Key Points
Brain tumor patients have a very high risk of VTE.
Podoplanin expression by primary brain tumors induces platelet aggregation and is associated with hypercoagulability and a high risk of VTE.
Abstract
Venous thromboembolism (VTE) is common in patients with brain tumors, and underlying mechanisms are unclear. We hypothesized that podoplanin, a sialomucin-like glycoprotein, increases the risk of VTE in primary brain tumors via its ability to induce platelet aggregation. Immunohistochemical staining against podoplanin and intratumoral platelet aggregates was performed in brain tumor specimens of 213 patients (mostly high-grade gliomas [89%]) included in the Vienna Cancer and Thrombosis Study, a prospective observational cohort study of patients with newly diagnosed cancer or progressive disease aimed at identifying patients at risk of VTE. Platelet aggregation in response to primary human glioblastoma cells was investigated in vitro. During 2-year follow-up, 29 (13.6%) patients developed VTE. One-hundred fifty-one tumor specimens stained positive for podoplanin (33 high expression, 47 medium expression, 71 low expression). Patients with podoplanin-positive tumors had lower peripheral blood platelet counts (P < .001) and higher D-dimer levels (P < .001). Podoplanin staining intensity was associated with increasing levels of intravascular platelet aggregates in tumor specimens (P < .001). High podoplanin expression was associated with an increased risk of VTE (hazard ratio for high vs no podoplanin expression: 5.71; 95% confidence interval, 1.52-21.26; P = .010), independent of age, sex, and tumor type. Podoplanin-positive primary glioblastoma cells induced aggregation of human platelets in vitro, which could be abrogated by an antipodoplanin antibody. In conclusion, high podoplanin expression in primary brain tumors induces platelet aggregation, correlates with hypercoagulability, and is associated with increased risk of VTE. Our data indicate novel insights into the pathogenesis of VTE in primary brain tumors.
Introduction
Patients with cancer are at risk of venous thromboembolism (VTE), with an estimated rate as high as 20% annually.1 Brain tumor patients belong to those with the highest risk. Because occurrence of VTE leads to increased mortality,2 the investigation of risk factors and mechanisms of developing VTE is of utmost clinical interest. Although several risk factors of VTE in patients with malignant brain tumors have been identified, such as patient age, leg paresis, glioblastoma tumor subtype, or intraluminal tumor thrombosis,3-5 underlying mechanisms explaining the high thrombotic risk remain to be elucidated.
Recently, Thaler et al showed that brain tumor patients with low blood platelet counts and high plasma levels of the cell adhesion molecule soluble P-selectin (sP-selectin), a marker of platelet and endothelial cell activation, are at high risk of VTE.6 This observation may indicate that platelets play a role in the development of VTE in patients with brain tumors.
A molecule that has the ability to induce blood platelet activation and that is frequently expressed by primary brain tumors is the sialomucin-like glycoprotein podoplanin. In healthy tissue, podoplanin is strongly expressed on lymphatic endothelial cells.7 Interestingly, podoplanin is the only known physiologic ligand for the platelet activation receptor C-type lectin receptor type 2 (CLEC-2), and binding of podoplanin to CLEC-2 induces platelet aggregation.8,9 Podoplanin-induced platelet aggregation is important in the separation of the lymphatic system from blood vessels during embryonic development.10
Malignant brain tumors express podoplanin to a various extent,11 and its expression was associated with worse survival in patients with glioblastoma.12,13 Because inhibition of podoplanin improved survival in a murine glioblastoma-model, it might be an anticancer drug target.14 Further, cancer cells expressing podoplanin can activate platelets via CLEC-2, thereby inducing platelet aggregation.15
We hypothesized that podoplanin, because of its ability in activating blood platelets, is involved in the procoagulatory state and the development of VTE in patients with primary malignant brain tumors. Thus, in this prospective cohort study, we explored the association of intratumoral podoplanin expression with hypercoagulability and intratumoral platelet aggregates and risk of VTE and mortality in patients with brain tumors. To substantiate a role of brain tumor–inducible platelet activation, we studied platelet response to glioblastoma cells in vitro.
