Cox-Proportional Hazard model showed that MFC MRD <0.1% at TP2 was the strongest predictor of DFS.
Overall survival (OS) analysis led to similar results.
However, at TP1, when performing MFC-MRD evaluation with a "Leukemia-associated immunophenotype" (LAP) approach, the choice of the deeper 0.025% cut-off for positivity identified patients at lower risk of relapse (2-year RFS of 88.9%) whereas patients with MFC MRD >0.1% had an high probability of relapse, with a 2-year RFS of only 21.5%.
Moreover, the combination of MFC and WT1- MRD assessment identified three subgroups of patients with significantly different outcome (2-years OS 100, 64.4 and 46.7%, respectively, for MFC-neg/WT1 neg, MFC-pos/WT1 neg, MFC-pos/WT1 pos), as we previously described (Marani C, 2013).
Consolidation chemotherapy with High Dose Ara-C was able to improve prognosis and increase MRD TP3 negativity rate only in patients not scheduled for HSCT in 1st CR and only if at least 2cycles were administered. The positive impact was higher if MFC MRD at TP2 was <0.1%.
Conclusions
Our data clearly show that the quantification of MFC-MRD assessment at different time-points during treatment retains a strong prognostic impact in AML and can improve patients-risk stratification.
In our experience the established threshold of 0.1% to define MFC negativity is confirmed as the most useful in post induction program setting. However, performing a MFC-MRD evaluation with LAP approach, choosing a deeper cut-off value can discriminate patients with a significantly lower risk of relapse. Prospective randomized trials evaluating the prognostic impact of MRD-driven therapeutic decisions are strongly needed.
Gobbi:Takeda: Consultancy; Janssen: Consultancy, Honoraria; Gilead: Honoraria; Celgene: Consultancy; Roche: Honoraria; Novartis: Consultancy, Research Funding; Mundipharma: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.