Key Points
Iron overload induces inflammatory responses in both hepatocytes and macrophages.
In hepatocytes, the HNF4α/miR-122 pathway is a molecular target of iron overload.
Abstract
Hepatic iron overload (IO) is a major complication of transfusional therapy. It was generally thought that IO triggers substantial inflammatory responses by producing reactive oxygen species in hepatic macrophages. Recently, a decrease in microRNA-122 (miR-122) expression was observed in a genetic knockout (Hfe−/−) mouse model of IO. Because hepatocyte-enriched miR-122 is a key regulator of multiple hepatic pathways, including inflammation, it is of interest whether hepatocyte directly contributes to IO-mediated hepatic inflammation. Here, we report that IO induced similar inflammatory responses in human primary hepatocytes and Thp-1–derived macrophages. In the mouse liver, IO resulted in altered expression of not only inflammatory genes but also >230 genes that are known targets of miR-122. In addition, both iron-dextran injection and a 3% carbonyl iron–containing diet led to upregulation of hepatic inflammation, which was associated with a significant reduction in HNF4α expression and its downstream target, miR-122. Interestingly, the same signaling pathway was changed in macrophage-deficient mice, suggesting that macrophages are not the only target of IO. Most importantly, hepatocyte-specific overexpression of miR-122 rescued IO-mediated hepatic inflammation. Our findings indicate the direct involvement of hepatocytes in IO-induced hepatic inflammation and are informative for developing new molecular targets and preventative therapies for patients with major hemoglobinopathy.
Introduction
Frequent blood transfusions among patients with thalassemia and sickle cell disease can commonly lead to iron overload (IO), also known as transfusional hemosiderosis.1,2 Excess iron is largely accumulated in the liver, the primary organ for iron recycling and storage, and eventually leads to significant hepatic injury.3 This transfusional IO (tIO)–induced damage is initiated by hepatic inflammation, such as increased transaminase levels.4 Recently, Das et al5 reported that intraperitoneal (i.p.) injection of dextran-iron increased hepatic inflammatory infiltration and the expression of proinflammatory cytokines, such as interleukin-6 (IL-6), IL-1β, and tumor necrosis factor α (TNF-α). However, the cellular and molecular factors underlining these innate immune responses were not further pursued. It is known that too much iron is toxic due to free radical generation through Fenton reactions, which triggers macrophage-mediated innate immune responses. Because excess iron is stored in hepatocytes, it is logical to hypothesize that in addition to the macrophage response, hepatocytes could also contribute to inflammatory signals. Indeed, in patients with hereditary hemochromatosis, hepatocytes are massively loaded with iron, which leads to liver cirrhosis, suggesting that hepatocyte iron loading might contribute to inflammation.6
MicroRNAs (miRNAs) are a class of short noncoding RNAs that regulate genes at the posttranscriptional level.7 MicroRNA-122 (miR-122), the predominant miRNA in hepatocytes, comprises up to 70% of the total hepatic miRNA population.8 Previous findings indicated that miR-122 is a key regulator of multiple hepatic pathways during liver development, fat metabolism, inflammation, and tumorigenesis.9 An increase in infiltrating inflammatory cells was observed in the liver of miR-122-deficient mice.10,11 Recently, miR-122 was observed to be downregulated in a genetic model of IO in hemochromatosis protein–deficient (Hfe−/−) mice.12 However, this genetic model does not mimic clinical observations, because most patients with tIO have no mutation in the Hfe gene.13 In addition, miR-122 is highly hepatocyte enriched,14 conflicting with the general concept that excess iron induces inflammatory responses through macrophages.15 In other words, it is unclear whether the miR-122 in hepatocytes directly contributes to IO-induced inflammation. Furthermore, the upstream molecular regulator that disrupts miR-122 expression under IO is currently unknown.
Taken together, the present study was designed to fully investigate, both in vitro and in vivo, the direct involvement of hepatocytes and the role of the HNF4α/miR-122 pathway in tIO-induced hepatic inflammation.
Materials and methods
Cell cultures and chemicals
Huh7 cells were described previously.16 HL-7702 and LX-2 cells were purchased from Shanghai Cell Biology Institute, Chinese Academy of Sciences. Cells were grown in high-glucose Dulbecco’s modified Eagle medium or 1640 medium, with 10% fetal bovine serum (v/v), 100 IU/mL penicillin, and 50 μg/mL streptomycin sulfate. PMA (Sigma) was used at 50 ng/mL for 48 hours to induce differentiation of Thp-1 cells from monocytes to macrophages. Human primary hepatocytes were purchased from ScienCell (catalog no. 5200), from which potential biological contaminants (HIV, hepatitis B virus, hepatitis C virus, and mycoplasma) were detected as negative. Mouse primary hepatocytes and macrophages were isolated from mouse liver using EasySep Do-It-Yourself Selection Kit (STEMCELL Technologies, catalog no. 18098). Fetal bovine serum was purchased from Gibco. All other medium, supplements, and Dulbecco’s phosphate-buffered saline (PBS) for cell culture were purchased from HyClone. Apo-transferrin (apo-Tf) and holo-transferrin (holo-Tf) were purchased from Merck Millipore. Both of these transferrin preparations lack lipolysaccharide, as indicated by Limulus amebocyte lysate assays using the Rapid Endotoxin Detection Pyrosate Kit (Associates of Cape Cod). The miR-122 inhibitor and miR-122 mimics were purchased from RiboBio.
Transfection in vitro
Cell transfection was performed by lipofectamine RNAiMAX (for miRNA inhibitors and mimics) or lipofectamine 2000 (for plasmids) according to the manufacturer’s instructions.17 Cells were harvested 24 hours posttransfection for the downstream experiments, such as quantitative reverse transcription polymerase chain reaction (qRT-PCR) and western blot analysis.
Western blot analysis
Western blot assays were performed as previously described,18 with modifications. Briefly, the Total Protein Extraction Kit from EMD Millipore was used. Following normalization for protein concentration, samples were separated using 12% SDS-PAGE electrophoresis, electrotransferred to a polyvinylidene fluoride membrane, and probed with relevant primary antibodies at 4°C overnight (CCL2 antibodies, 1:400; p65 antibody, 1:1000; IκBα antibody, 1:1000; p-IκBα antibody, 1:1000; TNF-α, 1:1000; and β-actin antibody, 1:3000). All antibodies were purchased from Cell Signaling Technology, Proteintech, or Santa Cruz Biotechnology. Grayscale image analysis using GeneSnap from the SynGene software package was normalized to β-actin.
qRT-PCR
Total RNA from tissues or cells were extracted using TRIzol from Thermo Fisher and purified using the RNeasy mini kit from QIAGEN. One microgram total RNA was reverse transcribed into complementary DNA using the RT regent kit with oligo-d(T) or random 6 mers primers for messenger RNA (mRNA) or special RT stem-loop primers for miRNA. Mixed complementary DNA templates, primers, and SYBR green reagents were subjected to the StepOnePlus Real-Time PCR System from Thermo Fisher. All primers are listed in supplemental Table 3 (available on the Blood Web site). The results were normalized to β-actin (for mRNA) or U6 (for miRNA).
ChIP analysis
Following treatments, Huh7 cells were crosslinked and processed according to EMD Millipore chromatin immunoprecipitation (ChIP) assay kit protocol. The antibodies against HNF4α were purchased from Abcam. The primers used for polymerase chain reaction were 5′-GACAAGGTTCCCCTATTATCAGTG-3′ (forward) and 5′-CAAAGCAAACGATGCCAAGAC-3′ (reverse), which amplified 145 bp of the human miR-122 promoter.
Animal studies
All animal studies were performed at the Second Military Medical University, Shanghai, China, under Institutional Animal Care and Use Committee approval. C57BL/6 mice, weighing 13 ± 2 g (mean ± standard deviation [SD]) at the age of 4 weeks, and nonobese diabetic/severe combined immunodeficiency (NOD/SCID) mice, weighing 15.5 ± 1.1 g (mean ± SD) at the age of 5 to 6 weeks, were purchased from the SLAC Laboratory Animal Co. (Shanghai, China). C57BL/6 mice were fed the AIN-93G growing rodent diet with no added iron (Research Diets), further divided into 3 subgroups, and received an i.p. injection. The iron-deficient (ID) subgroup was injected with dextran, the iron-adequate (IA) subgroup was injected with dextran-iron at 20 mg/kg mouse weight, which mimics daily consume from standard food. The IO group was injected with dextran-iron at 200 mg/kg mouse weight. All injections were administrated at a frequency of twice per week and a volume of 0.2 mL. Dextran-iron was purchased from Sigma-Aldrich. NOD/SCID mice were fed a cereal-based standard diet and i.p. injected with dextran-iron at 200 mg/kg mouse weight to induce IO. Alternatively, C57BL/6 mice were fed a 3% iron-rich diet for 1 month to induce IO.
Iron status parameters
Liver iron levels were quantitated using an atomic absorption spectrophotometer (Z-8100; Hitachi, Tokyo, Japan) and normalized to the wet tissue weight for each sample. Serum iron concentrations and total iron-binding capacity in nonhemolyzed serum samples were determined using the KX-21 automated hematology analyzer (Sysmex). Transferrin saturation was calculated as plasmatic iron/total iron-binding capacity.
Microarray analysis
A total of 15 RNA samples (5 in each group [ID, IA, and IO]) were isolated from mouse liver using TRIzol reagent (Invitrogen, Shanghai, China) and processed using the Affymetrix GeneChip Mouse Gene 1.0 ST Array (Invitrogen) to profile patterns of mRNA expression. The random-variance model (commonly used to compare >2 groups) F-test was applied to filter the differentially expressed genes for the control and experiment groups, because this test can raise the degrees of freedom effectively in cases with small samples. After the significance and false discovery rate analyses were performed, we selected differentially expressed genes according to the P value threshold.
Liver tissue immunofluorescence analysis
Tissue fixation was performed as previously described.19 Frozen liver sections (8 μm) were fixed and permeabilized in cold acetone and then blocked with 13% (v/v) donkey serum (Sigma) and 8.7% (v/v) fetal bovine serum in PBS. Primary mouse monoclonal anti–mouse CD68, TNF-α, and IL-1β antibodies (1:50 dilution) and the fluorescein isothiocyanate–conjugated secondary antibody were purchased from Abcam. DAPI, which stains nuclei, was also used. After staining, 5 images (20× objectives) from each liver section were analyzed using a fluorescence microscope.
Liver tissue inflammation score
Scoring was quantified by a pathologist based on hematoxylin and eosin (H&E)–stained slides. A semiquantitative scale was used (score 0, none; score 1, rare/mild; score 2, common/moderate; and score 3, frequent/severe).
rAAV vector production
Preparation and purification of recombinant adeno-associated virus (rAAV) vectors by triple-plasmid transfection have been described previously.20 All vector preparations were subjected to quality control tests, including western blot analysis,21 quantitative polymerase chain reaction,22 and Southern blot analysis,23 as previously described. The highly purified rAAV serotype 8 vectors were then administered intravenously via tail vein into IO mice at 10E11 viral genome per mouse in 100 μL PBS.
Statistics
A Student t test was used for 2-group comparisons. One-way analysis of variance, followed by Bonferroni test as a post-test, was performed to analyze the differences among the ≥3 groups. For data that did not follow a normal distribution, we used a Mann-Whitney test for 2-group comparisons and a Kruskal-Wallis test for multiple-group comparisons with the Dunn’s multiple comparison test as a follow-up test.
Results
Treatment of hepatic cell lines with excess iron results in inflammatory responses
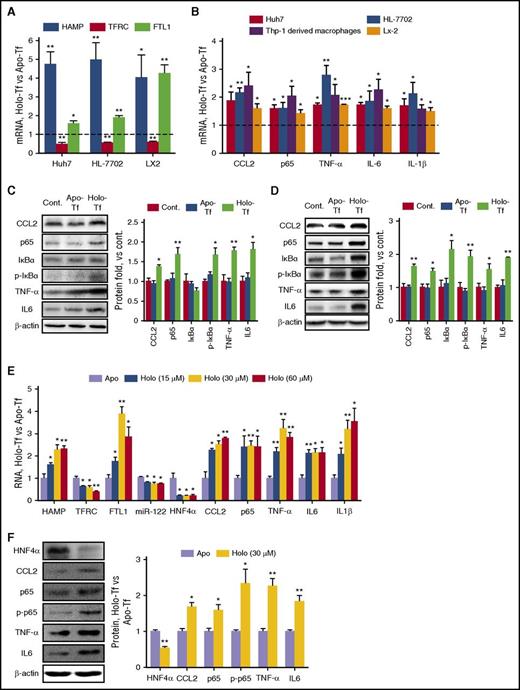
We first demonstrated that concentrations >30 µM holo-Tf had significant cytotoxicity on cell viability at 24 hours after exposure (supplemental Figure 1A). Meanwhile, this concentration led to substantial intracellular iron content (supplemental Figure 1B) and altered the expression of hepcidin (HAMP), transferrin receptor 1 (TFRC), and ferritin light polypeptide 1 (FTL1), which is consistent with other reports (Figure 1A).24 Interestingly, the mRNA levels of inflammatory factors (CCL2, NF-κB/p65, and TNF-α) and proinflammatory cytokines (IL-6 and IL-1β) were significantly increased upon iron treatment in various human hepatic cell lines (Figure 1B), including a hepatocellular carcinoma (HCC) cell line (Huh7), a hepatic cell line that retains many characteristics of primary human hepatocytes (HL-7702),25 and a hepatic stellate cell line (LX2). As an appropriate control, human Thp-1–derived macrophages were also included. Western blot assays also confirmed significantly increased protein expression upon treatment of holo-Tf, but not apo-Tf, an iron-free form of transferrin (Figure 1C-D). To further corroborate our observations based on cell lines, human primary hepatocytes were treated with increasing levels of holo-Tf, and similar results were obtained in a dose-dependent manner (Figure 1E-F). These results suggested that excess iron leads to CCL2-NF-κB–mediated inflammatory responses in both hepatocytes and macrophages in vitro.
IO induces inflammatory responses in hepatocyte cell lines in vitro. (A) Cells were treated with 30 μM apo-Tf or holo-Tf. mRNA expression levels were determined 24 hours posttreatment. (B) Various human hepatic cell lines and Thp-1–derived macrophages were treated with 30 μM apo-Tf or holo-Tf. Total mRNA was isolated 24 hours posttreatment. (C-D) Huh7 (C) and HL-7702 (D) cells were either mock treated (Cont.) or treated with 30 μM apo-Tf or holo-Tf. Protein levels of various inflammatory factors were determined 24 hours posttreatment. (E-F) Human primary hepatocytes were treated with holo-Tf at the indicated concentrations and apo-Tf as an appropriate control. Total RNA and proteins were isolated 24 hours posttreatment. Data were normalized to the control group and are presented as mean ± standard error of the mean (SEM) (N = 4). *P < .05; **P < .01 vs control.
IO induces inflammatory responses in hepatocyte cell lines in vitro. (A) Cells were treated with 30 μM apo-Tf or holo-Tf. mRNA expression levels were determined 24 hours posttreatment. (B) Various human hepatic cell lines and Thp-1–derived macrophages were treated with 30 μM apo-Tf or holo-Tf. Total mRNA was isolated 24 hours posttreatment. (C-D) Huh7 (C) and HL-7702 (D) cells were either mock treated (Cont.) or treated with 30 μM apo-Tf or holo-Tf. Protein levels of various inflammatory factors were determined 24 hours posttreatment. (E-F) Human primary hepatocytes were treated with holo-Tf at the indicated concentrations and apo-Tf as an appropriate control. Total RNA and proteins were isolated 24 hours posttreatment. Data were normalized to the control group and are presented as mean ± standard error of the mean (SEM) (N = 4). *P < .05; **P < .01 vs control.
