Background: Cardiac abnormalities in sickle cell anemia (SCA) include pan-chamber dilation, biventricular hypertrophy, ventricular diastolic dysfunction and elevated pulmonary artery pressures. Diastolic dysfunction in SCA has been reported with a variable prevalence, ranging from 11-77%. Its onset is in childhood, and data show an increase in prevalence with age [Pediatr Blood Cancer 2017, 64 (11): e26607]. While its etiology has remained elusive, it has been shown to be associated with early mortality in adults [J Am Coll Cardiol 2007, 49(4): 472-479] and decreased exercise capacity in children and adults [Circulation 2011, 124 (13):1452-1460]. The effect of hydroxyurea (HU), a SCA disease-modifying therapy, on ventricular diastolic dysfunction has not been investigated, and could represent a potential therapeutic strategy.
Objective: To determine the longitudinal effect of HU therapy on left ventricular diastolic function and to identify hematological correlates of altered left ventricular diastolic indices.
Methods: Children aged 5-18 years with HbSS or HbS/β0 thalassemia enrolled in the Long-Term Effects of Erythrocyte Lysis Trial [Pediatr Blood Cancer 2019, 66(7): e27717.], underwent prospective measurement of diastolic function by 2D-echocardiogram. Echocardiogram was obtained at steady state (≥ 4 weeks from illness, transfusion, or hospitalization), and then repeated 2 years later. Echocardiogram parameters used for clinical evaluation of diastolic function included early diastolic mitral annular velocity (lateral e'), an index of ventricular relaxation and an early marker of abnormal diastolic function, and the ratio between early mitral inflow velocity and early diastolic mitral annular velocity (lateral E/e'). Diastolic function was categorized as normal/abnormal, using normative age‐specific ranges [J Am Soc Echocardiogr 2004,17(3):212-221] and compared between baseline and 2 years. Hemoglobin and hemolysis markers including absolute reticulocyte count (ARC), and lactic dehydrogenase (LDH) were obtained concurrently.
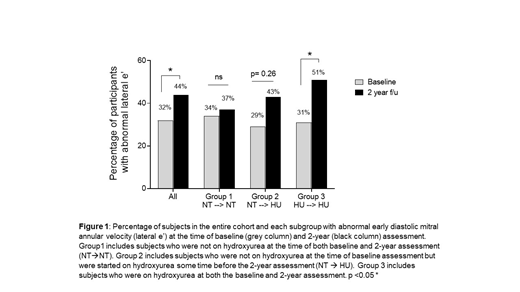
Results: 155 children with SCA, mean age 10.3 years (SD 3.54), were followed longitudinally. Of those, 59 (38%) did not receive HU, 61 (39%) were treated with HU both at baseline and at the 2-year evaluation, and 35 (23%) were untreated at baseline but initiated HU during the 2-year follow-up period after the initial echocardiogram. The prevalence of abnormal E/e' ratio did not change over time in the entire cohort (baseline 56% and 2-year 61%, p=0.23) or across the subgroups. However, the proportion of subjects with abnormal e' increased from 32% at baseline to 44% 2 years-later (p=0.01, Figure 1). In the subgroup analysis, participants who were continuously treated with HU were older [mean age 12 years (SD 3.54)] and had an increase in prevalence of abnormal e' at the 2-year time-point from 31% to 51% (p= 0.01, Figure 1). Individuals who were recently initiated on HU (baseline 29% and 2-year 43%, p = 0.27) or untreated (baseline 34% and 2-year 37%, p = 0.79) were comparatively younger [mean age 9.2 years (SD 3.02)] and did not have any significant change in diastolic function over time (Figure 1). A multivariate linear generalized mixed model was used to determine the association between abnormal diastolic and hematological parameters. An increase in hemoglobin concentration or a decrease in ARC and LDH did not affect the probability of having an abnormal e'. However, improvement in anemia among participants recently initiated on HU by 1 gm/dL, appeared to decrease the probability of having abnormal E/e' (Odds ratio, OR: 0.57, 95% CI: 0.32-1.03, p = 0.06). No such association was seen among participants on continuous HU therapy, whose hemoglobin did not significantly increase over the 2-year period from their baseline assessment.
Conclusion: Among children with SCA, HU therapy does not appear to significantly alter left ventricular diastolic indices over a follow-up of 2 years. Our findings may indicate HU's inability to affect cardiac remodeling in older children despite stable hematological parameters. Longer follow up is required to investigate clinically significant changes in diastolic indices during HU therapy. Additional therapeutic modalities that mitigate ventricular remodeling may need to be explored, to prevent the progression of diastolic dysfunction into adulthood, where it has been identified as a predictor of early mortality.
Kang:MBIO: Other: St. Jude Children's Research Hospital has an existing exclusive license and ongoing partnership with Mustang Bio for the further clinical development and commercialization of this XSCID gene therapy. Zhao:MBIO: Other: St. Jude Children's Research Hospital has an existing exclusive license and ongoing partnership with Mustang Bio for the further clinical development and commercialization of this XSCID gene therapy. Ataga:Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Global Blood Therapeutics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Emmaus Life Sciences: Honoraria, Membership on an entity's Board of Directors or advisory committees; Bioverativ: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer: Research Funding; Modus Therapeutics: Honoraria. Hankins:Bluebird Bio: Consultancy; NHLBI: Research Funding; ASPHO: Honoraria; Novartis: Research Funding; LYNKS Foundation: Research Funding; NHLBI: Honoraria; Global Blood Therapeutics: Research Funding; National Committee for Quality Assurance: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.