Acute myeloid leukemia (AML) is a hematopoietic cancer characterized by aberrant immature myeloid progenitor blasts in bone marrow and peripheral blood. Venetoclax (VEN), a BCL-2 inhibitor, is FDA-approved for AML treatment with hypomethylating agents (HMA). Despite the improvement of VEN and HMA, not all patients respond, and most patients develop resistance. This presents an urgent need for new therapies to overcome VEN resistance and enhance VEN efficacy. We propose SUMOylation inhibition as a novel therapy to address this need. SUMOylation regulates protein function by covalently attaching Small Ubiquitin-like MOdifier (SUMO) proteins to target proteins. SUMOylation plays an important role in tumor progression and immune response. Our study aims to evaluate the effects of SUMOylation inhibition on anti-AML activity of VEN and the mechanism.
We firstly generated VEN-resistant cell lines Molm13-VR and MV411-VR by culturing Molm13 and MV4-11 cells in increasing doses of VEN. SUMO E1 (SAE2) and global SUMOylation levels were remarkably upregulated in VEN-resistant cell lines, suggesting SUMOylation plays a role in VEN resistance. We evaluated the antileukemic effects of TAK-981 (subasumstat), a novel and specific SUMO E1 inhibitor, alone and in combination with VEN, using AML cell lines and primary blasts isolated from refractory/relapsed (R/R) AML patients. Different AML cell lines and blast samples showed various sensitivities to VEN (IC 50 10nM ~ 5,000nM), but significant synergism was observed with TAK-981 and VEN in cell viability assay. TAK-981 synergized with VEN at inducing AML cell apoptosis determined by annexin-V staining with flow cytometry as well as increased levels of cleaved-PARP and cleaved caspase-3 by western blotting.
The mechanism of action of VEN is dependent on BCL-2 mediated mitochondrial function and metabolism. We assessed mitochondrial membrane potential using TMRM probe. TAK-981 reduced mitochondrial membrane potential in both VEN-sensitive and resistant AML lines, further in combination with VEN. Because mitochondrial membrane potential is maintained by normal mitochondrial structure, we performed electron microscopy to determine mitochondrial structure. VEN had limited effect on mitochondria in resistant cells. However, TAK-981 induced abnormal ultrastructure mitochondria with fewer cristae, worsened by TAK-981+VEN combination.
Since TAK-981 treatment caused mitochondrial structure defects, we determine the mitochondrial function using Seahorse XF assay. VEN decreased oxygen consumption rate (OCR), indicating inhibition of oxidative phosphorylation (OXPHOS), further suppressed by TAK-981. Metabolomic profiling by mass spectrometry indicated TAK-981+VEN treated AML cells displayed striking decreases in TCA cycle intermediates and reduced electron transport chain (ETC) activity. Pathway analysis of the metabolomics data revealed significant enrichment in amino acids metabolism. Notably, RNA-sequencing and GSEA analysis showed consistent findings that TAK-981+VEN showed suppressed signaling pathways in mitochondrial function, glycolysis and amino-acid metabolisms, indicating TAK-981 targeted mitochondrial metabolism to enhance VEN activity.
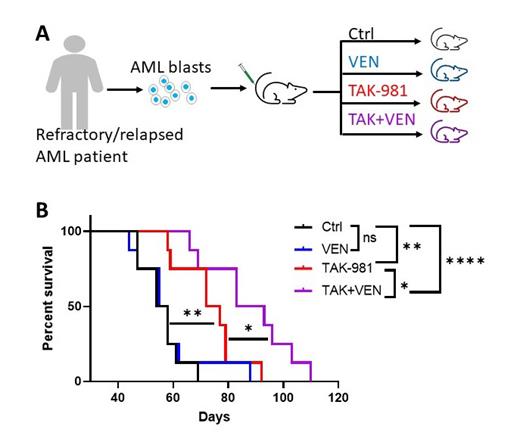
We then determined whether TAK-981 enhanced antileukemic effect of VEN ex vivo and in vivo. TAK-981 synergized with VEN at reducing colony formation capacity in colony formation assay using leukemia stem cells (LSCs) isolated from relapse AML blasts. More importantly, TAK-981 showed remarkable therapeutic effect in patient-derived xenograft (PDX) mouse model engrafted with blasts isolated from R/R AML patients, and significantly synergized with VEN at extending mice survival in vivo.
In conclusion, TAK-981 enhances the antileukemic activity of VEN by targeting mitochondria function and metabolism, offering a potent therapy for refractory/relapsed AML.
Disclosures
Marcucci:Ostentus Therapeutics: Current equity holder in private company, Research Funding. Rosen:Exicure: Current holder of stock options in a privately-held company; Pepromene Bio, Inc: Current holder of stock options in a privately-held company; Pepromene Bio, Inc: Membership on an entity's Board of Directors or advisory committees; NeoGenomics: Membership on an entity's Board of Directors or advisory committees; Verastem, Inc: Consultancy; Trillium Therapeutics, Inc: Consultancy; PharmaGene, LLC: Consultancy; Apobiologix/Apotex Inc: Consultancy; Exicure: Consultancy; Pheromone Bio, Inc: Consultancy; January Biotech: Current holder of stock options in a privately-held company; Trillium Therapeutics: Current holder of stock options in a privately-held company; SLAM BIOTHERAPEUTICS, INC: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees.