Abstract
Introduction: Current recommendations for the treatment of AML in newly diagnosed elderly patients (≥ 65 years) include different therapeutic options (intensive or semi-intensive chemotherapy, low-dose chemotherapy, hypomethylating agents, and supportive care). Regardless of the selected treatment, the results are disappointing because of low overall survival (OS) rates and significant toxicity.
Objectives: The primary objective of FLUGAZA trial is to compare the 1-year-OS in 350 patients aged ≥ 65 years diagnosed with AML assigned to azacytidine (n=175) or FLUGA (n=175) arms. Here we present the results of a pre-planned interim safety analysis for both outpatient treatment regimens.
Methods Inclusion criteria: Patients diagnosed with de novo or secondary AML according to WHO classification (except for APL), previously untreated, age ≥ 65 years, ECOG <4. Randomization arms (1:1): azacytidine (AZA) (75 mg/m2 SC days 1-7) and FLUGA (fludarabine 40 mg/m2 PO days 2-6, cytarabine 75 mg/m2 SC days 2-6, filgrastim 5 µg/Kg SC days 1-3), cycles of 28 days. The treatment schedule consisted of 3 induction cycles (C1, C2, C3), 6 consolidation cycles and a maintenance treatment until relapse or progression. FLUGA was reduced in patients older than 75 years and filgrastim in the FLUGA arm was omitted when white blood cells (WBC) were > 25 x109/L. Concomitant use of hydroxyurea was allowed in the AZA arm if WBC was between 15 and 50 x109/L. In patients with more than 50 x109/L WBC assigned to the AZA arm, the first induction cycle was FLUGA.
An interim analysis to assess myelotoxicity, early mortality and response rate was planned in the trial when the last of the first 100 randomized patients completed the first 3 induction cycles.
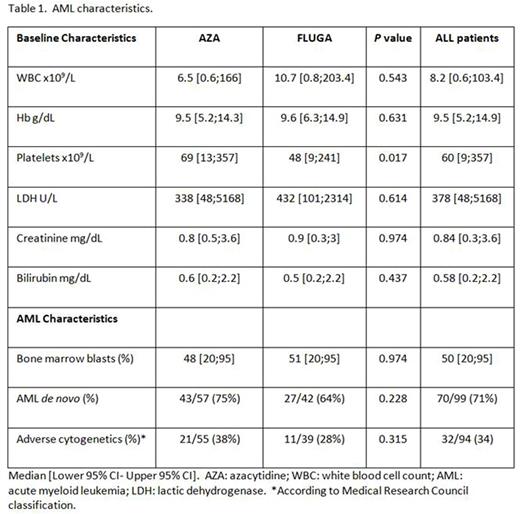
Results: From October 2014 to April 2016, 162 patients were enrolled in 26 Spanish centers from the PETHEMA group. Of them, 22 patients were screen failure and 16 patients did not receive the assigned treatment. The first 100 patients treated were analyzed (58 patients were assigned to AZA arm and 42 to FLUGA arm). Baseline characteristics are shown in Table 1. Median age was 75 [65;90] years in AZA arm vs. 77 [65;88] years in FLUGA arm; 36 (62%) vs. 19 (45%) males. Five patients from AZA arm (n=58) received the first cycle of FLUGA because of WBC > 50 x109/L. Outcomes: overall response rate (CR+CRu+PR) based on the best response accumulated with 3 cycles of treatment was 62% in the AZA arm and 57% in the FLUGA arm. CR+CRu was achieved in 14 patients (24%) from AZA arm and 15 (36%) from FLUGA arm. Early mortality rate (8 weeks) was 14% in AZA and 22% in FLUGA arm. Toxicities: there were no differences in hospitalization and non-hematological ≥ G3 serious adverse events (SAEs) between both arms. Overall, 71 patients (71%), 61 out of 79 (77%) vs. 41 out of 64 (64%) patients, developed neutropenia (< 0.5 x x109/L) after C1, C2 and C3, respectively. Median duration of neutropenia in the AZA arm was 14 days (6;40), 21 days (4;95) and 18 days (1;45) after in C1, C2 and C3, respectively, vs. 15 days (4;33), 15 days (3;36) and 20 days (7;41) after C1, C2 and C3, respectively, in the FLUGA arm (no statistically significant differences). Eighty-one (81%), 55 out of 79 (70%) vs. 40 out of 64 (62%) patients, developed thrombocytopenia (< 50 x109/L) after C1, C2 and C3, respectively. Median duration of thrombocytopenia was 14 days (5;29) after C1, 18 days (4;88) after C2 and 14 days (7;25) after C3 in the AZA arm; vs. 16 (3;63), 20 (6;42) and 13 days (7;56) after C1, C2 and C3, respectively, in the FLUGA arm (NS).
Conclusions: Outpatient treatment with AZA or FLUGA was feasible. Both treatment arms showed a similar rate of early mortality and relatively low CR/CRu rates. The study is currently enrolling patients until the planned accrual goal (350 patients) to assess the primary end-point (1-year OS). The study is registered at www.ClinicalTrials.gov as NCT02319135.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.