Methods
Study design and study population
The study was performed within the framework of the Vienna Cancer and Thrombosis Study (CATS), a single-center, prospective, ongoing observational cohort study on cancer patients that started in 2003 at the Medical University of Vienna. The aim of the study is to identify risk factors for cancer-associated VTE. Exact methods, inclusion, and exclusion criteria have been described in previous publications.16 Briefly, patients with newly diagnosed malignancy or progression of disease after remission are included. Exclusion criteria consist of chemotherapy within the prior 3 months, radiotherapy or surgery within the prior 2 weeks, diagnosis of VTE during the prior 3 months, and/or continuous oral anticoagulation or therapy with low-molecular-weight heparin. At study inclusion, past medical history is recorded in detail and a single blood draw is performed. Patients are then followed for a maximum time period of 2 years, as described in previous publications.16
Outcome measures of the study
The primary end point is occurrence of objectively confirmed, symptomatic, nonfatal and fatal VTE during a 2-year follow-up period. The secondary end point is death from any cause within the same time period.
For the current study, all patients with primary malignant brain tumors enrolled into CATS from October 2003 to March 2014, from whom a formalin-fixed and paraffin-embedded brain tumor sample was available in the bio-bank of the neuropathologic institute of the Medical University of Vienna were included.
Ethical approval for the study was received from the institutional ethics committee (EC number: 126/2003, ethik-kom@meduniwien.ac.at). The study was conducted in accordance with the Declaration of Helsinki.
Immunohistochemistry
Podoplanin expression and intravascular platelet clusters were determined by immunohistochemical staining of tissue samples. Sections at a thickness of 3-5 μm were cut from routine formalin-fixed and paraffin-embedded tumor tissue blocks.
Staining against podoplanin was performed with the monoclonal mouse antipodoplanin antibody D2-40 (Cell Marque, Rocklin, CA). For detection of platelets, the monoclonal mouse anti-CD61 (glycoprotein [GP] IIIa) antibody (NCL-CD61-308, Leica Biosystems, Newcastle, UK) was used in a dilution of 1:100. A lymph node sample served as positive control for podoplanin and a tonsil sample for CD61. A Benchmark Ultra immunostainer (Ventana Medical Systems, Tuscon, AZ) was used for immunohistochemistry.
All sections were reviewed by 2 observers experienced in histopathologic evaluation of primary brain tumors together on a multiheaded microscope. The observers were blinded to clinical data. Podoplanin staining intensity was semiquantitatively classified into the following degrees: (–) negative; (+) low expression, <50% of cancer cell express podoplanin and/or mild staining intensity; (++) moderate expression, 50% to 70% of cancer cell express podoplanin at a moderate to strong intensity; and (+++) high expression, >70% of cancer cells express podoplanin at a strong intensity level.
The intensity of intravascular platelet clusters, determined by staining against the platelet surface protein CD61, was categorized into the following levels: (–) negative; (+) isolated, small, CD61+ thrombotic vessels; (++) multiple CD61+ thrombotic vessels; (+++) very large and/or plenty of CD61+ thrombotic vessels.
Cell culture
Cancer cell line “Gli16” was established at the Institute of Cancer Research, Medical University of Vienna, Austria, from a patient with glioblastoma. Cancer cell line “AMCH” was established at the Department of Neurosurgery, Wagner-Jauregg Hospital, Linz, Austria, from a patient with gliosarcoma. Cancer cells were grown in RPMI 1640 medium with 10% fetal calf serum.
Podoplanin expression on cancer cells was investigated by flow cytometry using the rat anti-human podoplanin antibody NZ-1 (AngioBio, San Diego, CA) as a primary antibody and rat IgG for isotype control (Sigma-Aldrich, St. Louis, MO). The donkey anti-rat IgG Alexa Fluor 488-conjugate (Thermo Fisher, Waltham, MA) served as the secondary antibody. Stainings were performed on ice for 20 minutes, and cells were immediately analyzed with a Gallios flow cytometer (Beckman Coulter, Brea, CA).
Platelet aggregometry
Light transmission aggregometry (LTA) was performed using a PAP 8E (MoeLab, Langenfeld, Germany) aggregometer to investigate tumor-cell–induced platelet aggregation. Citrated blood from a healthy donor was centrifuged at 150g for 8 minutes to obtain platelet- rich plasma (PRP). For standard experiments, 5 × 105 cancer cells were added to 225 µL PRP and aggregation was recorded for 10 minutes. In dose-response experiments, the number of cancer cells was modified, and increasing cancer cell numbers were used. Thrombin-receptor activating peptide (TRAP)-6 (Bachem, Bubendorf, Switzerland) was used as a positive control for platelet aggregation. To confirm podoplanin-dependent platelet aggregation, experiments were repeated after preincubation of glioblastoma cells with the anti-human podoplanin antibody NZ-1 (AngioBio) or a control antibody (rat IgG control antibody, Sigma-Aldrich) at a final concentration of 50 µg/mL for 90 minutes at 37°C. It should be noted that NZ-1 treatment resulted in loss of podoplanin surface expression (presumably because of protein internalization) but did not affect cell viability.
