Abstract
Granulocytes for transfusion (GTX) continue to be administered, mostly to treat refractory infection 1. Single center series continue to be reported, but evidence of GTX effectiveness remains uncertain. Recent attempts at randomized trials of GTX have not been completed to target although the largest trial, RING 2, identified potentially promising results in a sub-group analysis by dose.
GTX can be produced by apheresis (from 'stimulated' donors), aGTX, or as a component pooled from whole blood (WB) donations. Logistic difficulties arise with aGTX, including recruitment of suitable donors willing to undergo cell mobilization and collection. In England GTX are now only supplied as pooled (10 WB donations/pool, mean volume single pooled unit 207 ml, mean granulocyte yield/unit 0.9 x10 10, typical adult dose 2 pools, available 6 days per week). In France, production moved from aGTX to pooled in 2020 (20 WB donations/pool, mean volume single pooled component 428 ml, mean granulocyte yield/unit 1.8 x10 10, typical adult dose 1 pool, available 6 days per week). There have been no recent published national data on the clinical use of pooled granulocytes, or comparisons between large datasets.
The aim of this study was to 1) describe the use of GTX, including pooled granulocytes, in two national cohorts of recipients; 2) estimate the effect of dose on patient mortality using statistical methods which could be applied to a larger dataset in the future.
A pre-piloted data collection form (DCF) was used to collect prospective audit data on all GTX given to patients in participating hospitals in England from March 2017 -Sept 2020 in England. The PROspective Granulocyte usage and outcomEs Survey (ProGrES) was deemed a national registry/audit with no individual patient consent required for anonymized data. Information on patient characteristics and outcomes was collected at the time of the request for GTX, following completion of GTX and at 28 days and 6 months follow up. DCFs were adapted for international use, and French data were collected from Jan 2018 to Dec 2020.
Descriptive analyses were performed to summarize GTX dose, patient characteristics, and outcomes. Using the English data, we investigated the association between GTX dose and mortality within 28 days of final GTX using logistic regression models with and without adjustment for possible confounders (age, infection source, use of renal therapy) and using the target trial framework. Ethical approval was obtained from London School of Hygiene and Tropical Medicine for analysis of English data.
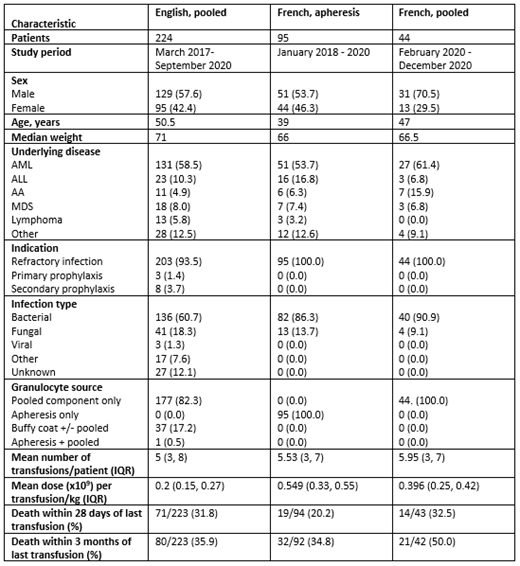
Results are presented on 224 (England, pooled) and 139 (France) patients who received GTX. In France, 95 and 44 patients received aGTX and pooled GTX, respectively. Table 1 shows patient features and outcomes. The most frequent underlying diagnosis was acute myeloid leukemia in all groups, and the most common indication was treatment of refractory infection. Median number of transfusions was similar in all groups (5.0, 5.3, 6.0). The mean dose/kg/transfusion was highest in the aGTX group (0.55 x10 9/kg), followed by French pooled (0.40) and English pooled (0.2). Death within 28 days of last GTX varied from 20.2% (French apheresis) to 32.5% (French pooled).
Additional analyses were performed on the English data; for death within 28 days, a 0.1 x10 9 increase in GTX dose was associated with a 22% reduction (95% CI -45% to +0%) in odds of death within 28 days of referral; odds ratio 0.78 (95% CI 0.57 to 1.00). After adjustment for potential confounders, the odds ratio was attenuated to 0.92 (95% CI 0.67, 1.23).
In summary, GTX continue to be requested by clinicians in patients with poor outcomes, despite an uncertain evidence-base. Regression analysis found dose was associated with lower odds of 28-day mortality. This was not significant after confounder adjustment, but this proof-of-principle analysis was limited by the sample size. A potential dose effect was also seen in the RING study. Our analysis using a target trial approach provides an approach for estimating GTX effectiveness in a larger cohort, which is required given the well-recognized challenges of undertaking randomized trials. The analysis provides a range of sample size calculations for testing an association between dose and outcomes for a defined effect size in a larger international cohort, supported by the BEST Collaborative 3.
References:
1. Transfusion 2019;59(1)160
2. Blood 2015;126(18)2153
3. Transfus Med Hemother 2018(45)318
Francis-Radice: GSK: Current equity holder in publicly-traded company; Smith & Nephew: Current equity holder in publicly-traded company; Bunzl: Current equity holder in publicly-traded company; Relx: Current equity holder in publicly-traded company; Cancer Research UK: Research Funding. McCullough: Fresenius Kabi: Honoraria; Terumo BCT: Honoraria.