In this issue of Blood, Hayden et al elegantly describe real-world data from their positron emission tomography (PET)-adapted treatment approach toward consolidative radiotherapy (RT) after rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone (R-CHOP) in primary mediastinal B-cell lymphoma (PMBCL).1
PMBCL is a rare, aggressive B-cell lymphoma with a propensity to affect young females and commonly presents with a bulky mediastinal tumor compressing and/or invading adjacent thoracic structures. Initially considered a diffuse large B-cell lymphoma subtype, the World Health Organization criteria designated it a distinct disease entity in 2008.2 Because of its rarity and the lack of widely available molecular testing, a centralized multidisciplinary approach to the clinicopathological diagnosis is key. Management is currently based on data from retrospective cohorts, diffuse large B-cell lymphoma trial subgroup analyses, or small phase 2 studies.
Treatment with CHOP-based chemotherapy plus consolidative RT was adopted from aggressive non-Hodgkin lymphoma studies from the pre–rituximab era demonstrating improved results through delivery of RT to areas of initial bulk or a residual anatomical mass. The addition of rituximab has led to an excellent prognosis, with long-term survival rates of 85% to 95%.3,4 Although rare, relapses, when they occur, do so almost universally within 2 years of treatment and were historically associated with poor outcomes.
Because most patients now survive long term, and PET imaging provides confidence in confirming complete response, the value of routine RT after chemotherapy-induced remission is increasingly questioned. Some retrospective reports have suggested an association between RT omission and inferior survival, leading clinicians to be uncomfortable with this approach.3,5
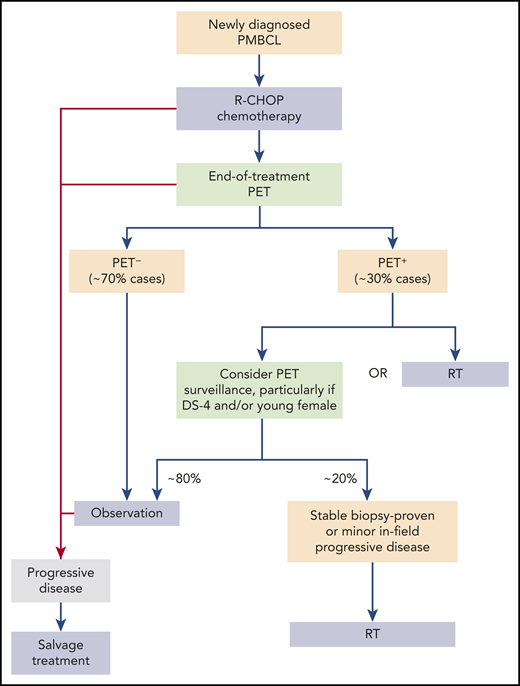
Adaptive therapy aims to minimize unnecessary toxicity for good-prognosis patients without compromising survival in poor-risk disease. Hayden et al report their institutional adaptive strategy in 109 PMBCL patients, employing end-of-treatment PET positivity after chemotherapy to determine the need for RT. A historic comparator group (n = 50) treated routinely with RT after R-CHOP was included. Although the usual caveats of retrospective studies apply, the authors should be praised for the central review of both histology and imaging in a majority of cases. Almost all received R-CHOP and PET-adapted RT as per prespecified institutional policy. The update in response assessment criteria during the study period was also addressed appropriately by applying blinded retrospective Deauville scores (DSs) to cases from before 2014. This PET-adapted strategy led to a 64% relative reduction in RT use and produced survival similar to that with routine RT and dose-intensive regimens. The reduction is likely to be further amplified with use of DSs in all cases, as evidenced by the small group downgraded from PET+ according to historical response assessment to PET− by DS. The 5-year overall survival (OS) rate for the entire cohort was 89%, and for the PET− group, OS was 97%; 1 of 71 patients in this PET group received RT. The 40% 5-year OS rate in relapsed/refractory patients is also noteworthy.
