Abstract
Background:
Selinexor is an oral, small molecule, selective inhibitor of nuclear export (SINE) compound that specifically blocks the karyopherin protein exportin 1 (XPO1, CRM1). In an shRNA library screen, we discovered that the survival of JAK2V167F mutant HEL cells is dependent on XPO1-mediated nuclear-cytoplasmic transport. Selinexor selectively suppressed primary myelofibrosis (MF) cells as compared with normal progenitor cells and induced hematologic responses in an MPN mouse model.
Methods:
An open label, prospective, investigator-initiated single center study is ongoing in adults with primary or secondary MF with resistance or intolerance to JAK inhibitor (JAKi) therapy with platelets > 30 K/μL and neutrophils > 500/μL. Selinexor was given orally once a week. Spleen volume was assessed by MRI at week 12 and week 24. The study was amended to include additional MRIs every 12 weeks in the year 1 and 24 weeks in the year 2. Primary end point is spleen response, defined as ≥ 35% spleen volume reduction (SVR) by MRI or CT, where applicable) at week 24. Bone marrow was evaluated at baseline and at week 24. The projected sample size of 24 will provide 83% power to reject a response rate of 15% and allow for up to a 25% dropout rate. We provide an interim report after completing 50% enrollment.
Results:
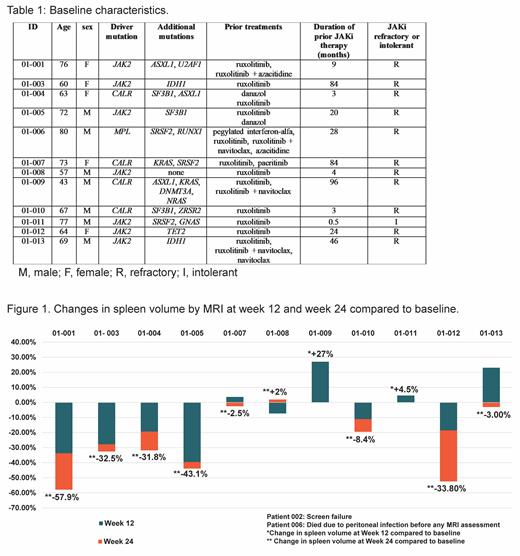
Between May 2019 and February 2021, 12 patients (pts) were enrolled. JAK2, CALR and MPL mutations were present in 7 (58.3%), 4 (33.3%) and 1 (8.3%) pts respectively. Eight pts (66.6%) had at least one high molecular risk mutation at baseline (Table 1). Median duration of prior JAKi therapy was 22 months (0.5 to 96 months) and 11 out of 12 were refractory to ruxolitinib at study enrollment. Median baseline spleen volume was 1454 cm 3(range 835 to 5792). Selinexor starting dose was 80 mg weekly in the first 6 pts and 60 mg for subsequent pts. At data cutoff, median duration of selinexor therapy was 36 weeks (range 11-114 weeks). One pt was not response evaluable and died due to liver abscess at week 12 (unrelated). One pt discontinued selinexor at week 18 due to grade 3 fatigue and was not evaluable for the primary end point. Of the 11 pts who had week 12 MRI or CT, 6 showed ≥ 10% SVR, 3 showed ≥ 25% SVR and 1 pt had early progression (Figure 1). At week 24, 5/9 (56%) pts had ≥ 25% SVR and 2/9 (22%) had ≥ 35% SVR (Figure 1). In 9 pts who had ≥24 weeks of selinexor, SVR ≥ 25% and ≥ 35% occurred at any point during study treatment in 4 (44%) and 3 (33%) pts, respectively. Two pts were red cell transfusion dependent at baseline; 1 became transfusion independent after 36 weeks of treatment, has not required transfusion for 49 weeks and remains on study treatment to date (114 weeks). Six pts (50%) discontinued selinexor. Reasons for treatment discontinuation are death in 1 pt, progressive disease in 1 pt, alternative treatment in 2 pts, and toxicity in 2 pts. Ten pts required dose reduction due to fatigue (1pt), anemia (1 pt), thrombocytopenia (2 pts), abdominal pain (1pt) and weight loss (5 pts). The most common treatment related adverse event was weight loss (grade 2 in 4 pts and grade 3 in 1 pt). This was manageable with treatment interruption and dose reduction, except in one pt who discontinued selinexor. As yet no changes in reticulin fibrosis MF grade were observed among 9 patients who received at least 24 weeks of treatment.
Conclusions:
Once weekly, oral selinexor showed single agent activity with sustained spleen responses in pts with JAKi refractory MF. Long-term administration of selinexor was well tolerated over time in MF pts. Correlatives studies including circulating inflammatory cytokine levels and mutant allele burden, as well as clonality studies by X-chromosome inactivation studies in woman, are underway and will be presented.
Tantravahi: BMS: Research Funding; Novartis: Research Funding; CTI BioPharma: Research Funding; Abbvie Inc.: Research Funding; Karyopharm Therapeutics Inc.: Consultancy, Honoraria, Research Funding. Patel: Stemline: Research Funding; Genentech: Research Funding; Roche: Research Funding. Chamoun: Karyopharm Therapeutics Inc.: Current Employment, Current equity holder in publicly-traded company. Shah: Karyopharm: Current Employment. George: Celgene: Consultancy; Bristol Meyers Squibb: Consultancy; Incyte Corporation: Consultancy; Blueprint Medicines: Consultancy. Deininger: Fusion Pharma, Medscape, DisperSol: Consultancy; SPARC, DisperSol, Leukemia & Lymphoma Society: Research Funding; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Part of a Study Management Committee, Research Funding; Blueprint Medicines Corporation: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Part of a Study Management Committee, Research Funding; Novartis: Consultancy, Research Funding; Sangamo: Consultancy, Membership on an entity's Board of Directors or advisory committees; Incyte: Consultancy, Honoraria, Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract