Abstract
Background:High dose therapy supported by autologous stem cell transplantation (HDT/ASCT) has been a treatment frequently indicated in follicular lymphoma (FL) patients and has contributed to modify the natural history of the disease. Secondary neoplasia is one of the concerns after HDT/ASCT, although its incidence after transplant-free treatments is considerable (RummelM et al. Lancet Oncol 2016; Sacchi S et al. Haematologica 2008). The high incidence of secondary neoplasia in some of the studies, together with the lack of OS advantage in transplanted patients compared to those treated with conventional Chemo/R, has led to abandon HDT/ASCT by many groups. However, its incidence is very variable among studies due in part to cohort different in terms of age, pre-ASCT treatments, used of TBI-based or TBI-free conditioning regimen or length of follow-up.
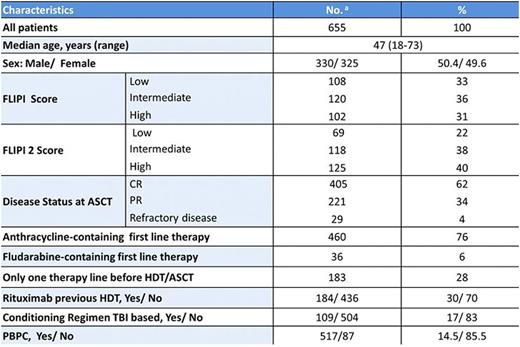
Methods:The Incidence of secondary neoplasia, the factors associated with its development and the survival of 655 FL patients reported to the Spanish GELTAMO registry and intensified with HDT/ASCT between 1989 and 2007 with a median follow-up of 12.3 years from HDT/ASCT and 14.5 years from diagnosis were analyzed. Baseline characteristics and therapeutic-related data are listed in the table. Standardized Incidence Ratios (SIR) were calculated to assess the risk of a second malignancy by dividing the number of observed second malignancies with the number of expected sex matched incidence using the 2008 crudes rates in the Spanish population (J Farley et al. EJC 2013). Data were updated with a cut-off date of 31 June 2016.
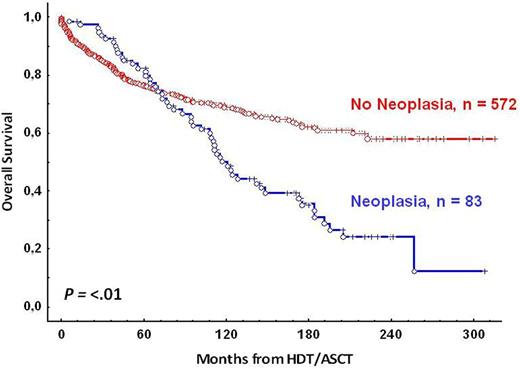
Results:Median OS were 21.3 years from HDT/ASCT and 22.6 years from the time of FL diagnosis. A total of 83 patients (12.75%) developed 85 second malignancies. There were 45 cases of solid tumors (51%), including 8 skin cancers; 34 cases of Acute Myeloid Leukemia or Myelodysplastic Syndromes (AML/MDS) (40%); 2 Acute Lymphoblastic Leukemia (ALL), 1 chronic myeloid leukemia (CML), 1 Hodgkin lymphoma (HL) and 2 cases of other cancers. The accumulated incidence at 5, 10 and 15 years were 6.7%, 12% and 17.8%, respectively. The incidence for solid tumors and AML/SMD were 2.1%, 4.1%, 9.5% and 3.1%,6.4% and 8,1% at 5, 10 and 15 years, respectively. Median time from HDT/AST to the diagnosis of the second malignancy was 5.5 years (IQR 3.2-9.6). Solid tumors and AML/SMD were documented with a median time of occurrence since HDT/ASCT 7.7 years (IQR 3.2-9.9) and 4.2 years (IQR 1.7-7.3), respectively. The SIR for second neoplasia was 2.8 (CI 95%: 2.6-2.9) and only 1.4 (CI 95% 1.3-16) in the case of solid tumors. Only male sex (P=.006) and the use of anthracycline at first line therapy in the case of solid tumors (P=.02) were associated with an increased number of second neoplasia. Older age and a status of disease before HDT/ASCT different to complete response showed a tendency. There were no differences according to the use of fludarabine or rituximab previously to HDT/ASCT, number of therapy lines before HDT/ASCT, time from diagnosis to HDT/ASCT or conditioning regimen. Median OS for patients with second neoplasia is 12 years from the time of FL diagnosis, 9.4 years from ASCT (fig 2)[14.5 y. for solid tumors and 8 y. for sMDS/sAML (P=.01)] and 1.5 years from the time of diagnosis of second neoplasia [2.3 y. for solid tumors and 1.25 y. for sMDS/sAML (P=.01)], respectively
Conclusion: This very long follow-up study, that includes patients from the rituximab era, indicates that FL patients undergoing an ASCT are at an increased risk of developing a 2nd malignancy, especially AML-MDS. However, the incidence is not higher than the reported in other series without transplantation. Only male sex and the use of anthracyclines were associated with an increased risk of 2nd neoplasia. Given the favorable survival obtained by ASCT in FL, the risk of secondary neoplasia shouldn't preclude its application. However, once a neoplasia is diagnosed the prognosis is dismal. Thus, a carefully selection of patients candidates to HDT/ASCT is necessary.
Lopez-Jimenez:Abbvie: Membership on an entity's Board of Directors or advisory committees; Velgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Roche: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; MSD: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; MundiPharma: Honoraria, Membership on an entity's Board of Directors or advisory committees. Martín:Sevier: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.