These results demonstrate achieving PET negativity after completing R-CHOP confers excellent survival, and RT can be safely omitted. Still, 30% to 35% of patients have residual PET+ masses irrespective of chemotherapy used.6,7 Management of this group remains less clear, although reported rates of both biopsy-proven disease and treatment failure are <20%, respectively, in PET+ patients.6,7
Assessment of prognostic factors in the Hayden et al study had limitations similar to those in previous analyses. However, it is important to observe that those with DS-4 disease who received RT experienced outcomes comparable8 to those of PET− patients, whereas DS-5 conferred a 5-year OS rate of only 64%. This warrants further investigation, particularly pertaining to the need for RT in DS-4 disease and prioritization of alternative strategies in DS-5 patients.
Dose intensification of chemotherapy has gained extensive support as an RT-sparing strategy. No randomized data support superiority of a particular regimen, with outcomes overall similar across studies, as summarized by Hayden et al in their supplement. The recent analysis from the German Lymphoma Alliance randomized phase 3 UNFOLDER study described virtually identical survival in those randomized to R-CHOP alone and those in the DA-EPOCH (dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin) studies (3-year OS, 96%), with no OS benefit provided by dose densification of R-CHOP or routine RT, in line with the prior UK study.4 The inherent problem with all of these analyses is that in the context of excellent outcomes, the small numbers are inadequate to detect meaningful differences between comparators. The landmark PMBCL phase 2 study of 51 patients treated with DA-EPOCH yielded a 5-year OS of 97%, with only 4% receiving RT,6 leading to widespread adoption. The study protocolized serial postchemotherapy PET imaging in patients with a residual PET+ mass until PET avidity normalized. This strategy, rather than regimen choice, is likely to have contributed to the low rate of RT. If employed after R-CHOP, the reduced rate of RT is likely to be replicated. The low positive predictive value of PET positivity by DS coupled with challenges faced with attempting biopsy has led others to propose a postchemotherapy serial imaging approach in PET+ disease as an alternative to routine RT; however, the risk of this strategy remains uncertain.7 Better methods of identifying disease through evaluation of PET metrics beyond DS, optimal timing of PET imaging, and integration of circulating tumor DNA monitoring are more likely to achieve successful individualized care.
Judicious use of RT in PMBCL is essential, given the long-term risks of irradiating large mediastinal fields in a predominantly young, female disease population. Advancement in RT techniques have almost certainly reduced risks of late organ dysfunction and second malignancies, although this is difficult to quantify. Additionally, the ability to serially monitor PET+ patients coupled with more meaningful options available in relapsed disease, such as checkpoint inhibitors, chimeric antigen receptor T cells, and allografts, allows clinicians more flexibility in the decision to irradiate. RT does remain an important, albeit diminishing, component of the treatment paradigm for PMBCL (see figure); therefore, RT omission must be carefully balanced with both excess toxicity from dose-intensive regimens and outcomes in relapse.
The IELSG37 study results are eagerly awaited (registered at www.clinicaltrials.gov as #NCT01599559). In the absence of these randomized data, the excellent outcomes from the Canadian PET-adapted approach favor omitting RT in PET− patients undergoing R-CHOP, which is further supported by the encouraging results from the PMBCL patients in the UNFOLDER study who received R-CHOP alone.
International research efforts should focus on maintaining these excellent outcomes while minimizing unnecessary toxicity by refinement of prognostication, incorporation of biomarker research, and prudent use of novel agents.
Conflict-of-interest disclosure: E.A.H. has received research funding from Bristol-Myers Squibb, Merck KgA, Astra Zeneca, Merck Sharp & Dohme, Celgene, and Roche; consulting/advisory compensation or honoraria from Roche, Janssen, Merck Sharp & Dohme, Gilead, and AstraZeneca; and travel expenses from Janssen and Roche.