Abstract
Background. Chronic myeloid leukemia (CML) treatment is based on company-sponsored and academic trials testing different tyrosine kinase inhibitors (TKIs) as first-line therapies. However these clinical trials included patients selected according to different inclusion and exclusion criteria, particularly age and comorbidities, and with specific treatment obligations. In daily clinical practice (real life), no inclusion and exclusion criteria do exist, and treatment outcomes depend not only on choice of the first-line TKI, but also on doctor- and patient-choice of second- and third-line TKIs.
Aims. To investigate the response and the outcome on first-line imatinib, with switch to 2nd generation TKIs in case of failure or toxicity, in BCR-ABL1+ CML patients enrolled in a population-based registry (real-life setting).
Methods. We report here the results from a prospective study, enrolling all newly diagnosed patients (N = 236, median age 60 years) living in two Italian regions, that were registered according to population-based criteria and treated front-line with imatinib. The decision to switch to second-line treatment was up to the Local Investigator (no predefined criteria) Definitions: major molecular response (MMR or MR3.0): BCR-ABL1IS ratio <0.1%; deep molecular response (MR4.0): BCR-ABL1IS ratio <0.01% with >10,000 ABL1 copies; failure and suboptimal response: according to 2009 European LeukemiaNet (ELN) criteria; progression: transformation to advanced phases according to ELN criteria; leukemia-related death (LRD): death after progression.
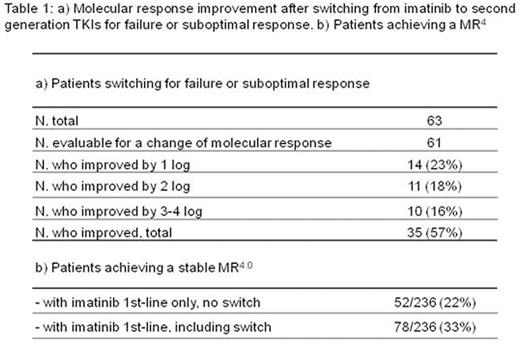
Results. Median age was 60 years at diagnosis and 64 years at last contact, with a median follow up of 48 months (range 2-86 months). High risk patients according to Sokal score and new EUTOS long-term survival (ELTS) score were 23% and 18%, respectively. A switch from imatinib to a second generation TKI was reported in 14% of patients for side-effects, and in 24% for failure or suboptimal response, with an improvement of molecular response in 57% of them. The median time to switch was 8 months for toxicity and 20 months for failure. At 12 months, 55% of all evaluable patients were in MR3.0 or better. At last contact, 75% of all evaluable patients were in MR3.0 or better and 48% were in MR4.0 or better. The molecular response of patients switching for failure or suboptimal response was evaluable in 61/63 patients: improved in 57%, not modified in 38%, worsened by at least 1 log in 5% of patients. Overall, the treatment switch was associated with a remarkable increase of MR3.0 rate: from 10% at the time of switching to 52% at last qPCR. Overall, 33% achieved a stable MR4.0, of whom 22% with imatinib alone, and 11% after switching to a second generation TKI (Table 1). The 5-year overall survival (OS) and leukemia-related survival (LRS) were 85% and 93%, respectively. Twenty-seven patients (11%) died, of whom 13 (6%) of leukemia, after disease progression and 14 (6%) of "other" causes, without any evidence of progression. Cardiovascular complications were reported in 4% of patients treated with imatinib alone and in 6% of patients who received nilotinib in second-line. OS was not affected by age in the interval between 18 and 69 years (5-year OS 92%, ranging from 89% to 95 % in the different decades), but 5-year OS probabilities of elderly (70-79 years) and very elderly patients (≥ 80 years) were significantly lower (78%, P = 0.003, and 34%, P < 0.0001, respectively). The probability of LRS was not affected by age. The Sokal score predicted OS, but not LRS. The ELTS score clearly separated low, intermediate, and high risk patients for OS and also for LRS.
Conclusions. The results of this prospective population-based study emphasize the importance of second-line treatment in the clinical practice: the CML management with the 3 available TKIs in the period 2008-2012, according to current treatment recommendations for switching and without any predefined obligation, resulted in response rates and outcomes, that are in the high range of prospective interventional studies investigating the efficacy and the safety of a single TKI in selected patients. This study provides a robust dataset on the results of CML treatment with TKI in clinical practice, that will be important for interpreting and evaluating the results of the next prospective studies, arguably limited to selected patients.
Castagnetti:Bristol-Myers Squibb: Consultancy, Honoraria; ARIAD Pharmaceuticals: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Gugliotta:Bristol Myers Squibb: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Marasca:Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria; Roche: Honoraria. Soverini:Novartis: Consultancy; Bristol-Myers Squibb: Consultancy; Ariad: Consultancy. Martinelli:Genentech: Consultancy; ARIAD: Consultancy; Roche: Consultancy; Bristol-Myers Squibb: Speakers Bureau; Novartis: Speakers Bureau; MSD: Consultancy; Pfizer: Consultancy, Speakers Bureau; Amgen: Consultancy. Cavo:Bristol-Myers Squibb: Honoraria; Amgen: Honoraria; Takeda: Honoraria; Celgene: Honoraria, Research Funding; Janssen: Honoraria, Research Funding. Rosti:Novartis: Honoraria, Research Funding, Speakers Bureau; Pfizer: Honoraria, Research Funding, Speakers Bureau; Roche: Honoraria, Research Funding, Speakers Bureau; Incyte: Honoraria, Research Funding, Speakers Bureau; BMS: Honoraria, Research Funding, Speakers Bureau. Baccarani:Ariad: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau; Pfizer: Consultancy, Honoraria, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.