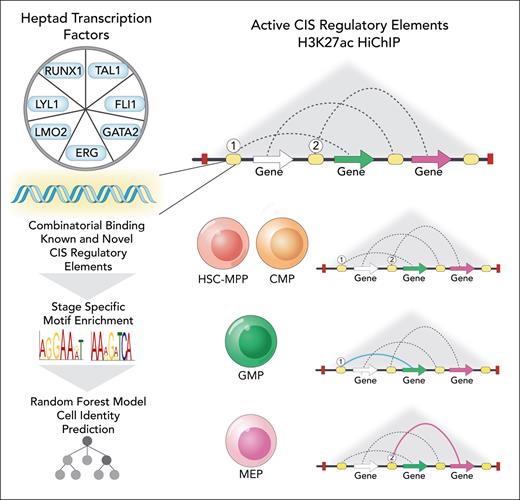
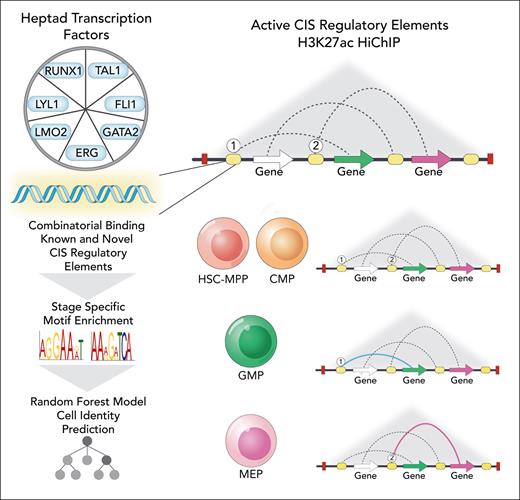
In this edition of Blood, Subramanian et al provide important new insights into the complex transcriptional regulatory networks governing human hematopoietic differentiation.1 Following recent work from this group that identified a triad of transcription factors (TFs) (GATA2-TAL1-ERG) that formed regulatory circuits controlling erythroid transition in bulk CD34+ cells,2 this study defined a “connectivity map” of selective TF binding sites for a predefined "heptad" factors (FLI1, ERG, GATA2, RUNX1, TAL1, LYL1, LMO2)3 as well as other chromatin marks in several populations of human hematopoietic stem and progenitor cells. This vast resource was analyzed for patterns of binding and chromatin interactions across these populations, providing insights into the dynamic remodeling of proposed regulatory circuits controlled by heptad TFs during early human hematopoiesis. Importantly, this analysis led to novel regulatory elements linked to key hematopoietic genes being proposed, including heptad genes themselves, with motif analysis of active noncoding cis-regulatory elements (CREs) demonstrating a “grammar” underlying cell type–specific putative enhancer activity, with a random forest-based machine learning approach able to link regulatory motifs to cell identity (see figure).
Analysis of “heptad” TF binding and chromatin interactions identifies a connectivity map of known and novel putative CREs in CD34+ mobilized HSC-MPP (hematopoietic stem cells and multipotential progenitor), CMP (common myeloid progenitor), GMP (granulocytic and macrophage progenitor), and MEP (megakaryocyte-erythroid progenitor) populations. 1 and 2 symbolically indicate heptad-bound CIS regulatory elements (CREs) associated with select lineage-specific gene expression in GMPs and MEPs, respectively. Professional illustration by Somersault18:24.
Analysis of “heptad” TF binding and chromatin interactions identifies a connectivity map of known and novel putative CREs in CD34+ mobilized HSC-MPP (hematopoietic stem cells and multipotential progenitor), CMP (common myeloid progenitor), GMP (granulocytic and macrophage progenitor), and MEP (megakaryocyte-erythroid progenitor) populations. 1 and 2 symbolically indicate heptad-bound CIS regulatory elements (CREs) associated with select lineage-specific gene expression in GMPs and MEPs, respectively. Professional illustration by Somersault18:24.
Historically, the importance of CREs had been demonstrated in early studies that identified the first lineage-specific enhancers in hematopoietic cells such as the β-globin locus control region governing erythroid gene expression.4 The discovery of several key hematopoietic TFs such as TAL1, GATA1, and PU.1, along with identification of target gene promoters and the enhancers that regulated their cell lineage–specific gene expression, allowed TF networks, including those regulating CREs, to be developed.
Interestingly, recent work has identified the importance of CREs in helping to characterize hematopoietic cell states. Using ATACseq and RNAseq, Corces et al identified chromatin accessibility was more adept than messenger RNA expression levels at classifying cell types and, intriguingly, distal elements enriched for CTCF binding, providing significantly improved cell-type classification in comparison with promoters.5
Although Subramanian et al identified that 3-dimensional genome structures were relatively stable across all progenitor populations, increasing complexity of putative distal active enhancer interactions was noted with early myeloid lineage differentiation. In this case, a subset of interactions at select lineage-specific gene loci were presaged by binding or cobinding of heptad factors to CREs at an earlier developmental stage. This led to an interesting proposition that binding of 1 or more heptad factors to a CRE may “prime,” in a noncanonical sense, distal CRE regulation of gene transcription of a subset of lineage-specific genes, providing a conceptual framework to perceive the relationship between initial TF binding to active CREs and distal chromatin looping to specific gene loci that occurs with lineage differentiation.
These observations permit several interesting questions to be posed. Which enhancer CREs are the most important in a given biological process, such as cell lineage determination? How can the proposed network models integrate with other known lineage-determining TFs? Of the heptad TFs associated CREs, how important are specific heptad factors at a given CRE in governing lineage determination in this early-stage myeloid transition, and how may this apply at a clonal level?
For some of these questions, orthogonal studies have provided several insights. For instance, analysis of γ-retroviral integration sites in patients undergoing gene therapy have identified CREs selected with hematopoietic stem cell (HSC) function through representational analysis in hematopoietic lineages following transplantation,6 a subset of which can be shown and predicted in the authors’ random forest model, to be enriched in HSC-MPP–specific CREs.
Importantly, analysis of PU.1 binding as related to the heptad TFs by the authors in this and previous studies3 suggests that PU.1 does not significantly affect enrichment of heptad TFs to binding regions, implying that PU.1, in its pioneering TF role and dose-dependent function for lymphoid/monocytic lineage determination, may act relatively independently of the heptad networks that the authors propose here. Similar questions can also be posed against this framework for other non–heptad lineage defining TFs such as CEBPA, MEIS1, and CBFB. For CTCF and cMYC that have defined roles in controlling hematopoietic stem cell quiescence and lineage transition,7 heptad network models would need to be developed in nonmobilized stem and progenitor cell populations.
Finally, with technological improvements in experimental techniques, single-cell and genome-editing approaches can help address the known heterogeneity underlying clonal hematopoiesis. For example, whereas the network models the authors propose here are built on population-averaged contact maps across bulk-cell populations, single-cell approaches have been applied in other model systems to identify the relative importance of individual factors in cell fate specification,8 and systematic perturbation of conserved elements using genome editing can provide a functional approach to identification and validation of novel CREs.9
As this and other studies have shown, expression of genes that determine cell fate and function rely on a series of gene-regulatory networks that define and tune target gene expression as well as important molecular machinery that regulate chromatin organization and interaction. As computational approaches continue to be developed using more sophisticated machine learning architectures to help understand these increasingly complex relationships,10 the fun, as they say, has only just begun.
Conflict-of-interest disclosure: The author declares no competing financial interests.