Abstract
Background:225Ac-lintuzumab is a radioimmunoconjugate composed of 225Ac (t½=10 days), which emits 4 α-particles, linked to a humanized anti-CD33 monoclonal antibody. Short-ranged (50-80 µm), high-energy (~100 keV/µm) α particle-emitting isotopes such as 225Ac may result in more specific tumor cell kill and less damage to normal tissues than β-emitters. An initial phase I trial in 20 patients with relapsed/refractory AML showed that a single infusion of 225Ac-lintuzumab is safe at doses ≤ 3 µCi/kg and has anti-leukemic activity (Jurcic et al. ASH, 2011). We conducted a multicenter, phase I dose-escalation trial to determine the maximum tolerated dose (MTD), toxicity, and biological activity of fractionated-dose 225Ac-lintuzumab in combination with LDAC.
Patients and Methods: Patients ≥ 60 years with untreated AML not candidates for standard induction therapy (e.g., antecedent hematologic disorder, poor-risk cytogenetic or molecular features, and significant comorbidities) were eligible. Patients received LDAC 20 mg twice a day for 10 days every 4-6 weeks for up to 12 cycles. During Cycle 1, 2 fractions of 225Ac-lintuzumab were given 1 week apart, beginning 4-7 days following completion of LDAC. To prevent radiation-induced nephrotoxicity, patients were given furosemide while receiving 225Ac-lintuzumab then spironolactone for 1 year afterward. Four dose levels of 225Ac-lintuzumab were studied using a 3+3 design. Dose escalation proceeded if < 33% of patients in a cohort experienced dose-limiting toxicity (DLT).
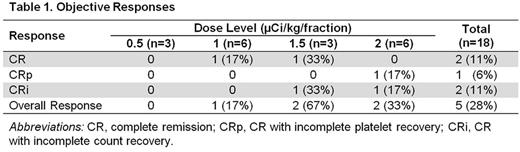
Results: Eighteen patients (median age, 77 years; range, 68-87 years) completed therapy. Twelve (67%) had prior myelodysplastic syndrome (MDS), for which 10 (83%) received therapy with hypomethylating agents (n=9) or allogeneic hematopoietic cell transplantation (n=1). One patient (6%) had chronic myeloid leukemia in molecular remission prior to developing AML. Eleven patients (61%) had intermediate-risk and 7 (39%) had poor-risk disease by NCCN criteria. Median CD33 expression was 81% (range, 30-100%). 225Ac-lintuzumab was given at 0.5 (n=3), 1 (n=6), 1.5 (n=3), or 2 (n=6) μCi/kg/fraction. Up to 4 cycles of LDAC were administered. Two patients experienced DLT (grade 4 thrombocytopenia with marrow aplasia for > 6 weeks following therapy), one each in the 1 and 2 µCi/kg/fraction cohorts. Although the MTD was not reached, 2 µCi/kg/fraction was chosen as the phase II dose to limit prolonged myelosuppression. Hematologic toxicities included grade 4 neutropenia (n=5) and thrombocytopenia (n=9). Grade 3/4 non-hematologic toxicities included febrile neutropenia (n=6), pneumonia (n=5), other infections (n=3), atrial fibrillation/syncope (n=1), transient creatinine increase (n=1), generalized fatigue (n=1), hypokalemia (n=1), mucositis (n=1), and rectal hemorrhage (n=1). Thirty- and 60-day mortality rates were 0% and 17%, respectively. Eleven of 14 patients (79%) evaluated after Cycle 1 had bone marrow blast reductions (mean reduction, 66%; range, 19-100%). Objective responses (2 CR, 1 CRp, 2 CRi) were seen in 5 of the 18 patients (28%), but only at doses ≥ 1 µCi/kg/fraction (Table 1). One of the responders received 15 cycles of azacitidine for prior MDS. All responses occurred after 1 cycle of therapy, in contrast to historical data with LDAC alone, where the median time to response was 3 cycles. Median progression-free survival (PFS) for all patients was 2.7 months (range, 1.0-31.8+ months). Median overall survival (OS) was 5.6 months (range, 1.6-32+ months). Median response duration was 5.6 months (range, 4.9-32+ months). Peripheral blood blast counts were a strong predictor of response. Among 38 patients treated in the current and initial phase I trials, responses were seen in 8 of 19 patients (42%) with blast counts < 200/µL, compared with 0 of 17 patients with blast counts ≥ 200/µL (P=0.002). This difference is likely due to decreased marrow targeting in patients with higher circulating blast counts when the subsaturating antibody doses used in this trial are given.
Conclusions: Fractionated-dose 225Ac-linutuzmab can be safely combined with LDAC and induce remission in older patients with untreated AML. A phase II trial of 225Ac-lintuzumab monotherapy at 2 µCi/kg/fraction using hydroxyurea, if needed, to lower peripheral blast counts prior toadministration will be undertaken to determine response rate, PFS, and OS in this patient population.
Jurcic:Forma Therapeutics: Research Funding; Seattle Genetics: Research Funding; Kura Oncology: Research Funding; Celgene: Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees; Bayer: Consultancy; Alexion Pharmaceuticals: Consultancy; Merck & Co.: Consultancy; Astellas: Research Funding; Actinium Pharmaceuticals, Inc.: Research Funding; Daiichi-Sankyo: Research Funding. Levy:Janssen: Speakers Bureau; Actinium Pharmaceuticals, Inc.: Research Funding; Seattle Genetics: Research Funding; Amgen: Speakers Bureau; Takeda Pharmaceuticals International Co.: Speakers Bureau. Park:Amgen: Consultancy; Genentech/Roche: Research Funding; Juno Therapeutics: Consultancy, Research Funding. Ravandi:Actinium Pharmaceuticals, Inc.: Research Funding. Perl:Actinium Pharmaceuticals, Inc.: Research Funding; Seattle Genetics: Consultancy. Smith:Actinium Pharmaceuticals, Inc.: Research Funding. Cicic:Actinium Pharmaceuticals, Inc.: Employment, Equity Ownership. Scheinberg:Actinium Pharmaceuticals, Inc.: Equity Ownership, Patents & Royalties: Ac-225-Lintuzumab.
Author notes
Asterisk with author names denotes non-ASH members.