Release of platelet factor 4
After co-incubation of 5 × 105 cancer cells with 225 µL PRP for 10 minutes (37°C, stirring) plasma was centrifuged at 3000g for 5 minutes, supernatant was collected, and aliquots were stored at −80°C until further processing. Platelet factor 4 (PF4) was measured by using a commercially available enzyme-linked immunosorbent assay (ELISA) kit (human PF4 ELISA, Quantikine, R&D Systems, Minneapolis, MN).
Statistical analyses
Statistical analyses were performed using IBM SPSS Statistics (Version 2014, IBM Corp., Armonk, NY) and Stata (Windows version 13.0, Stata Corp., Houston, TX). Continuous variables were summarized using medians with interquartile ranges, and categorical variables by absolute frequencies and percentages. We compared means between groups with simple and multiple linear regression, and used χ2 tests to evaluate the association of 2 categorical variables. The follow-up period was defined from study inclusion until VTE, death, or censoring alive at 2 years after baseline. The inverse Kaplan-Meier estimator was applied to calculate the median follow-up time.17 We estimated and graphed the cumulative incidence of VTE in the presence of competing mortality using Stata’s stcompet suite, which implements the cumulative incidence and variance estimators proposed by Marubini and Valsecchi, and Choudhury, respectively.18 In all analyses, death from any cause except fatal VTE was considered as the competing event of interest. To study the association between podoplanin and other variables with the risk of VTE, we performed an “etiologic-type” analysis of cause-specific VTE hazards using log-rank tests and uni- and multivariable Cox proportional hazards models.19,20 In addition, Gray’s tests for comparing cumulative VTE incidences were performed. The all-cause mortality end point was analyzed with Kaplan-Meier product limit estimators, log-rank tests, and uni- and multivariable Cox models. Fitting an interaction term between podoplanin and the natural logarithm of follow-up time did not reveal evidence for nonproportional hazards in any of the presented models. This study is reported in concordance with STROBE recommendations.21
Results
Study population
From October 2003 until March 2014, 285 patients with primary malignant brain tumors were included in CATS. Of those, 13 had to be excluded because of not meeting the exact inclusion and exclusion criteria after reevaluation, and in 59 cases no tumor sample was available. Therefore, a total of 213 patients were eligible for the current study and were followed for a median of 731 days (range, 3-731).
Patients were included in the present study for a median 17 days after tumor biopsy or tumor surgery. In total, 75 patients had complete tumor resection, 77 incomplete tumor resection, and 61 had tumor biopsy before study inclusion.
According to the classification of the World Health Organization (WHO), brain tumor subtypes consisted mainly of high-grade gliomas: in 152 patients (71.4%), a glioblastoma (WHO grade IV) was diagnosed, and 38 patients (17.8%) had an anaplastic glioma (WHO grade III). Table 1 shows characteristics of all included patients. Tumors were newly diagnosed in 185 patients (86.9%), and 28 patients (13.1%) had a recurrent brain tumor after complete or partial remission.
Thromboembolic events and deaths during follow-up
During follow-up, 29 patients (13.6%) developed objectively confirmed VTE and 113 patients (53.1%) died. The 6-, 12-, and 24-month risks of VTE were 10.1%, 13.3%, and 14.8%, respectively. Thromboembolic index events consisted of 15 deep vein thrombosis of the lower extremity (51.7%), 13 pulmonary embolisms (44.8%), and 1 deep vein thrombosis of the upper extremity (3.4%). Fatal pulmonary embolism was objectively documented in 2 cases.
The 6-, 12-, and 24-month probabilities of survival were 84.3%, 65.1%, and 40.6%, respectively.
Immunohistologic staining pattern
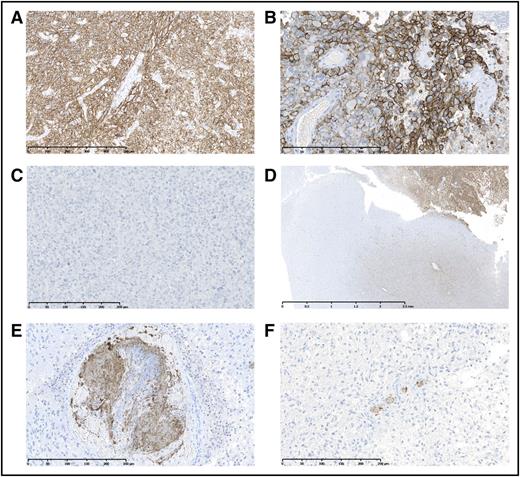
In tumor specimens staining positive for podoplanin, expression patterns were diffuse and patchy throughout the tumor. Podoplanin was mostly membrane-bound on tumor cells. Normal brain tissue of cerebrum, cerebellum, and white matter showed no podoplanin expression. In some slides, macrophages phagocytizing podoplanin-positive tumor cells were observed. We did not observe podoplanin-positive lymphatic vessels. Representative slides are shown in Figure 1.
Representative samples showing podoplanin expression and intravascular platelet clusters (antiplatelet surface protein CD61) in brain tumor specimens. (A-B) Representative examples of high podoplanin (+++)-expressing tumors. (C) Representative example of a podoplanin-negative (–) tumor. (D) The invasion zone of a podoplanin-expressing tumor is depicted. (E) A very large CD61+ intravascular thrombosis (+++) is shown. (F) Tumor sample with multiple CD61+ thrombotic vessels (++). NDP Viewing Software (NDP.view2; Hamamatsu Photonics, Japan) was used for microscopy photographs. Full scale bars represent 500 μm (A), 250 μm (B-C,E-F), and 2.5 μm (D).
Representative samples showing podoplanin expression and intravascular platelet clusters (antiplatelet surface protein CD61) in brain tumor specimens. (A-B) Representative examples of high podoplanin (+++)-expressing tumors. (C) Representative example of a podoplanin-negative (–) tumor. (D) The invasion zone of a podoplanin-expressing tumor is depicted. (E) A very large CD61+ intravascular thrombosis (+++) is shown. (F) Tumor sample with multiple CD61+ thrombotic vessels (++). NDP Viewing Software (NDP.view2; Hamamatsu Photonics, Japan) was used for microscopy photographs. Full scale bars represent 500 μm (A), 250 μm (B-C,E-F), and 2.5 μm (D).
Podoplanin expression correlates with intratumoral intravascular platelet clusters
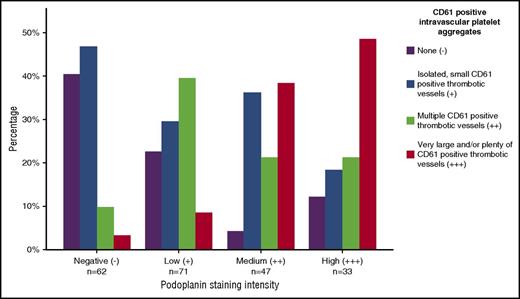
Of 213 tumor specimens, 151 (70.9%) stained positive for podoplanin (71 low [+]), 47 medium [++], and 33 high [+++] expression). Intravascular, CD61+ platelet clusters were detected in 166 specimens (73 low grade [+], 51 medium [++], 42 high grade [+++]). Podoplanin expression was positively correlated to grade of intravascular platelet clusters (P < .001; Figure 2).
Association between podoplanin expression levels and grade of intratumoral intravascular platelet aggregates. Increasing levels of podoplanin expression intensity correlated with higher levels of CD61+ vascular platelet aggregates (χ2, P < .001).
Association between podoplanin expression levels and grade of intratumoral intravascular platelet aggregates. Increasing levels of podoplanin expression intensity correlated with higher levels of CD61+ vascular platelet aggregates (χ2, P < .001).
Detailed information about podoplanin and CD61 staining according to brain tumor histology is given in supplemental Table 1, available on the Blood Web site.
Podoplanin expression correlates with high D-dimer and low blood platelet counts
Table 2 shows selected peripheral blood parameters in patients with podoplanin expression and podoplanin negative tumors. Patients with podoplanin-expressing tumors had significantly lower blood platelet counts, higher D-dimer, higher leukocyte counts, higher coagulation factor VIII (FVIII) activity, and higher prothrombin fragment 1 + 2 (F 1 + 2) levels. After adjusting for age, sex, and tumor type, blood platelet count was significantly lower, and D-dimer was significantly higher in patients with podoplanin-expressing tumors, compared with podoplanin-negative tumors.
There was no relevant correlation between sP-selectin and platelet count (r = 0.16; P = .029).
