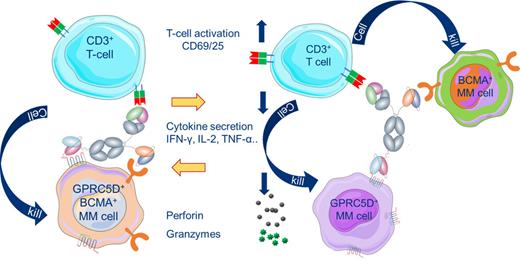
Background: Multiple myeloma (MM) is a genetically complex and heterogenous malignancy with a poor survival rate and accounts for approximately 10% to 15% of all hematologic cancers. Despite advances in myeloma therapy, MM remains incurable. Deep and durable clinical responses have been observed when targeting the tumor associated antigens BCMA and GPRC5D with immunotherapies. Additionally, data presented at ASCO this year demonstrated a further improvement in ORR when BCMA and GPRC5D bispecifics were given in combination to relapsed or refractory MM patients. These data suggest that broader antigen coverage by redirecting T cells to two tumor surface antigens is an effective means to harness the immune system to deplete cancer cells. Targeting two tumor antigens may also reduce the likelihood of antigen escape as a primary reason for relapse. Here, we describe JNJ-79635322, a trispecific antibody targeting B-cell maturation antigen (BCMA) and G-Protein-coupled receptor class 5 member D (GPRC5D), which are both highly expressed on plasmablasts and plasma cells in myeloma patient samples. Dual antigen recognition on plasma cells with a trispecific T-cell engaging antibody engineered with a CD3 arm has the potential to enhance tumor binding through avidity that could result in efficient depletion of malignant clonal populations and prevent tumor antigen loss mediated resistance.
Methods: JNJ-79635322 is a trispecific antibody composed of an anti-CD3 binding domain an anti-BCMA binding domain, and an anti-GPRC5D binding domain. It was selected based on biophysical assessment and showed activity in multiple myeloma models.
Results: JNJ-79635322 induced potent cytotoxicity in various myeloma cell lines in vitro with concomitant T-cell activation. Importantly, JNJ-79635322 was able to deplete both dual (BCMA and GPRC5D) and single (BCMA or GPRC5D) target expressing cells efficiently. In addition, dose dependent depletion of H929 myeloma cells was observed in a more physiological healthy fresh whole blood co-culture assay. In vivo, JNJ-79635322 exhibited potent antitumor activity compared to vehicle and antibody controls in a murine MM xenograft prevention model (single target expressing clonal H929 cells) and two tumor regression models (RPMI 8226 and MM.1S cells).
Conclusion: JNJ-79635322 is a potential first-in-class trispecific antibody targeting BCMA and GPRC5D with the ability to deplete dual and single target expressing MM clones in preclinical studies vitro and in vivo. A Phase 1 dose-escalating study of JNJ-79635322 in myeloma patients is ongoing (NCT05652335).
Disclosures
Pillarisetti:Janssen Pharamceuticals: Current Employment, Current equity holder in publicly-traded company, Current holder of stock options in a privately-held company. Yang:Janssen Pharmaceuticals: Ended employment in the past 24 months. Yao:Janssen Pharmaceuticals: Current Employment, Current equity holder in publicly-traded company. Smith:Janssen Pharmaceuticals: Current Employment, Current equity holder in publicly-traded company. Luistro:Janssen Pharmaceuticals: Current Employment, Current holder of stock options in a privately-held company. Vulfson:Janssen Pharmaceuticals: Current Employment, Current equity holder in publicly-traded company. Testa:Janssen Pharmaceuticals: Current Employment, Current equity holder in publicly-traded company. Packman:Janssen: Current Employment, Current equity holder in publicly-traded company. Brodeur:Janssen Pharmaceuticals: Ended employment in the past 24 months. Attar:Janssen Pharmaceuticals: Current Employment, Current equity holder in publicly-traded company. Elsayed:Janssen: Current Employment, Current equity holder in publicly-traded company. Philippar:Janssen: Current Employment, Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties.