Introduction
Chimeric antigen receptor (CAR)-T cell therapy is considered a therapeutic breakthrough in cancer immunotherapy with high and sustained remission rates. However, it can cause serious side effects such as cytokine release syndrome (CRS). CRS is characterized by systemic hyper-inflammation leading to hypotension, hypoxia, and progressive organ failure. Management of CRS commonly requires administration of the Interleukin-6 receptor antagonist (IL6-RA) tocilizumab. CRS-related coagulopathy is considered the result of disseminated intravascular coagulation (DIC) and concomitant progressive liver dysfunction. Overall, coagulopathy is observed in about every second patient after CAR-T cell therapy with a disproportional rate of hypofibrinogenemia. In this study, we investigated the incidence, dynamics, and possible causes of hypofibrinogenemia after CAR-T cell therapy.
Methods
We included all patients (n=41) with hematological malignancies receiving CAR-T cell therapies at the University Hospital Regensburg between 2020 and May 2023. All patients were followed up until death or 07/31/2023. Median age was 69 (range: 38-83) years, half of the patients were female and 73% had diffuse large cell B-cell lymphoma.
Results
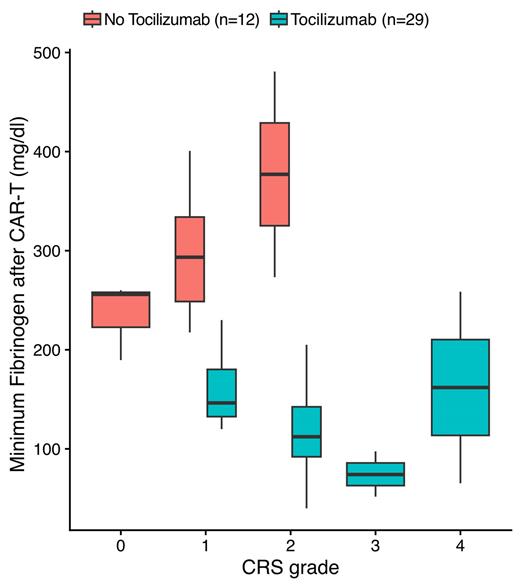
93% of patients developed CRS (12% ≥ grade 3), 71% received tocilizumab and 34% high-dose corticosteroids. While DIC and liver dysfunction almost exclusively occurred in CRS grade 3 or higher, 38% of patients with CRS grade 1 and 90% with CRS grade 2 developed de novo hypofibrinogenemia. Hypofibrinogenemia was severe or life-threatening (CTCEA ≥ grade 3) in 39% of cases and frequently required substitution. Median day of fibrinogen recovery (> lower limit of normal) was day 52 after CAR-T cell therapy. Patients with de novo hypofibrinogenemia initially displayed rising fibrinogen levels alongside development of CRS and rapidly declined in fibrinogen levels shortly after the first administration of tocilizumab. Minimum fibrinogen levels and duration of hypofibrinogenemia further correlated to tocilizumab dosing (r=-0.44 p=0.004 and r=0.7 p<0.001, respectively). Resolution of hypofibrinogenemia was associated to normalization of suppressed C-reactive protein (CRP) after tocilizumab treatment. In contrast, patients with CRS grades 1 and 2 who did not receive tocilizumab (7/13 and 2/20, respectively) had increased fibrinogen levels throughout the course and until resolution of CRS. Logistic regression showed that administration of tocilizumab was the most important, independent risk factor for development of hypofibrinogenemia (OR = 517, p<0.001).
Conclusions
In summary, hypofibrinogenemia is a frequent complication of CAR-T cell therapy and tocilizumab administration. We conclude that fibrinogen synthesis in CRS is upregulated in an IL-6-dependent acute phase reaction compensating for CRS-induced consumptive coagulopathy. IL-6RA tocilizumab inhibits fibrinogen upregulation and thereby causes protracted hypofibrinogenemia. These observations provide novel insights into the emergence and pathophysiology of hypofibrinogenemia after CAR-T cell therapy and emphasize the need for close fibrinogen monitoring after tocilizumab treatment of CRS.
Disclosures
Perl:Takeda: Honoraria; Kite/Gilead: Other: Travel grants; Amgen: Other: Travel grants. Hansmann:Amgen: Other: Travel expenses; Sanofi: Membership on an entity's Board of Directors or advisory committees. Herr:Amgen: Honoraria, Other: Travel expenses; Janssen-Cilag: Honoraria, Other: Travel expenses. Poeck:Gilead: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Pfizer: Consultancy; Novartis: Consultancy, Honoraria; Servier: Consultancy, Honoraria; BMS: Consultancy, Honoraria, Research Funding; Janssen-Cilag: Honoraria. Wolff:Gilead: Honoraria; Takeda: Honoraria; Behring: Honoraria; Novartis: Honoraria, Research Funding; Incyte: Honoraria; Sanofi: Honoraria; Mallickrodt: Honoraria. Fante:Amgen: Honoraria, Other: Travel expenses; Novartis: Honoraria, Other: Travel expenses.