Podoplanin expression is associated with risk of VTE
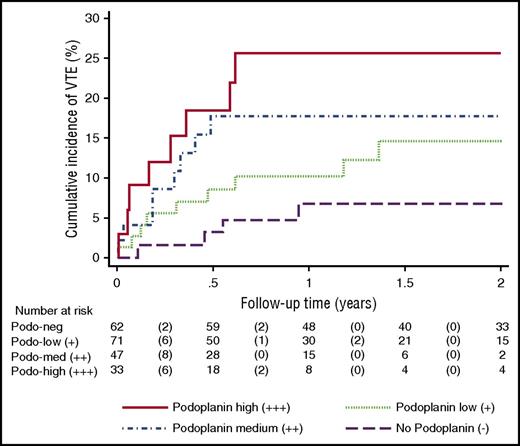
In competing risk analysis, the cumulative 6-, 12-, and 24-month incidences of VTE were 3.3%, 6.7%, and 6.7%, respectively, in patients with podoplanin-negative tumors; 8.7%, 10.3%, and 14.6% for tumors with low podoplanin expression,;17.8%, 17.8%, and 17.8% for moderate podoplanin expression; and 18.4%, 25.7%, and 25.7% for high podoplanin expression (log-rank test for comparing cause-specific VTE hazards P = .019, Gray’s test for comparing cumulative VTE incidences P = .062; Figure 3).
Cumulative incidence of venous thromboembolism (VTE) accounting for competing risk (death of any cause other than fatal VTE) according to expression levels of podoplanin. Podoplanin expression was associated with a significantly increased risk of VTE; the probability of VTE increased with increasing levels of podoplanin staining intensity. Log-rank test comparing the cause-specific hazards of VTE (null hypothesis: all 4 podoplanin staining groups have the same VTE rate): P = .019; Gray’s test comparing the competing risk cumulative incidences (null hypothesis: all 4 podoplanin staining groups have the same VTE risk): P = .062.
Cumulative incidence of venous thromboembolism (VTE) accounting for competing risk (death of any cause other than fatal VTE) according to expression levels of podoplanin. Podoplanin expression was associated with a significantly increased risk of VTE; the probability of VTE increased with increasing levels of podoplanin staining intensity. Log-rank test comparing the cause-specific hazards of VTE (null hypothesis: all 4 podoplanin staining groups have the same VTE rate): P = .019; Gray’s test comparing the competing risk cumulative incidences (null hypothesis: all 4 podoplanin staining groups have the same VTE risk): P = .062.
In the univariable analysis of cause-specific VTE hazards, patients with podoplanin-expressing tumors had a significantly higher risk of VTE compared with patients with podoplanin-negative tumors (hazard ratio [HR] = 3.44 [95% confidence interval [CI], 1.19-9.95]; P = .022, supplemental Table 2). This univariable association prevailed in competing risk regression accounting for death as a competing risk (subdistribution hazard ratio [SHR] = 2.30 [95% CI, 1.12-4.73]; P = .023). Table 3 shows uni- and multivariable HR for VTE in patients with low, medium, and high podoplanin expression compared with podoplanin-negative tumors; corresponding estimates from competing risk regression are reported in supplemental Table 3.
Intravascular platelet aggregates are associated with risk of VTE
In univariable Cox regression analysis, occurrence of multiple and/or very large (++ and +++) intravascular platelet aggregates (n = 93) was associated with an increased risk of VTE compared with tumor samples with no or only small (– and +; n = 120) CD61+ thrombotic vessels (HR 2.85 [1.28-6.37]; P = .011), and this association remained statistically significant after adjustment for age, sex, and glioblastoma subtype (HR 2.59 [1.10-6.10]; P = .029). Similar results were observed in the corresponding univariable (SHR 2.73 [1.23-6.03]; P = .013) and multivariable (SHR 2.55 [1.03-6.32]; P = .044) competing risk regressions, respectively.
Podoplanin expression is associated with overall mortality
Podoplanin expression was associated with increased risk of overall mortality during the 2-year follow-up period. The univariable HR for low, medium, and high podoplanin expression compared with no podoplanin expression were 2.78 (1.59-4.87, P < .001), 4.70 (2.63-8.40, P < .001), and 4.44 (2.37-8.31, P < .001), respectively. This association remained statistically significant in multivariable analysis adjusting for age, sex, and tumor type (WHO grade IV, III, II, and others): 2.14 (1.18-3.89, P = .012), 3.03 (1.62-5.68, P = .001), and 2.58 (1.29-5.15, P = .007), respectively.
In Kaplan-Meier analysis, the probability of survival after 6, 12, and 24 months was 98.4%, 88.2%, and 68.0%, respectively, in patients with podoplanin-negative tumors; 81.5%, 61.4%, and 39.0% for low podoplanin expression; 78.0%, 50.8%, and 12.2% for moderate podoplanin expression; and 72.2%, 49.3%, and 21.9% for high podoplanin expression (log-rank P < .001; supplemental Figure 1).
