Abstract
Background:
The dynamic international prognostic scoring system (DIPSS)-plus currently offers the most comprehensive risk assessment tool in primary myelofibrosis (PMF) (JCO 2011;29:392). More recently, DIPSS-plus-independent genetic determinants of prognosis have been described; type 1/like CALR mutations predicted superior (Blood 2014;124:2465) and ASXL1 / SRSF2 mutations and very high risk (VHR) karyotype inferior survival (Leukemia 2013;27:1861; Blood 2011;118: 4595) . The objective of the current study was to enhance the prognostic value of DIPSS-plus, by incorporating genetic information.
Methods:
Learning and validation cohorts were recruited from the Mayo Clinic and University of Florence, respectively. Diagnosis was according to WHO criteria (Blood 2016;127:2391). The study required availability of complete information on DIPSS-plus, type 1/like CALR, ASXL1 and SRSF2 mutations and karyotype. Previously published methods were used for mutation analyses and determination of CALR variants (Blood 2014;124:2465; Leukemia 2013;27:1861) . VHR karyotype included monosomal, inv(3), i(17q) and -7/7q-. Survival was calculated from date of first referral for the Mayo cohort and date of diagnosis for the Florence cohort; patients receiving stem cell transplant were censored at the time of transplantation.
Results: Mayo Clinic cohort (n=465)
Presenting clinical and genetic features and clinical course
465 Mayo Clinic patients (median age 63 years; 64% males) served as the learning cohort; DIPSS-plus risk distribution at presentation was 32% high, 37% intermediate-2, 18% intermediate-1 and 13% low. Driver mutations included JAK2 in 54%, type 1/like CALR 23%, type 2/like CALR 4%, MPL 7% and triple-negative 12%. ASXL1 and SRSF2 mutational frequencies were 38% and 15%, respectively. Ten patients displayed VHR karyotype. During median follow-up of 4.6 years, 330 (71%) deaths, 55 (12%) leukemic transformations, and 30 (6.5%) transplants were documented.
Survival analysis and development of the "revised DIPSS-plus"
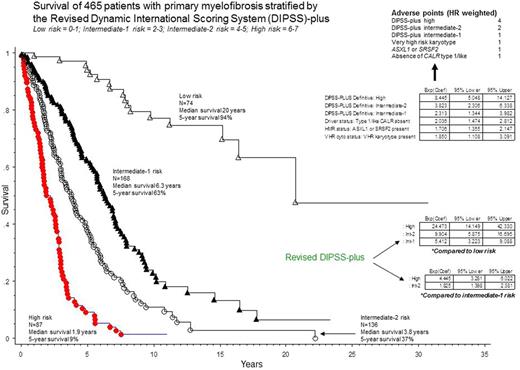
Multivariable analysis confirmed independent prognostic contribution from DIPSS-plus, type 1/like CALR, ASXL1 / SRSF2 mutations and VHR karyotype; HRs (95% CI) were 8.4 (5.1-14.1) for DIPSS-plus high, 3.8 (2.3-6.3) intermediate-2, 2.3 (1.3-4.0) intermediate-1, 2.0 (1.5-2.8) for absence of type 1/like CALR, 1.7 (1.4-2.1) for presence of ASXL1 or SRSF2 mutation, and 1.9 (1.1-3.1) for VHR karyotype.
HR-weighted scores were then assigned to absence of type 1/like CALR (1 point), presence of ASXL1 / SRSF2 mutation (1 point), presence of VHR karyotype (1 point) and DIPSS-plus intermediate-1 (1 point), intermediate-2 (2 points) and high (4 points) risk categories. Subsequent adverse point summation resulted in four new "revised DIPSS-plus (RDIPSS-plus)" risk categories: low (0-1 points; n=74; 16%); intermediate-1 (2-3 points; n=168; 36%), intermediate-2 (4-5 points; n=136; 29%) and high (6-7 points; n=87; 19%); the respective median survivals were 20, 6.3, 3.8 and 1.9 years (p<0.0001; Figure). RDIPSS-plus was also effective in predicting leukemic transformation; HRs (95% CI) were 19.3 (6.1-61.0), 5.8 (1.9-18.2) and 3.5 (1.2-10.6), for RDIPSS-plus high, intermediate-2, and intermediate-1 risk categories, respectively.
Risk group distributions in "RDIPSS-plus" vs "DIPSS-plus"
All 87 patients in the "RDIPSS-plus" high risk group belonged to DIPSS-plus high risk category. Among 136 intermediate-2 risk patients in the "RDIPSS-plus" model, 60 belonged to DIPSS-plus high and 76 DIPSS-plus intermediate-2; among 168 patients in the "RDIPSS-plus" intermediate-1 risk group, 98 belonged to DIPSS-plus intermediate-2, 60 intermediate-1 and 10 low risk; among 74 patients in the "RDIPSS-plus" low risk group, 25 belonged to DIPSS-plus intermediate-1 and 49 low risk groups.
Results: Florence cohort (n=347)
The aforementioned observations were validated in an external cohort of 347 patients from the University of Florence (details to follow at time of presentation).
Conclusions:
" RDIPSS-plus " incorporates clinical, cytogenetic and molecular information, in order to provide a comprehensive prognostic tool for facilitating treatment decision-making in PMF. RDIPSS-plus was most effective in extracting patients with better than expected outcome, from specific DIPSS-plus risk categories.
Vannucchi: Novartis: Honoraria, Speakers Bureau; Shire: Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.