Abstract
PV and AL contributed equally
Multiple studies from the EBMT registry and others have shown excellent survival rates after allogeneic haematopoietic stem cell transplantation (HSCT)for Wiskott-Aldrich syndrome (WAS) patients (Ozsahin et al, Blood 2008). The importance of myeloid engraftment for full disease correction has also been demonstrated (Moratto et al, Blood 2011). However, the vast majority of HSCTs in these studies were performed with (oral) busulfan/cyclophosphamide-based conditioning and in the early 2000 years or before. In 2005, the inborn errors working party (IEWP) of EBMT and ESID first recommended busulfan/fludarabine (BuFlu) or treosulfan/fludarabine (TreoFlu) based conditioning for primary immunodeficiencies such as WAS, with some centers deciding to add thiotepa (TT) to the conditioning.
We performed a retrospective analysis via the EBMT and SCETIDE registries of WAS patients transplanted between 01/01/20006 and 12/31/2016 with these two regimens. The primary objective was to compare the overall (OS) and event-free survival (EFS) after HSCT with either BuFlu±TT or TreoFlu±TT conditioning. Secondary objectives included the influence of either conditioning regimen on acute and chronic GVHD, the degree of donor chimerism, incidence of secondary procedures after HSCT (2nd HSCT, stem cell boost, DLI, gene therapy or splenectomy) and rates of disease-specific complications after HSCT.
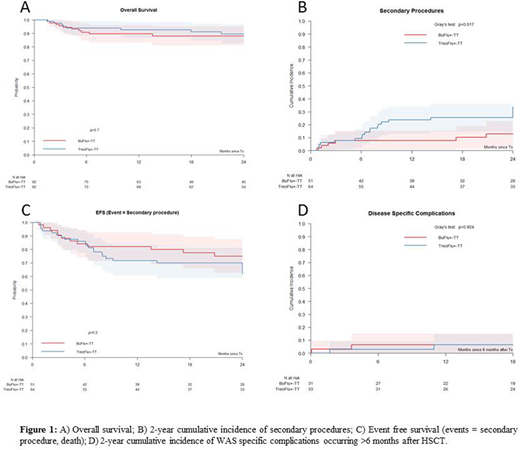
At the time of this interim analysis, 174 patients were included, 92 (53%) with BuFlu±TT and 82 (47%) with TreoFlu±TT conditioning with a median age of 1.57 years (range 0.21-29.96) at HSCT and a median follow-up of 32.9 months (range 1.5-128.9). The donor was an HLA-matched sibling (MSD) in 30, a matched related donor (MRD) in 5, a matched unrelated donor (MUD, 9/10 or 10/10) in 105, a mismatched unrelated donor (MMUD, <9/10) in 9 and a mismatched family donor (MMFD) in 25 (18 with ex-vivo T-cell depletion). Stem cell source was bone marrow in 93 (53%), peripheral blood in 62 (36%) and cord blood in 18 (10%). Two year overall survival (OS) of the entire cohort was 88.6% (95% confidence interval 83.5%-93.6%). There was no significant difference in OS between patients treated with BuFlu±TT or TreoFlu±TT conditioning (2-year OS 88.1% vs. 89.5%; log-rank test p=0.7). Patients aged >5 years had a worse OS as compared to those 5 years or younger at HSCT (74.9% vs. 90.8%; log-rank test p=0.005). The type of donor had no influence on OS: 96.4% for MSD/MFD, 86.8% for MUD/MMUD and 87.7% for MMFD (log-rank test p=0.4). Whole blood chimerism was complete (>90% donor) in 60/75 evaluable patients (80%) at last follow-up or before secondary procedure (if a patient had one), 39/40 (98%) in the BuFlu±TT group and 21/35 (60%) in the TreoFlu±TT group. Twenty-six patients required a secondary procedure: stem cell boost in 4 patients, donor lymphocyte infusion in 9, 2nd HSCT in 15 and splenectomy in 1. Twenty-two of these 26 (84.6%) are alive and 14 of 16 with available chimerism data have a complete donor chimerism (>90%donor) at last follow-up. The 2-year cumulative incidence (CI) of secondary procedures was higher at 33.9% in the TreoFlu±TT versus 12.8% in the BuFlu±TT group (Gray's test p=0.017), and 2-year EFS (secondary procedure or death as event) was 61.4% in the TreoFlu±TT and 75.0% in the BuFlu±TT group (log-rank test p=0.2). Grade II-IV acute GVHD had the same incidence in both groups (100 day CI 24.4% vs. 26.3%; Gray's test p=0.849) and chronic GVHD of any grade was borderline more frequent in the TreoFlu±TT group (2 year CI 17.2% vs 6.7%; Gray's test p=0.054). The cumulative incidence of disease-specific complications occurring more than 6 months post HSCT (severe infections, bleeding, autoimmunity) was not different between the two groups (6.5% vs. 6.4%; Gray's test p=0.92). There was no malignancy reported after HSCT except for one EBV-post-transplant lymphoproliferative disorder (PTLD) 2.7 months after HSCT.
In summary, HSCT with either BuFlu±TT or TreoFlu±TT conditioning reliably cures almost 90% of patients with WAS regardless of donor type. WAS-related complications are very rare events more than 6 months post HSCT. More patients required secondary procedures after treosulfan-based than busulfan-based conditioning. These data confirm the feasibility and efficacy of the regimens currently recommended by the IEWP.
Slatter:Medac: Other: Travel assistance. Chiesa:Gilead: Consultancy; Bluebird Bio: Consultancy. Kalwak:Sanofi: Other: travel grants; medac: Other: travel grants. Locatelli:Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; bluebird bio: Consultancy; Bellicum: Consultancy, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Miltenyi: Honoraria. Sykora:Aventis-Behring: Research Funding; medac: Research Funding. Zecca:Chimerix: Honoraria. Veys:Pfizer: Honoraria; Servier: Research Funding; Novartis: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.