Podoplanin induces platelet aggregation and platelet activation in vitro
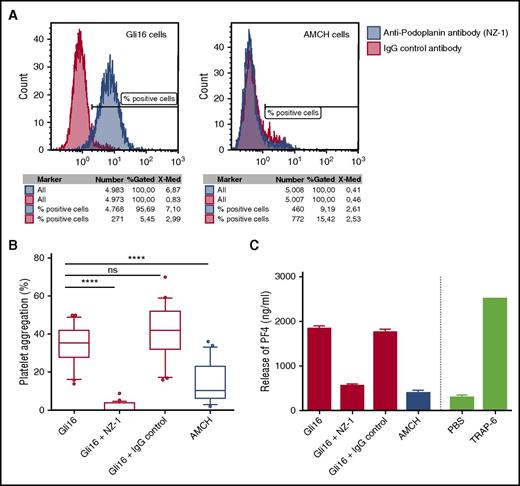
Glioblastoma cell line “Gli16” was isolated from a 46-year-old male patient who was included in the Vienna CATS and who developed massive, symptomatic, bilateral PE during the follow-up period. Immunohistochemical staining of tumor tissue, obtained from surgery at first diagnosis of glioblastoma, revealed podoplanin expression on tumor cells and large intravascular platelet aggregates in the tumor tissue (supplemental Figure 2). As a negative control, a podoplanin-negative cancer cell line (“AMCH”), derived from a patient with gliosarcoma, was used. Cell line Gli16 stained highly positive for podoplanin in flow cytometric analysis, whereas AMCH stained negative for podoplanin expression (Figure 4A). Gli16 cells induced platelet aggregation in a dose-dependent manner, as investigated by LTA (supplemental Figure 3). Furthermore, Gli16 cells led to significantly higher platelet aggregation compared with AMCH (P < .001) (Figure 4B). The anti-podoplanin antibody NZ-1 significantly abrogated tumor-cell–induced platelet aggregation by Gil16, whereas this was not observed when a control antibody was used (Figure 4B).
Podoplanin expression and tumor cell–induced platelet aggregation and platelet activation in vitro. (A) Podoplanin expression on cancer cell lines “Gli16” and “AMCH” was investigated by flow cytometry. (B) The ability of the cell lines to induce platelet aggregation was investigated by LTA: A total number of 5 × 105 cancer cells were added to 225 µL of PRP,and maximal platelet aggregation (%) after 10 minutes was recorded. Gli16 cells induced significantly stronger platelet aggregation compared with control cell line AMCH (***P < .001). Platelet aggregation in response to Gli16 was abrogated by the antipodoplanin antibody NZ-1, but not by control antibody (***P < .001). ns, not significant. Box plots show data from 5 individual experiments (performed in multiple replicates; total n = 22). (C) Platelet activation and degranulation upon coincubation of 225 µL PRP with 5 × 105 cancer cells was determined by a PF4 ELISA. Gli16 cells induced marked release of PF4 from platelets, which was not observed for AMCH cells. The ability of Gli16 cells to induce PF4 release from platelets could be inhibited by the anti-podoplanin antibody NZ-1, but not by control antibody. The experiment was performed in duplicates.
Podoplanin expression and tumor cell–induced platelet aggregation and platelet activation in vitro. (A) Podoplanin expression on cancer cell lines “Gli16” and “AMCH” was investigated by flow cytometry. (B) The ability of the cell lines to induce platelet aggregation was investigated by LTA: A total number of 5 × 105 cancer cells were added to 225 µL of PRP,and maximal platelet aggregation (%) after 10 minutes was recorded. Gli16 cells induced significantly stronger platelet aggregation compared with control cell line AMCH (***P < .001). Platelet aggregation in response to Gli16 was abrogated by the antipodoplanin antibody NZ-1, but not by control antibody (***P < .001). ns, not significant. Box plots show data from 5 individual experiments (performed in multiple replicates; total n = 22). (C) Platelet activation and degranulation upon coincubation of 225 µL PRP with 5 × 105 cancer cells was determined by a PF4 ELISA. Gli16 cells induced marked release of PF4 from platelets, which was not observed for AMCH cells. The ability of Gli16 cells to induce PF4 release from platelets could be inhibited by the anti-podoplanin antibody NZ-1, but not by control antibody. The experiment was performed in duplicates.
To further investigate the ability of the 2 cell lines to induce platelet activation, release of PF4 upon coincubation of platelet rich plasma with cancer cells was investigated. Gli16 cells induced marked release of PF4 from platelets (PF4 concentration in plasma, mean ± standard deviation: 1955 ± 75 ng/mL). Release of PF4 by Gli16 cells could be inhibited by NZ-1 (564 ± 39 ng/mL), but not by control antibody (1764 ± 224 ng/mL). AMCH cells induced only minor release of PF4 from platelets (403 ± 50 ng/mL), comparable with the negative control (phosphate-buffered saline: 301 ± 45 ng/mL) (Figure 4C).
Discussion
In this study of patients with primary brain tumors, we provide evidence for an association of intratumoral podoplanin expression with increased risk of VTE and mortality. The risk of developing VTE was approximately 6 times higher in patients with high podoplanin–expressing tumors compared with patients with podoplanin-negative tumors. Podoplanin expression was linked to intravascular platelet clusters and thrombotic vessels in tumor samples. In vitro glioblastoma cells induced aggregation of human platelets via podoplanin. Interestingly, podoplanin expression was associated with decreased blood platelet counts, as well as with increased plasma levels of D-dimer. These results support the concept that podoplanin expression represents the missing pathobiologic link between cancer and thrombotic risk in patients with primary brain tumors. Our study suggests that determination of podoplanin expression in brain tumor samples might help to identify patients with very high risk of VTE who might benefit from primary thromboprophylaxis.
The detailed pathophysiology of VTE in patients with brain tumors is still under investigation. It was postulated that cancer cells may cause an activation of the coagulation cascade via upregulation of tissue factor (TF), a potent procoagulatory protein.3 However, definite evidence for this hypothesis is lacking, and recent studies found no association between intratumoral TF expression levels or microparticle-associated TF-activity and risk of VTE in malignant brain tumors.22,23 Our current study indicates a role of podoplanin in development of VTE in those patients through podoplanin-induced platelet aggregation, because the intensity of podoplanin expression was positively correlated to the grade of intratumoral intravascular platelet clusters reflected by CD61 staining. Our in vivo data are supported by in vitro experiments, which confirmed that glioblastoma cells can induce platelet aggregation via podoplanin. We showed that primary glioblastoma cells, newly established from a patient in whom a PE developed during his disease, induced platelet aggregation in vitro, which could be reversed by a podoplanin-blocking antibody. Our experimental data are in line with previously published studies that demonstrated the ability of cancer cells to induce platelet activation24 through podoplanin. Previous studies have identified CLEC-2 as the receptor for podoplanin on platelets.15 Interestingly, a recent study found the interaction of podoplanin and CLEC-2 to be an important driver for thrombosis in inflammation, showing that upregulation of podoplanin on inflammatory cells in the liver and consequent platelet activation induce thrombosis in mice with systemic Salmonella typhimurium infection.25 That study proves the concept that high expression of podoplanin can induce thrombosis in vivo. In future studies, it will be of particular interest to investigate detailed pathophysiologic mechanisms of how podoplanin expression in brain tumor tissue induces thrombosis at distant sites (ie, in the deep vein system of the legs) and PE. With the current state of knowledge, we propose several possible explanations: First, podoplanin might be released into the circulation of brain tumor patients, either in a soluble form or on the surface of tumor-derived microvesicles. The development of reliable measurement techniques to determine podoplanin in the circulation is therefore of utmost importance. Second, circulating tumor cells might be a source of circulating podoplanin. Interestingly, a recent study detected circulating tumor cells in 20% of patients with glioblastoma.26 Trapping of tumor cells in the venous system might lead to local platelet activation and aggregation, potentially initiating and promoting VTE development. Third, local platelet activation and coagulation activation in the brain tumor tissue might induce a hypercoagulable state, which leads to an increased propensity of thrombus formation at sites of slow blood flow and stasis (ie, in the deep vein system of the legs).
In our study, patients harboring podoplanin-expressing tumors had a lower blood platelet count. We propose this might be a consequence of platelet consumption caused by podoplanin-induced platelet aggregation. Podoplanin expression was also associated with higher plasma levels of D-dimer, indicating that high podoplanin expression correlates with a procoagulatory state. We propose that podoplanin-induced platelet aggregation leads to hypercoagulability in patients with brain tumor. Expression of podoplanin was not associated with plasma levels of sP-selectin. High sP-selectin is associated with increased risk of VTE in patients with brain tumor.6 In our current analysis, sP-selectin was only weakly correlated to blood platelet count, and our data may suggest that the contribution of podoplanin to VTE might be independent of sP-selectin.
In line with previous reports, we found an association between high podoplanin expression and poor prognosis of patients with brain tumor.13,14 During the 2-year observation period, patients with high podoplanin–expressing tumors had a 2.6-fold higher risk of mortality compared with patients with podoplanin-negative tumors, after adjusting for tumor type. Podoplanin is known to contribute to tumor progression by inducing cancer cell migration and tumor invasion,27 and it correlates with tumor grade of gliomas.28 Mortality in high podoplanin–expressing tumors might therefore be linked to tumor progression itself, which is the major cause of death in patients with brain tumor. In our study, only 2 of 213 patients were diagnosed with fatal PE during the observation period. However, autopsy is only rarely performed in these patients, and a higher number of fatal PEs can therefore not be excluded.
Some limitations of our study need to be discussed. We cannot provide experimental in vivo evidence for a mechanistic relationship between podoplanin expression and development of VTE. However, a specific advantage of our clinical study is its prospective design, which was a priori and specifically chosen to investigate risk factors for VTE in patients with cancer. VTE rates observed in our study are in concordance with previously published VTE incidence rates, indicating that our study population is representative for patients with brain tumor. Podoplanin expression levels and CD61+ intravascular platelet clusters were determined semiquantitatively. However, 2 researchers experienced in brain histopathology independently reviewed all sections. Podoplanin expression may reflect a more malignant tumor biology, which, through other pathways, leads to a higher risk of VTE. Because the association between podoplanin and tumor aggressiveness might confound our data, we have aimed to limit this potential confounding by multivariable inclusion of tumor grade into time-to-event regression models. Future studies may investigate the role of specific molecular alterations typically found in brain tumors on podoplanin expression and VTE.29 We believe that our data provide a coherent rationale for a role of podoplanin in development of VTE in malignant brain tumors. We propose future in vivo studies using animal models of glioblastoma and thrombosis to confirm our findings. Moreover, we propose future studies on the efficacy and safety of primary thromboprophylaxis in patients with brain tumor who are at high risk of VTE, based on podoplanin expression levels in tumor tissue.
In conclusion, we have shown that expression of podoplanin on primary malignant brain tumors induces platelet aggregation, and is associated with risk of VTE and mortality. Podoplanin expression was linked to vascular platelet aggregates in tumor specimens, decreased blood platelet counts, and high D-dimer, suggesting a functional role of podoplanin-induced platelet activation in hypercoagulability in patients with brain tumor. Because podoplanin might be involved in development of VTE, as well as in cancer progression, the podoplanin-CLEC-2 axis might provide a potential drug target for both reducing the risk of VTE and improving prognosis in patients with brain tumor. Our study provides new insights into the pathophysiology of VTE in patients with primary brain tumors and identifies podoplanin as a novel risk factor for cancer-associated VTE.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Orsoyla Rajky, Anna Berghoff, and Adelheid Wöhrer for their kind help with immunohistologic staining, and Elisabeth Dirnberger and Gerda Ricken for preparation of tumor sections for immunohistolochemistry (Institute of Neurology, Medical University of Vienna); Sabine Spiegl-Kreinecker (Kepler Universitätsklinikum GmbH, Neuromed Campus, Linz, Austria) for kindly providing cancer cell line AMCH; and Anna Zommer (Department of Surgery) for assistance with immunostaining and flow cytometry. They also acknowledge the members of the adjudication committee: Andrea Willfort-Ehringer (Department of Angiology, Medical University of Vienna) and Sylvia Metz-Schimmerl (Department of Diagnostic Radiology, Medical University of Vienna). Finally, they thank Tanja Altreiter for proof-reading of this manuscript.
This study was supported by the Austrian Science Fund (FWF), Special Research Program (SFB) 54, and by a fund of the “Hans und Blanca Moser Stiftung.” The Vienna Cancer and Thrombosis Study (CATS) was supported by funds of the Austrian National Bank (Anniversary Fund, project 14744). Establishment of glioblastoma cell lines was part of a project funded by the Austrian National Bank (project 14279).
Authorship
Contribution: J.R., I.P., and C.A. designed the study; J.R., M.P., P.M.S.N., P.B., S.P., C.B., W.B., D.L., and J.A.H. designed and performed the experiments; J.R., M.P., C.M., J.T., and C.A. recruited patients; J.R., C.A., and F.P. performed statistical analyses; and J.R. and C.A. wrote the article, which was reviewed and edited by all other authors.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Cihan Ay, Clinical Division of Hematology and Hemostaseology, Department of Medicine I, Comprehensive Cancer Center, Medical University of Vienna, Waehringer Guertel 18-20, A-1090 Vienna, Austria; e-mail: cihan.ay@meduniwien.ac.at.