Abstract
Background and Aim:It is increasingly recognized that patients with a de novomyelodysplastic syndrome (MDS) onset as young adults, lacking any other feature of a congenital disorder, may share a pathogenic overlap due to the presence of both germline and somatic variants. Identifying an inherited pathogenic variant has important therapeutic implications beyond family counselling: adapting the selection of sibling donor, the use of highly cytotoxic therapy and the monitoring for other cancer development. However, most studies have focused on patients with suspected inherited disorders based on the presence of physical abnormalities and/or family history. In addition, a mixture of pediatric and adult cases is usually reported. The aim of this study is to characterize the germline and tumor variants in a group of adult MDS patients without accompanying congenital physical anomalies and or family antecedent of bone marrow failure.
Methods: We included 72 patients from 15 Spanish centers with a diagnosis of MDS between 18 and 60 years old (y.o). Patients with a previously diagnosed or suspected (one physical anomaly or family history) congenital syndrome were excluded. Diagnoses were made in accordance with the WHO 2016 classification. Whole-exome sequencing (WES) libraries were prepared using SureSelectXT Target Enrichment and sequenced on a HiSeq4000 platform (IlluminaInc.). Mean number of reads per sample was 138,726,017 with a Phred Quality Score >30 in 95.05% of bases. Read mapping sequence alignment and variant calling were performed using Biomedical Workbench (Qiagen). WES was performed on 72 tumor and 32 paired germinal DNA (buccal swab). To identify potential germline-causal mutations, a selection tool was implemented incorporating 239 genes associated with cause or predisposition to bone marrow failure or cancer. Variants with an ExAC, TOPMed and/or European 1000 Genomes minor allele frequency ≥0.01 were discarded.
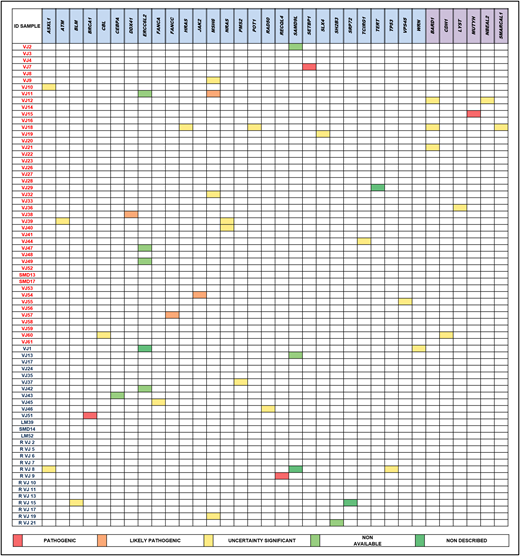
Results: The median age at diagnosis was 49 y.o. The cohort was categorised into two groups, less or equal 50 y.o. (62.5%) and between 50 and 60 y.o. (37.5%). In the first group, the frequency according to the WHO classification were 12% MDS with single lineage dyplasia (MDS-SLD), 8% MDS with ring sideroblasts (MDS-RS), 11% MDS with multilineage dyplasia (MDS-MLD), 24% MDS with excess blasts(MD-EB), 4% MDS with isolated del(5q)(MDS-del5q), 4% MDS unclassifiable and 4% chronic myelomonocytic leukemia (CMML). Meanwhile, in the group with age more than 50 y.o., the subtypes were 3.7% MDS-SLD, 7.4% MDS-RS, 29.6% MDS-MLD, 40.7% MD-EB, 3.7% MDS-del5q, and 14.8% CMML.Patients less or equal 50 y.o. were stratified based on IPSS-R as very low (4%), low (64%), intermediate (20%), high (12%) and very high (0%); and the group of more than 50 y.o. as very low (14.8%), low (33.3%), intermediate (29.6%), high (11.1%) and very high (11.1%).The mean number of somatic mutations was 0.68 in patients with less or equal 50 y.o. and 1.37 in those between 50 and 60 y.o., p=0.033 (U Mann-Whitney); and regarding germline variants, the first group mean number was 2.44 (p25-75, 1-3) and the second group showed a mean of 1.85 (QI 25-75, 1-3), p= 0,331.In the whole cohort, germline variants were found in 62 out of 72 patients, with the following frequencies: ATR(N=5, 6.9%), followed by BARD1(N=5, 6.9%), ERCC6L2(N=4, 5.6%), MSH6(N=4, 5.6%), TCIRG1(N=4, 5.6%), NBEAL2(N=4, 5.6%), ASXL1(N=3, 4.2%), ATM(N=3, 4.2%), MPL(N=3, 4.2%), NF1(N=3, 4.2%), RECQL4(N=3, 4.2%), SAMD9L(N=3, 4.2%), WRN(N=3, 4.2%).Among germline variants, those reported previously as pathogenic or likely pathogenic, or involving genes associated with familial MDS/AML included: ERCC6L2(N=4, 5.6%), SAMD9L(N=3, 4.2%), and one case mutated for DDX41, FANCC, JAK2, MSH6, SETBP1, MUTYH, BRCA1and RECQL4. In the whole cohort, somatic variants were found in 38 patients, with the following frequencies: TP53(N=7, 9.7%), ASXL1(N=7, 9.7%), SETBP1(N=5, 6.9%), NF1(N=5, 6.9%), SRSF2(N=4, 5.5%).
Conclusion:In this subset of young adults with de novo MDS without congenital anomalies and/or familial history suggesting the presence of an undiagnosed congenital syndrome, 18% of the cohort harbored a likely causative germline variant. In addition, we noted a predominance of variants affecting genes with a cancer predisposition limited to the hematopoietic system, rather than classical telomere, DNA damage genes with an established mendelian link.
Díez-Campelo:Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.