Excess iron reduces HNF4α and miR-122 expression in vitro
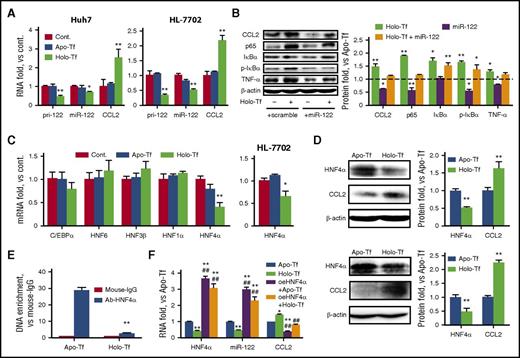
Because miR-122 plays an essential role in hepatic inflammation and is expressed in a hepatocyte-enriched manner, we wished to evaluate the effect of excess iron on miR-122 expression. The results, as shown in Figure 2A, indicated that the expression of both mature miR-122 and pri–miR-122 in Huh7 and HL-7702 cells was significantly inhibited by holo-Tf treatment. In addition, the iron-induced expression of inflammatory factors, such as CCL2, NF-κB/p65, IκBα, and TNF-α, can be restored by transfection of miR-122 mimics, but not control scramble miRNA (Figure 2B). To date, 5 transcriptional factors, namely HNF1α, HNF4α, HNF3β, HNF6, and c/EBPa, have been reported to regulate miR-122 expression.26 Only the expression of HNF4α was significantly reduced in the presence of holo-Tf (Figure 2C), and this finding was confirmed by western blot analysis (Figure 2D). Furthermore, ChIP assays were performed using either anti–HNF4α antibody or negative control immunoglobulin G. The results indicated that holo-Tf significantly reduced the physical interaction between HNF4α and its core elements in the miR-122 promoter (Figure 2E). To further investigate the role of HNF4α, it was overexpressed by a plasmid (oeHNF4α), as shown in supplemental Figure 2A. Consistently, expression levels of miR-122, CCL2, and other HNF4α-targeting proteins27,28 were significantly altered (supplemental Figure 2B). Most importantly, when holo-Tf–treated cells were transfected with oeHNF4α, the expression of CCL2 was restored to a normal level, suggesting that holo-Tf regulates CCL2 expression through the HNF4α signaling pathway (Figure 2F).
IO negatively regulates HNF4α and miR-122 expression in hepatocyte cell lines in vitro. (A) Huh7 and HL-7702 cells were treated as indicated. Total RNA was isolated 24 hours posttreatment. (B) Huh7 cells were treated with 30 μM apo-Tf or holo-Tf. Both groups were transfected with either miR-122 mimics or scramble controls. The protein levels of various inflammatory factors were determined 24 hours posttreatment. (C) Huh7 (left) and HL-7702 (right) cells were treated as indicated. Total RNA was isolated at 24 hours posttreatment. (D) Huh7 (top) and HL-7702 (bottom) cells were treated with 30 μM apo-Tf or holo-Tf. CCL2 and HNF4α protein expression was determined 24 hours posttreatment. (E) Huh7 cells were treated with 30 μM apo-Tf or holo-Tf. ChIP assays were performed using anti–HNF4α monoclonal antibody, followed by quantitative polymerase chain reaction analysis using primers specific to the miR-122 promoter region. Mouse immunoglobulin G (IgG) was used as a negative control. (F) Huh7 cells were treated with 30 μM apo-Tf or holo-Tf. Both groups were transfected with either HNF4α overexpression plasmids (oeHNF4α) or control plasmids. Total RNA was isolated 24 hours posttreatment. Data were normalized to the control group and are presented as mean ± SEM (N = 4). *P < .05; **P < .01 vs apo-Tf; #P < .05; ##P < .01 vs holo-Tf.
IO negatively regulates HNF4α and miR-122 expression in hepatocyte cell lines in vitro. (A) Huh7 and HL-7702 cells were treated as indicated. Total RNA was isolated 24 hours posttreatment. (B) Huh7 cells were treated with 30 μM apo-Tf or holo-Tf. Both groups were transfected with either miR-122 mimics or scramble controls. The protein levels of various inflammatory factors were determined 24 hours posttreatment. (C) Huh7 (left) and HL-7702 (right) cells were treated as indicated. Total RNA was isolated at 24 hours posttreatment. (D) Huh7 (top) and HL-7702 (bottom) cells were treated with 30 μM apo-Tf or holo-Tf. CCL2 and HNF4α protein expression was determined 24 hours posttreatment. (E) Huh7 cells were treated with 30 μM apo-Tf or holo-Tf. ChIP assays were performed using anti–HNF4α monoclonal antibody, followed by quantitative polymerase chain reaction analysis using primers specific to the miR-122 promoter region. Mouse immunoglobulin G (IgG) was used as a negative control. (F) Huh7 cells were treated with 30 μM apo-Tf or holo-Tf. Both groups were transfected with either HNF4α overexpression plasmids (oeHNF4α) or control plasmids. Total RNA was isolated 24 hours posttreatment. Data were normalized to the control group and are presented as mean ± SEM (N = 4). *P < .05; **P < .01 vs apo-Tf; #P < .05; ##P < .01 vs holo-Tf.
Genome-wide microarray analysis in vivo
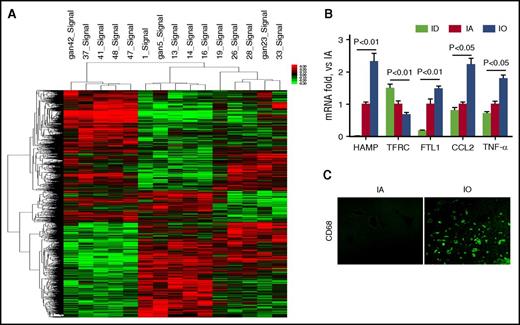
To examine changes in gene expression in the liver, C57BL/6 mice were fed an iron-free diet and separated into ID, IA, and tIO groups as described in “Materials and methods.” Two weeks after iron injection, total mRNA was isolated from mouse liver and subjected to Affymetrix GeneChip Mouse Gene 1.0 ST Array analysis to profile gene expression. As shown in Figure 3A, out of 28 853 mouse genes, 3282 candidates were identified as differentially expressed (P < .05). The expression of HAMP, FTL1, and bone morphogenetic protein 6 (Bmp6) was significantly upregulated upon tIO, while the expression of TFRC was downregulated (supplemental Table 1), both validating the tIO treatment.29,30 Interestingly, at least 230 altered genes are known to be regulated by liver-enriched miR-122. In addition, as shown in supplemental Table 1, functional annotation of the gene list revealed enrichment of genes related to inflammatory responses, such as surface antigens of monocyte/macrophage, chemokines, chemokine receptors, and proinflammatory cytokines. Selected genes were further confirmed by qRT-PCR assays using the same RNA samples (Figure 3B). Furthermore, immune-fluorescent staining against CD68 confirmed iron-induced liver inflammation (Figure 3C).
Genome-wide microarray assay reveals differential expressed genes under IA conditions. C57BL/6 mice (N = 5) were fed a purified diet with no added iron and separated into ID, IA, and IO groups. Two weeks after iron injection, total hepatic RNA was subjected to Affymetrix GeneChip Mouse Gene 1.0 ST Array analysis to profile gene expression. (A) Cluster analysis of microarray showed different transcriptional profiles that were influenced by iron homeostasis. (B) Total hepatic RNA was subjected to qRT-PCR to determine HAMP, TFRC, FTL1, CCL2, and TNF-α expression. (C) Liver tissue was subjected to the immunofluorescent staining against CD68. Original magnification ×400.
Genome-wide microarray assay reveals differential expressed genes under IA conditions. C57BL/6 mice (N = 5) were fed a purified diet with no added iron and separated into ID, IA, and IO groups. Two weeks after iron injection, total hepatic RNA was subjected to Affymetrix GeneChip Mouse Gene 1.0 ST Array analysis to profile gene expression. (A) Cluster analysis of microarray showed different transcriptional profiles that were influenced by iron homeostasis. (B) Total hepatic RNA was subjected to qRT-PCR to determine HAMP, TFRC, FTL1, CCL2, and TNF-α expression. (C) Liver tissue was subjected to the immunofluorescent staining against CD68. Original magnification ×400.
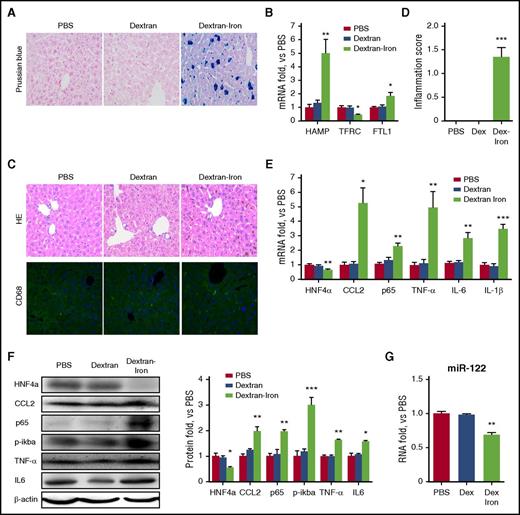
tIO reduces HNF4α and miR-122 expression in the mouse liver
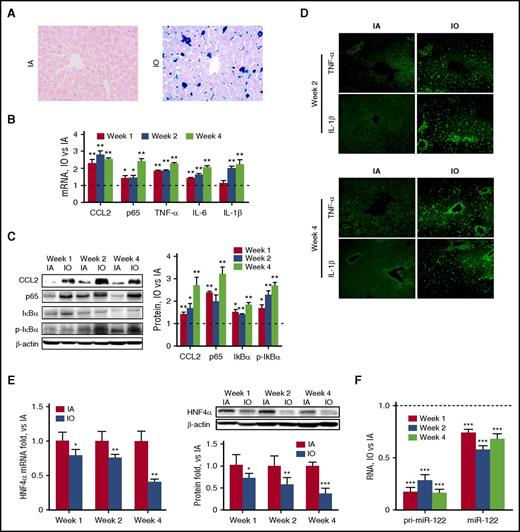
Next, another group of C57BL/6 mice were divided into IA and tIO groups. As shown in Table 1, liver and serum iron content, as well as other serum iron indices, was significantly enhanced over time upon tIO. Prussian blue–stained liver sections showed normal tissue architecture with few stainable cells in the IA group. In contrast, heavy iron accumulation was observed in the tIO group in not only nonparenchymal cells but also hepatocytes (Figure 4A). Consistent with other reports,5 tIO led to a time-dependent increase in inflammatory factors and proinflammatory cytokines at both the mRNA (Figure 4B) and protein levels (Figure 4C-D). The observation of immune cell infiltration by H&E staining further confirmed hepatic inflammation (supplemental Figure 3A). Consequently, both hepatic inflammation scores (supplemental Figure 3B) and serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activity (supplemental Figure 3C) were significantly increased upon iron administration. To investigate the molecular mechanism in vivo, we first evaluated that tIO significantly reduced the expression of HNF4α in a time-dependent manner (Figure 4E). The levels of mature and pri–miR-122 were also determined, indicating that both were significantly reduced, presumably as a result of the reduction in HNF4α (Figure 4F). To further distinguish the role of parenchymal and nonparenchymal cells, double immune-fluorescent staining assays were performed, and results indicated that downregulation of HNF4α and upregulation of CCL2 upon tIO treatment occurred in both hepatocytes and macrophages (supplemental Figure 4).
