Background: Determining which AML patients (pts) are "fit" for intensive chemotherapy (IC), both in real-world scenarios and for clinical trial eligibility (particularly as it relates to regulatory indications) is challenging. While age, AML etiology, and cardiac function have been invoked, these oversimplify the clinical assessment and recommendation for IC vs. Non-IC (NIC). We compared baseline characteristics among pts receiving IC and NIC, and evaluated associations between treatment receipt and pt characteristics. Adjusting for treatment received, pts' attributes were further evaluated for association with overall survival (OS).
Methods: Adult (≥18 years) AML pts who received IC or NIC at Cleveland Clinic from 2003 to 2019 were included. The following characteristics were analyzed: age, sex, self-reported race, insurance, etiology of AML, comorbidities, hepatic and renal function tests, left ventricular ejection fraction (LVEF), and corrected QT interval (QTc). AML risk was categorized according to the 2017 European LeukemiaNet (ELN) risk stratification. Fisher's exact and Welch's t-tests were performed to compare baseline characteristics. Multivariate logistic and Cox regression were used to identify significant prognostic factors for treatment receipt and OS, respectively. The Kaplan-Meier method estimated distributions of OS, which were compared among pt cohorts using the log-rank test. Classification and regression tree (CART) analysis was performed to characterize the best decision process used to select pts for NIC.
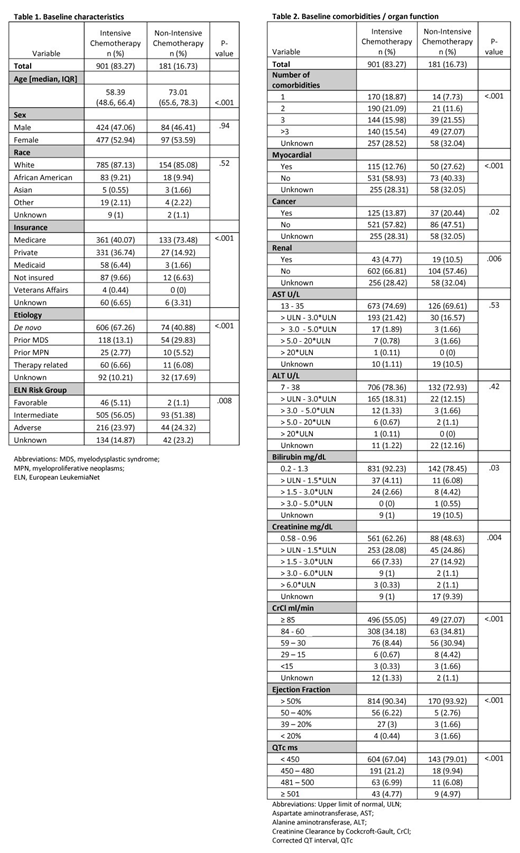
Results: Of 1,082 AML pts analyzed, 901 (83.2%) were treated with intensive, cytarabine-based chemotherapy, and 181 (16.8%) with non-intensive (low-dose cytarabine or hypomethylating agents) regimens. As expected, baseline characteristics were significantly imbalanced between groups; pts receiving NIC were more likely to be older (P<.001), have ≥3 comorbidities (P<.001), another cancer (P=.02), myocardial (P<.001), renal (P=.006) conditions, abnormal renal (creatinine (P=.004), creatinine clearance by Cockcroft-Gault (CrCl, P<.001)), and abnormal liver function (bilirubin (P=.03)). And less likely to have de novo disease (P<.001), favorable ELN risk (P=.008), private insurance (P<.001), and mild cardiac abnormalities (QTc 450-480ms (P<.001), LVEF 50-40% (P<.001)) (Tables 1 & 2).
Multivariable stepwise regression revealed independent associations with the following attributes (favoring NIC over IC): older age (OR=1.10, P<.001), etiology (prior MDS vs. de novo; OR=2.52 , P<.001), liver (bilirubin >1.5xULN vs. normal; OR=3.04, P=.02), renal (CrCl <60 ml/min vs. normal; OR=3.17, P=.002), myocardial conditions (atrial fibrillation/congestive heart failure/coronary artery disease vs. normal); OR=2.33, P=.001) and mild cardiac abnormalities (QTc 450-480ms vs. normal: OR=0.46, P=.01; LVEF 50-40% vs. normal: OR=0.33, P=.03). Clinically, in making decisions about NIC, the first step is age, followed by comorbidity. CART analyses revealed that when a comorbidity is identified, age is then re-assessed; depending upon the type of comorbidity present, various age thresholds determined NIC vs. IC.
With a median follow-up of 12.9 (IQR: 4.67-43.13) months, the median OS for the cohort was 15.1 months (range 0.03-189.25). OS was significantly different between groups (median OS, NIC vs. IC: 4.9 vs. 20.4 months, P<.001). In multivariate analyses controlling for treatment (IC vs. NIC), age, sex, insurance, number of comorbidities, AML etiology, and ELN risk, having another cancer (HR= 0.97, P=.78), renal (HR=1.10, P=.56) or myocardial comorbidity (HR= 0.94, P=.60) were not significantly associated with OS. Liver (bilirubin >1.5xULN: HR=1.70, P=.01), renal (creatinine >1.5xULN: HR=1.55, P=.001; CrCl<60ml/min: HR=1.37, P=.01), and cardiac dysfunction (QTc >480-500ms: HR=1.36, P=.04; LVEF 50-40%: HR=1.38, P=.04) were, however, associated with worse survival.
Conclusions: Age, comorbidities, and organ dysfunction are associated with AML treatment determination. Pts who presented with cancer/renal/myocardial comorbidities and renal/liver dysfunction were more likely to receive NIC, despite the comorbid conditions not having an impact on OS. Further research needs to elucidate the extent to which NIC is the optimal treatment decision for pts exhibiting varying severities of cancer/renal/myocardial comorbidities.
Hobbs:SimulStat Inc.: Consultancy; Amgen: Research Funding. Mukherjee:McGraw Hill Hematology Oncology Board Review: Other: Editor; Partnership for Health Analytic Research, LLC (PHAR, LLC): Consultancy; Celgene Corporation: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Projects in Knowledge: Honoraria; Bristol-Myers Squibb: Speakers Bureau; Takeda: Membership on an entity's Board of Directors or advisory committees; Pfizer: Honoraria. Advani:Glycomimetics: Consultancy, Research Funding; Amgen: Research Funding; Kite Pharmaceuticals: Consultancy; Macrogenics: Research Funding; Abbvie: Research Funding; Pfizer: Honoraria, Research Funding. Gerds:Sierra Oncology: Research Funding; Celgene Corporation: Consultancy, Research Funding; Pfizer: Consultancy; Incyte: Consultancy, Research Funding; CTI Biopharma: Consultancy, Research Funding; Imago Biosciences: Research Funding; Roche: Research Funding. Nazha:Daiichi Sankyo: Consultancy; Tolero, Karyopharma: Honoraria; MEI: Other: Data monitoring Committee; Novartis: Speakers Bureau; Jazz Pharmacutical: Research Funding; Incyte: Speakers Bureau; Abbvie: Consultancy. Sekeres:Millenium: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Syros: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.