Background. Warm antibody autoimmune hemolytic anemia (wAIHA) is a rare and potentially serious disease in which autoantibodies bind to red blood cells (RBCs), leading to phagocytosis by Fc receptor-bearing macrophages via a spleen tyrosine kinase (SYK) dependent signaling pathway. Fostamatinib is a potent SYK inhibitor that is orally administered and was approved for the treatment of chronic ITP in April 2018 by the US FDA and has demonstrated activity in patients with wAIHA. A phase 2, open-label, multicenter study (NCT02612558) showed that 11 of 25 (44%) patients with wAIHA had markedly improved hemoglobin (Hgb) levels after treatment with fostamatinib. Adverse events (AEs) from the phase 2 studies in wAIHA were manageable and consistent with those in the fostamatinib safety database of >3500 patients across multiple disease states. Therefore, the phase 2 results support the conduct of a phase 3 study in wAIHA.
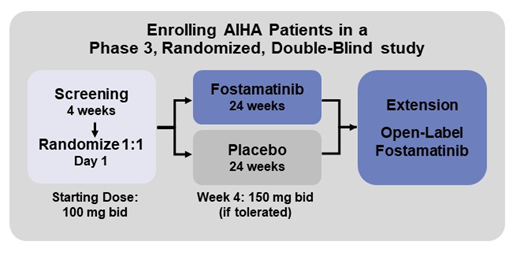
A phase 3, randomized, double-blind, placebo-controlled global study (NCT03764618) to investigate the safety and efficacy of fostamatinib in subjects with wAIHA is currently enrolling patients. The study intends to enroll approximately 80 patients at 109 sites in 19 countries across North America, Europe and Australia. This is the first randomized, double-blind, placebo-controlled study to explore the effect of a SYK inhibitor in the treatment of wAIHA.
Study Design and Methods
Inclusion Criteria: Eligible adult patients must have: primary or secondary wAIHA, documented by IgG or IgA positive direct antiglobulin test (DAT); failed ≥1 prior treatment for AIHA; haptoglobin <LLN (lower limit of normal) or total bilirubin >ULN (upper limit of normal) or lactate dehydrogenase (LDH) >ULN; and baseline hemoglobin level ≤9 g/dL or, if hemoglobin is >9 g/dL to <10 g/dL, subject must be on an allowed wAIHA treatment AND have symptoms related to anemia.
Exclusion Criteria: Patients should not have other types of AIHA, uncontrolled or poorly controlled hypertension, a neutrophil count <1,000/µL, platelet count <30,000/μL (unless due to Evans syndrome), or transaminase levels >1.5 x ULN.
Treatment: Patients will be randomized 1:1 to receive fostamatinib or placebo for 24 weeks. Patients will receive an initial dose of 100 mg BID which will be increased to 150 mg BID at Week 4, based on patient tolerability. In the event of dose-limiting AEs, the dose may be reduced at any time. Randomization will be stratified by concomitant steroid use at baseline and severity of anemia at screening. All patients will be allowed to continue concurrent steroid therapy and other selected wAIHA therapy (maximum of 2) throughout the 24-week study. A rescue medication protocol is allowed as needed. A steroid taper protocol allows reduced used of steroids in responsive patients. Patients who complete the phase 3 study will be encouraged to enroll in an open-label extension study to receive fostamatinib.
Endpoints: Efficacy endpoints will include hemoglobin response, defined as a hemoglobin level >10 g/dL with a ≥2 g/dL increase from baseline (Day 1) in the absence of rescue medication; duration of hemoglobin response; and use of wAIHA rescue therapy. Safety endpoints include the incidence of adverse events. Patients will be evaluated at clinic visits approximately every two weeks for safety assessments and measurement of hemoglobin levels.
Statistics: Based on results of the phase 2 study, a sample size of 80 subjects (randomized 1:1) would be required to provide 80% power to detect a difference in the response between the active and placebo groups using the Cochran-Mantel-Haenszel test at a two-sided significance level of 0.05. The response rate will be compared between groups using a chi-square test adjusting for randomization stratification factors.
Cooper:Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Rigel: Consultancy, Membership on an entity's Board of Directors or advisory committees; Principia: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Gorham:Rigel: Employment, Equity Ownership. Lowe:Rigel: Consultancy. Numerof:Rigel: Employment, Equity Ownership. Tong:Rigel: Employment, Equity Ownership. Kuter:Actelion (Syntimmune): Consultancy, Honoraria, Research Funding; Bristol Myers Squibb (BMS): Consultancy, Honoraria, Research Funding; Protalix: Consultancy, Honoraria; Rigel: Consultancy, Honoraria, Research Funding; Sanofi: Consultancy, Honoraria; Rigel: Consultancy, Honoraria, Research Funding; Genzyme: Consultancy, Honoraria; Dova: Consultancy, Honoraria; Agios: Consultancy, Honoraria, Research Funding; Agios: Consultancy, Honoraria, Research Funding; Alnylam: Consultancy, Honoraria, Research Funding; Argenx: Consultancy, Honoraria, Research Funding; Shinogi: Consultancy, Honoraria; Platelet Disorder Support Association: Consultancy, Honoraria; Principia: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Kyowa-Kirin: Consultancy, Honoraria; Amgen: Consultancy, Honoraria, Research Funding; Sanofi: Consultancy, Honoraria; Shinogi: Consultancy, Honoraria; Genzyme: Consultancy, Honoraria; UCB: Consultancy, Honoraria; Takeda (Bioverativ): Consultancy, Honoraria, Research Funding; Zafgen: Consultancy, Honoraria; Momenta: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Principia: Consultancy, Honoraria, Research Funding; Protalex: Consultancy, Honoraria, Research Funding; Shire: Consultancy, Honoraria; UCB: Consultancy, Honoraria; Takeda (Bioverativ): Consultancy, Honoraria, Research Funding; Up-to-Date: Consultancy, Honoraria, Patents & Royalties: 3 Up-to-Date chapters; Up-to-Date: Consultancy, Honoraria, Patents & Royalties: 3 Up-to-Date chapters; Platelet Disorder Support Association: Consultancy, Honoraria; Kezar: Research Funding; Pfizer: Consultancy, Honoraria; Merck Sharp Dohme: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Caremark: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Protalix: Consultancy, Honoraria; Dova: Consultancy, Honoraria; Merck Sharp Dohme: Consultancy, Honoraria; Kyowa-Kirin: Consultancy, Honoraria; Protalex: Consultancy, Honoraria, Research Funding; Momenta: Consultancy, Honoraria; Kezar: Research Funding; Pfizer: Consultancy, Honoraria; Actelion (Syntimmune): Consultancy, Honoraria, Research Funding; Alnylam: Consultancy, Honoraria, Research Funding; Argenx: Consultancy, Honoraria, Research Funding; Bristol Myers Squibb (BMS): Consultancy, Honoraria, Research Funding; Caremark: Consultancy, Honoraria; Zafgen: Consultancy, Honoraria; Shire: Consultancy, Honoraria.
Fostamatinib is a tyrosine kinase (SYK) inhibitor for the treatment of adult patients with chronic immune thrombocytopenia (ITP) who have had an insufficient response to a previous treatment. The use of fostamatinib in other diseases is off-label.
Author notes
Asterisk with author names denotes non-ASH members.