IO induces significant hepatic inflammation in vivo. C57BL/6 mice (N = 6) were fed a purified diet with no added iron and subjected to i.p. injection of 20 mg/kg (IA) or 200 mg/kg (IO) dextran-iron twice per week. Mice were sacrificed, and liver tissues were harvested at various time points. (A) Prussian blue staining shows heavy iron accumulation. Original magnification ×400. (B) RNA and (C) protein levels of various inflammatory factors were determined. (D) Immunofluorescence analysis showed upregulation of TNF-α and IL-1β expression. Original magnification ×200. (E-F) Expression of HNF4α at the mRNA and protein levels (E) and expression of miR-122 in the pri- and mature forms (F) were determined. Data were normalized to the IA group and are presented as mean ± SEM. *P < .05; **P < .01; ***P < .001 vs IA.
IO induces significant hepatic inflammation in vivo. C57BL/6 mice (N = 6) were fed a purified diet with no added iron and subjected to i.p. injection of 20 mg/kg (IA) or 200 mg/kg (IO) dextran-iron twice per week. Mice were sacrificed, and liver tissues were harvested at various time points. (A) Prussian blue staining shows heavy iron accumulation. Original magnification ×400. (B) RNA and (C) protein levels of various inflammatory factors were determined. (D) Immunofluorescence analysis showed upregulation of TNF-α and IL-1β expression. Original magnification ×200. (E-F) Expression of HNF4α at the mRNA and protein levels (E) and expression of miR-122 in the pri- and mature forms (F) were determined. Data were normalized to the IA group and are presented as mean ± SEM. *P < .05; **P < .01; ***P < .001 vs IA.
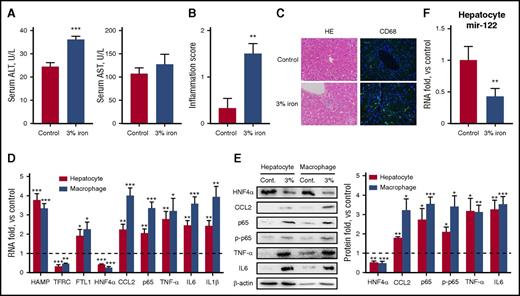
Mice fed an iron-rich diet shows decreased HNF4α and miR-122 expression in hepatocytes
To extend our findings to other IO conditions, a group of mice were fed a 3% carbonyl iron-containing diet for 1 month and then compared with age-matched animals kept on standard chow. Consistent with previous reports,31 liver iron content and other serum iron indices were significantly higher (Table 1). Meanwhile, serum transaminase levels (Figure 5A), hepatic inflammation scores (Figure 5B), and immune cell infiltration (Figure 5C) were significantly higher in iron-fed mice. Most importantly, hepatocytes and macrophages were isolated from both control and IO mice, and it was evident that under chronic IO conditions, upregulation of CCL2 and its downstream inflammatory genes occurred in both macrophages and hepatocytes (Figure 5D-E). Regarding the molecular mechanisms involved, and similar to the results obtained in iron-injected mice, an iron-rich diet led to a significant reduction in hepatic HNF4α and miR-122 expression (Figure 5E-F).
An iron-rich diet leads to increased inflammatory responses and reduced HNF4α/miR-122 expression in hepatocytes. C57BL/6 mice (N = 6) were fed with either a standard diet or a 3% iron-rich diet for 1 month. (A-B) Serum ALT and AST levels (A) and hepatic inflammation scores (B) were determined. (C) H&E and immunofluorescent staining against CD68 showed inflammatory infiltration in the liver. Blue represents DAPI stain. Original magnification ×400. (D-E) mRNA (D) and protein (E) levels of various inflammatory factors and HNF4α were determined from isolated hepatocytes and macrophages. (F) Expression of miR-122 was determined. Data were normalized to the control group and are presented as mean ± SEM. *P < .05; **P < .01 vs control.
An iron-rich diet leads to increased inflammatory responses and reduced HNF4α/miR-122 expression in hepatocytes. C57BL/6 mice (N = 6) were fed with either a standard diet or a 3% iron-rich diet for 1 month. (A-B) Serum ALT and AST levels (A) and hepatic inflammation scores (B) were determined. (C) H&E and immunofluorescent staining against CD68 showed inflammatory infiltration in the liver. Blue represents DAPI stain. Original magnification ×400. (D-E) mRNA (D) and protein (E) levels of various inflammatory factors and HNF4α were determined from isolated hepatocytes and macrophages. (F) Expression of miR-122 was determined. Data were normalized to the control group and are presented as mean ± SEM. *P < .05; **P < .01 vs control.
Hepatic macrophages are not essential for tIO-induced inflammatory responses
To further distinguish the role of macrophages and hepatocytes, immune-deficient NOD/SCID mice (deficient in macrophages and other immune cells) were fed a standard diet and intraperitoneally injected with PBS, dextran, or dextran-iron (supplemental Table 2). Hepatic iron accumulation was confirmed by Prussian blue staining (Figure 6A). As expected, only upon administration of dextran-iron was the expression of hepcidin and FTL1 significantly increased in mouse liver, whereas the expression of TFRC was reduced, further confirming the tIO condition (Figure 6B). Interestingly, although both H&E and immunofluorescence staining failed to detect infiltration of macrophages in the liver (Figure 6C), the hepatic inflammation score was still significantly elevated (Figure 6D). Further experiments revealed that tIO treatment led to increased expression of chemokines and proinflammatory cytokines, such as CCL2, TNF-α, and IL-6 (Figure 6E-F). This was accompanied by significantly reduced expression of HNF4α and its downstream target, miR-122 (Figure 6G), similar to previous results observed in immune-competent mice. Given that there was no hepatic macrophage infiltration after tIO treatment in NOD/SCID mice and that hepatocytes express levels of miR-122 that are at least 100-fold higher than those of macrophages,14 we concluded that macrophages are not the only source of inflammatory responses induced by tIO.
IO-induced hepatic inflammation is independent of macrophages. NOD/SCID mice (N = 6) were fed a standard diet and subjected to i.p. injection of 200 mg/kg (IO) dextran-iron twice per week. Mice were sacrificed, and liver tissues were harvested at week 4. (A) Prussian blue staining showed heavy iron accumulation. Original magnification ×400. (B) Total RNAs were subjected to qRT-PCR assays. (C) H&E and immunofluorescent staining against CD68 showed no macrophage infiltration in the liver. Blue represents DAPI stain. Original magnification ×400. (D) The inflammation scores of liver sections were determined. (E-F) Total RNA (E) and proteins (F) were subjected to qRT-PCR and western blot assays, respectively. (G) The level of mature miR-122 expression was determined. Data were normalized to the the PBS group and are presented as mean ± SEM. *P < .05; **P < .01 vs PBS.
IO-induced hepatic inflammation is independent of macrophages. NOD/SCID mice (N = 6) were fed a standard diet and subjected to i.p. injection of 200 mg/kg (IO) dextran-iron twice per week. Mice were sacrificed, and liver tissues were harvested at week 4. (A) Prussian blue staining showed heavy iron accumulation. Original magnification ×400. (B) Total RNAs were subjected to qRT-PCR assays. (C) H&E and immunofluorescent staining against CD68 showed no macrophage infiltration in the liver. Blue represents DAPI stain. Original magnification ×400. (D) The inflammation scores of liver sections were determined. (E-F) Total RNA (E) and proteins (F) were subjected to qRT-PCR and western blot assays, respectively. (G) The level of mature miR-122 expression was determined. Data were normalized to the the PBS group and are presented as mean ± SEM. *P < .05; **P < .01 vs PBS.
Overexpression of miR-122 reduces tIO-mediated hepatic inflammation in a hepatocyte-specific manner
To substantially support our hypothesis that hepatocyte-enriched miR-122 is involved in the inflammatory responses induced by tIO, we took advantage of a hepatocyte-targeting rAAV vector that is well known to provoke a small and transient innate immune response in mice.32 A miR-122 expression cassette was embedded inside the intron region between a Gaussia luciferase (Gluc) reporter transgene and a hepatocyte-specific TTR promoter (supplemental Figure 5A). The experimental protocol is schematically presented as supplemental Figure 5B. Two weeks after viral injection, levels of serum Gluc in both rAAV vector-injected groups were determined to be similar (supplemental Figure 5C), validating the dose of viral vectors. Results of qRT-PCR analysis indicated that administration of rAAV-122 vectors led to a 12-fold increase in miR-122 expression in the liver (Figure 7A). On the other hand, only a modest increase (<1.5-fold) in HNF4α expression was observed (supplemental Figure 6A). Upon administration of either rAAV or rAAV-122 vectors, liver and serum iron indices were unchanged (Table 2) and hepatic iron-related molecules were unaffected (Figure 7B). Interestingly, overexpression of miR-122 in hepatocytes significantly reduced the expression of tIO-induced inflammatory factors (Figure 7B). Immunofluorescence staining further confirmed a significant reduction in CD68, TNF-α, and IL-1β levels in tIO + rAAV-122–injected mouse liver compared with mouse liver injected with the viral vector counterparts (Figure 7C; supplemental Figure 6B). Western blot analysis also revealed that, consistent with our above experiments, tIO led to significantly enhanced CCL2 and NF-κB/p65 protein expression in mouse liver. Meanwhile, overexpression of miR-122 in hepatocytes resulted in a significant reduction in these proteins, whereas the control viral vector had no effect (Figure 7D). Consequently, both hepatic inflammation scores and serum AST and ALT activities were significantly reduced upon miR-122 overexpression (Figure 7E). Taken together, we conclude that tIO results in a reduction in hepatocyte-enriched miR-122 levels and that miR-122 overexpression in hepatocytes has the ability to restore, at least partially, tIO-mediated hepatic inflammation.
Overexpression of miR-122 in hepatocytes rescues the hepatic inflammation mediated by IO in vivo. C57BL/6 mice (N = 6) were fed a purified diet with no added iron and subjected to i.p. injection of 20 mg/kg (IA) or 200 mg/kg (IO) dextran-iron. rAAVs with or without a miR-122 transgene were injected via tail vein at day 6. Mice were sacrificed, and liver tissues were harvested at week 4. (A-B) RNA levels of miR-122 (A) and various iron homeostasis-related genes (B) and inflammatory factors were determined by qRT-PCR. (C) The protein levels of CCL2 and NF-κB/p65 were determined by western blot assays. Both representative figures and quantitative data are shown. (D) Immunofluorescence analysis showing the expression of CD68, TNF-α, and IL-1β expression after treatment. Original magnification ×200. (E) The inflammation score in the liver and serum ALT and AST levels were determined. Data were normalized to the IA group and are presented as mean ± SEM. *P < .05; **P < .01 vs IO.
Overexpression of miR-122 in hepatocytes rescues the hepatic inflammation mediated by IO in vivo. C57BL/6 mice (N = 6) were fed a purified diet with no added iron and subjected to i.p. injection of 20 mg/kg (IA) or 200 mg/kg (IO) dextran-iron. rAAVs with or without a miR-122 transgene were injected via tail vein at day 6. Mice were sacrificed, and liver tissues were harvested at week 4. (A-B) RNA levels of miR-122 (A) and various iron homeostasis-related genes (B) and inflammatory factors were determined by qRT-PCR. (C) The protein levels of CCL2 and NF-κB/p65 were determined by western blot assays. Both representative figures and quantitative data are shown. (D) Immunofluorescence analysis showing the expression of CD68, TNF-α, and IL-1β expression after treatment. Original magnification ×200. (E) The inflammation score in the liver and serum ALT and AST levels were determined. Data were normalized to the IA group and are presented as mean ± SEM. *P < .05; **P < .01 vs IO.
Discussion
Frequent blood transfusions are essential for patients with major hemoglobinopathy33 and bone marrow disorders.34 Unfortunately, as each unit of transfused blood contains ∼250 mg iron and most patients receive 1 blood transfusion of 2 U each month, excess iron rapidly accumulates in the liver.35 It was generally thought that tIO triggers hepatic inflammation via macrophages and reactive oxygen species.36 However, a recent clinical observation indicated that antioxidant therapy has little effect on hepatic inflammation in β-thalassemia patients with tIO.37 These findings triggered us to further pursue the potential underlying cellular and molecular mechanisms.
In our studies, we used both transfusional and iron-rich diet models to exclude artificial effects. Similar results were observed in 2 models in which inflammatory responses occurred not only in hepatic macrophages but also in hepatocytes. The use of NOD/SCID mice further pointed out that these responses are at least partially independent of macrophages. The most conclusive evidence came from the results of hepatocyte-specific overexpression of miR-122 by rAAVs. It is worth noting that a large vector dose is required to transduce 100% of hepatocytes in the mouse liver,38 which is at least 4 times higher than the dose used in our studies. This may account for the partial restoration of liver inflammation scores in Figure 7E. Interestingly enough, Das et al5 reported that resveratrol, a plant phenol, has the therapeutic ability to prevent the inflammatory phenotypes associated with hepatic IO. Meanwhile, an independent group observed that the same chemical increases miR-122 expression in the liver.39 These observations, along with our present data that overexpression of miR-122 in hepatocytes significantly reduced hepatic inflammation scores, further expand treatment strategies. Next, we would like to perform similar studies using a hemoglobin-deficient mouse model, such as human hematopoietic stem cell transplanted thalassemia mice.
The risk of developing HCC was recently reported to be significantly higher in patients with thalassemia than in an age-matched healthy population,40 yet the molecular mechanism is unknown. We demonstrate here that excess iron significantly reduced miR-122 expression. The miR-122 level was reported to be reduced in human HCC biopsy specimens.41 In 2012, 2 independent groups observed significantly increased rates of HCC development in miR-122–knockout mouse models.10,11 Furthermore, Hsu et al suggested that the tumor-suppressor effect of miR-122 is independent of its role in nutrition metabolism or hepatic inflammation.10 Thus, our data provide a reasonable explanation for IO-induced HCC via inhibition of miR-122 expression. Next, it would be of interest to determine whether IO accelerates HCC development in miR-122–knockout mice.
Based on our data, we propose that both hepatocytes and macrophages are the cellular targets of IO that trigger hepatic inflammation. However, the exact molecular mechanisms may differ between these 2 cell types. Our data, both in vitro and in vivo, suggest that the HNF4α/miR-122 pathway in hepatocytes contributes to IO-mediated hepatic inflammation. Because miR-122 is a highly hepatocyte-enriched miRNA, macrophages may use other molecular mechanisms, such as reactive oxygen species generation and lysosomal injury, as recently proposed.31 Taken together, our study will be important and informative in the elucidation of new molecular targets of IO-induced hepatic inflammation and for the development of clinical strategies used to treat patients with major hemoglobinopathy.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Changquan Ling, Yuan Wang, and Zifei Yin at the Second Military Medical University for the production of rAAV vectors.
C.L. is sponsored by the Shanghai Eastern Scholarship and the Shanghai Sailing Program (grant 17YF1401300). This work was supported by the National Institutes of Health, National Institute of General Medical Sciences (grant 1R01GM119186-01A1) (C.L.) and the National Natural Science Foundation of China (grant 31671236) (M.L.). Y.T. is a PhD candidate at the Second Military Medical University, and this work was submitted in partial fulfillment of the PhD requirement.
Authorship
Contribution: X.W. and R.Q. performed in vitro experiments; H.Z. and Y.T. performed animal intervention; L.W. and H.L. assessed, measured, and quantified the results of in vivo experiments; F.M. produced the rAAVs and performed related in vivo experiments; M.L. and C.L. designed experiments; and M.L., A.S., and C.L. analyzed the data and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Chen Ling, Room C125, School of Life Science, 2005 Songhu Rd, Shanghai 200438, China; e-mail: lingchenchina@fudan.edu.cn; and Min Li, Department of Military Hygiene, 800 Xiangyin Rd, Shanghai 200433, China; e-mail: linlimin115@hotmail.com.
References
Author notes
M.L., Y.T., and L.W. contributed equally to this study.