Abstract
Introduction: CTL019 is an anti-CD19 genetically modified autologous T-cell immunotherapy developed at the University of Pennsylvania (Penn) that was recently approved for treatment of relapsed/refractory pediatric and young adult B-cell acute lymphoblastic leukemia (ALL) and adult relapsed/refractory diffuse large B-cell lymphoma (DLBCL) as tisagenlecleucel (Novartis). For ALL, the FDA-approved dose is 0.2 to 5.0 x 106 CAR-positive viable T cells per kg of body weight for patients ≤ 50 kg or 0.1 to 2.5 x 108 CAR-positive viable T cells for pts > 50 kg; for DLBCL, the FDA-approved dose is 0.6 to 6.0 x 108 CAR-positive viable T cells. For CTL019 manufactured at Penn, the dose is determined by flow cytometric staining of CAR-positive T cells, which are cryopreserved in product bags along with replicate aliquots of the final formulation in vials, simultaneously cryopreserved for release testing. The CTL019 product release criteria include a post thaw viability assessment using a vial of replicate aliquot of the final formulation for Trypan blue exclusion or dual fluorescence automated cell counting (Luna-FL, Logos Biosystems). There are no published data examining the relationship between CTL019 viability release testing and clinical outcomes.
Methods: We analyzed CTL019 post thaw viability release testing in patients treated on one prospective single institution clinical trial of CD19-expressing non-Hodgkin lymphomas (NHL) (NCT02030834) and two single-institution prospective pediatric ALL clinical trials (NCT01626495 and NCT02906371). Patients were assessed for response to therapy and CAR T-cell expansion. Receiver operating characteristic (ROC) curves were constructed for prediction of complete responses based on sensitivity and specificity of CAR T-cell product post thaw viability release test results.
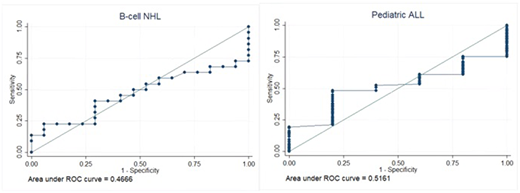
Results: 39 pts with relapsed/refractory NHL (24 diffuse large B-cell lymphoma and 15 follicular lymphoma) were enrolled and received the protocol-specified dose of CTL019. Best response rate was 56% (22/39) complete responses (CR). 123 pts with relapsed/refractory pediatric ALL were enrolled and received the protocol-specified dose of CTL019. Best response rate was 96% (118/123) CR/complete remission with incomplete blood count recovery (CRi). For patients with NHL infused with CTL019, product % viability had a median of 89.8% viability (range: 73.7%-97.7%); product % viability quintiles were as follows: 20%-tile=81.7%, 40%-tile=88.3%, 60%-tile=91.1%, 80%-tile=94.8%). ROC area for NHL patients was 0.47 (95%CI: 0.28-0.65). For patients with ALL infused with CTL019, product % viability had a median of 89.3% viability (range: 56.0%-98.4%); product % viability quintiles were as follows: 20%-tile=82.3%, 40%-tile=87.5%, 60%-tile=90.9%, 80%-tile=94.4%). ROC area for ALL patients was 0.52 (95%CI: 0.32-0.71). For patients with NHL, progression-free survival (PFS) was not significantly influenced by product viability release test results by Cox proportional hazards (HR: 1.0, 95%CI: 0.94-1.09, p=0.7). For patients with NHL, peak CAR T-cell expansion was not significantly correlated with product viability release test results (r2=0.12, p=0.5). Data collection for Cox analysis to investigate the effect of release test viability on PFS and correlation of release test viability with peak CTL019 expansion in ALL is ongoing and will be presented.
Conclusions: Our data suggest that, within the ranges obtained in these trials, there is no clear dose-response relationship between CTL019 product viability release test results and clinical response rates in pediatric and young adult ALL or DLBCL.
Chong:Novartis: Consultancy. Levine:Cure Genetics: Consultancy; Brammer Bio: Consultancy; CRC Oncology: Consultancy; Incysus: Consultancy; Novartis: Consultancy, Patents & Royalties, Research Funding; Tmunity Therapeutics: Equity Ownership, Research Funding. Grupp:Novartis Pharmaceuticals Corporation: Consultancy, Research Funding; Adaptimmune: Consultancy; University of Pennsylvania: Patents & Royalties; Jazz Pharmaceuticals: Consultancy. Davis:Novartis Institutes for Biomedical Research, Inc.: Patents & Royalties. Siegel:Novartis: Research Funding. Maude:Novartis Pharmaceuticals Corporation: Consultancy, Membership on an entity's Board of Directors or advisory committees. Frey:Novartis: Consultancy; Servier Consultancy: Consultancy. Porter:Genentech: Other: Spouse employment; Novartis: Other: Advisory board, Patents & Royalties, Research Funding; Kite Pharma: Other: Advisory board. June:Immune Design: Membership on an entity's Board of Directors or advisory committees; Tmunity Therapeutics: Equity Ownership, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding; Celldex: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis Pharmaceutical Corporation: Patents & Royalties, Research Funding; Immune Design: Membership on an entity's Board of Directors or advisory committees; Tmunity Therapeutics: Equity Ownership, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding; Novartis Pharmaceutical Corporation: Patents & Royalties, Research Funding. Schuster:Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Dava Oncology: Consultancy, Honoraria; Merck: Consultancy, Honoraria, Research Funding; Novartis Pharmaceuticals Corporation: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Nordic Nanovector: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Genentech: Honoraria, Research Funding; Gilead: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.