Abstract
BACKGROUND: Allo-HSCT is an accepted treatment option for patients with relapsed and refractory B cell lymphomas or high-risk chronic lymphocytic leukemia (CLL). This treatment option is currently reshaped by the introduction of pathway inhibitors (PWI) as ibrutinib, idelalisib, and venetoclax. However, the mid- and long-term efficacy and toxicity of these drugs are not yet fully defined and most patients need to discontinue their treatment over time because of disease progression or intolerance. Allo-HSCT could induce long-term disease control with curative potential especially in poor prognosis patients. In case of disease relapse after transplantation, patient's outcome is a major concern and the prognosis is dismal. In this context, PWI could be very promising in the context of lowering the bulky leukemic cells before proceeding to allo-HSCT but also when used after transplantation to treat or prevent disease relapse.
The purpose of the present study is to provide information on the safety and efficacy of ibrutinib when administered after allo-HSCT for mantle cell lymphoma (MCL) or CLL.
DESIGN: We included in this study adult patients who had been registered with the EBMT for an allo-HSCT for CLL or MCL and who received ibrutinib for treating disease recurrence or persistence at any time after transplant. Baseline patient, disease, and transplant data were collected from MED-A forms. Centers were requested to provide additional post-allo-HSCT treatment and follow-up information.
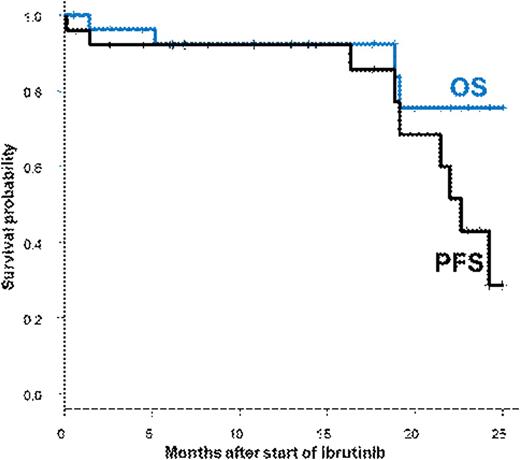
RESULTS: A total of 30 patients, 22 (73%) males, who had undergone allo-HSCT between September 2002 and December 2015 were included in this study. Diagnosis was CLL in 27 (90%) patients and MCL in 3 patients. The median age at transplantation was 55 (range: 38-66) years and the median number of treatment lines prior to transplantation was 3 (1-8). Before transplantation, in CLL patients, del17p was present in 10/27 (37%) of cases and del11q was present in 4/27 (15%) other cases. Prior to allo-HSCT, 4 patients (2 CLL and 2 MCL) had received an auto-HSCT and 4 other CLL patients had been exposed to ibrutinib for a median of 261 (205-376) days. Disease response at allo-HSCT was sensitive in 20/27 (74%) of the CLL patients, and in 1/3 of the MCL patients. Conditioning was reduced-intensity in 20/30 (67%) of the transplants and included in-vivo T cell depletion in the majority of cases (17/20). Donors were identical siblings in 23% with PBSC being the predominant stem cell source (28/30). Acute GVHD grade II-IV was observed in 5/30 (16.6%) patients (3 grade II, 2 grade III), while chronic GVHD had occurred in 15/29 patients but had resolved in 11/15 patients prior to ibrutinib. Patients received ibrutinib for relapse after allo-HSCT (n=27, median time from allo-HSCT to progression: 22 (0.5-53) months), 1 for stable disease and 2 after positive MRD. Donor lymphocyte infusions (DLI) were administered concomitantly with ibrutinib in 3/30 patients. Ibrutinib was generally well tolerated; major toxicities observed after ibrutinib commencement were secondary cancers in 3 patients and toxic epidermiolysis in 1 patient. De novo chronic GVHD occurred in 2 patients, in one of them in the context of DLI. Overall, 23/30 (77%) patients had responsive disease after ibrutinib (33% reached CR), 21/27 with CLL (8 CR) and 2/3 with MCL (2CR). At the last follow-up, 20 (67%) patients are still on ibrutinib after a median exposure of 395 (17-703) days, 19 of them with ongoing response (8 in CR) and one too early for response assessment. Ten patients had discontinued ibrutinib, 4 because of toxicity (2 secondary cancers, 2 skin toxicity), and 6 because of disease progression. In total, 4 deaths (all CLL) were observed and were due only to progression. With altogether 8 progression events, the median PFS after start of ibrutinib of all 30 patients was 23 months, the one year OS and PFS probabilities were 92%. There was no effect of del17p or time from allo-HSCT to ibrutinib start on PFS.
CONCLUSION: Ibrutinib can be safely administered for CLL/MCL relapse after allo-HSCT, with an efficacy at least similar to non-transplanted patients with high-risk disease.
Dreger:Gilead: Speakers Bureau; Janssen: Consultancy; Novartis: Speakers Bureau; Novartis: Consultancy; Gilead: Consultancy; Roche: Consultancy. Corradini:Gilead: Honoraria; Servier: Honoraria; Sanofi: Honoraria; Takeda: Honoraria; Roche: Honoraria; Novartis: Honoraria; Janssen: Honoraria; Celgene: Honoraria; Takeda: Consultancy. Schetelig:Sanofi: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.