Abstract
Introduction: In an Oncology Care Model (OCM) setting, practices may earn a Performance-Based Payment (PBP) for a reduction in the costs of treating participating Medicare patients during a 6-month episode of care. An Excel-based decision analytics model was developed to evaluate the cost-savings associated with implementing changes in the usage of tyrosine kinase inhibitors (TKIs) among patients with chronic myeloid leukemia (CML) within a typical OCM practice and the impact it could have on a practice potentially receiving a PBP.
Methods: The default scenario is based on an OCM practice that treats 1,000 cancer patients during a 6-month episode of care. The types of cancers treated and the proportions of patients treated in the OCM practice were estimated from an OCM baseline report; all-cause healthcare costs for each cancer type were obtained from published literature. CML patients were stratified into newly-diagnosed and established TKI-treated patients. The percentages of CML patients on each of the TKIs (branded and generic imatinib [1st-gen TKIs], as well as dasatinib and nilotinib [2nd-gen TKIs]) within each stratum were estimated using market share data from April 2018. The 2018 Wholesales Acquisition Costs for the TKIs were obtained from RedBook. It was assumed that, if a practice implements the policy of restricting utilization of branded TKIs as a cost-cutting measure, 80% of the current market share of branded imatinib would shift to the generic and 50% of the current market shares of 2nd-gen TKIs would shift to generic imatinib. Among established TKI-treated patients, it was assumed that 80% of the current market share of branded imatinib would shift to the generic, whereas no patients treated with 2nd-gen TKIs would be switched to generic imatinib due to the lack of supporting evidence, physician and patient apprehension, some patients already having used imatinib, among other reasons. The relationship between the savings achieved from restricting utilization of 2nd-gen TKIs and the savings required for the OCM practice to receive a PBP using either a one-sided or two-sided risk model was evaluated.
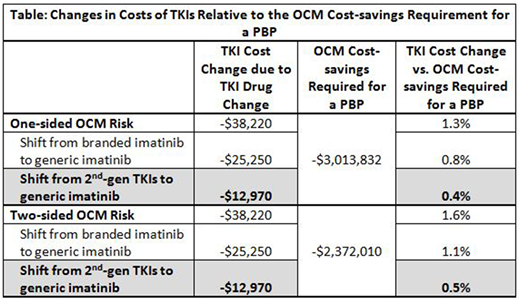
Results: The total healthcare costs of an OCM practice that treats 1,000 cancer patients for 6 months were estimated at $51,345,812. It was estimated that there would only be 4 CML patients in a 1,000-patient OCM practice, 1 newly-diagnosed and 3 established TKI-treated patients. Implementing the policy of restricting utilization of 2nd-gen TKIs for patients with CML would save a practice $12,970 during the 6-month episode of care, while $25,250 would be saved through a branded to generic imatinib shift (Table). For a 1,000-patient OCM practice participating in a one-sided risk model, a total cost-savings of $3,013,832 is required for it to be eligible for a PBP. In this scenario, the cost reduction associated with a shift from 2nd-gen TKIs to generic imatinib amounts to only 0.4% of the required total cost-savings threshold before the practice is eligible for a PBP. For a 1,000-patient OCM practice participating in a two-sided risk model, a total cost-savings of $2,372,010 is required for it to be eligible for a PBP. In this case, the cost reduction associated with a shift from 2nd-gen TKIs to generic imatinib amounts to only 0.5% of the required total cost-savings threshold before the practice is eligible for a PBP.
Conclusions: This economic model indicates that the cost-savings associated with restricting branded TKI utilization among CML patients in an OCM setting will represent only a very small portion of the cost-savings required before an OCM practice is eligible for a PBP. Of the reduction in TKI costs, approximately two-thirds was attributed to the shift from branded to generic imatinib. Restricting utilization of the 2nd-gen TKIs contributed a negligible amount of savings required for a PBP. The cost-savings opportunities in CML in the OCM setting are limited by how few CML patients would be affected by restrictions.
Jabbour:Pfizer: Consultancy, Research Funding; Novartis: Research Funding; Takeda: Consultancy, Research Funding; Bristol-Myers Squibb: Consultancy, Research Funding; Abbvie: Research Funding. Mendiola:Bristol-Myers Squibb: Employment. Lingohr-Smith:Novosys Health: Employment. Menges:Novosys Health: Employment. Lin:Bristol-Myers Squibb: Consultancy; Novosys Health: Employment. Makenbaeva:Bristol-Myers Squibb: Employment.
Author notes
Asterisk with author names denotes non-ASH members.