Multiple myeloma (MM) is a plasma cell malignancy characterized by the extensive expansion of monoclonal aberrant plasma cells in the bone marrow and the second most prevalent hematological malignancies worldwide. Despite the promising outcomes of advanced therapies, relapsed/refractory (RR) diseases remain incurable, highlighting an unmet clinical need to identify novel therapeutic targets. An emerging field of investigation is the role of aberrant post-translational modifications in multiple myeloma pathology. Arginine methylation is one of the most predominant post-translational modifications in cells, catalyzed by protein arginine methyltransferases (PRMTs) that plays a crucial role in multiple cellular processes. Type I PRMTs catalyze mono- (MMA) and asymmetric demethylation (ADMA) of arginine; Type II PRMTs catalyze mono- and symmetric demethylation (SDMA) of arginine on proteins; Type III PRMT mediates MMA only. Aberrant expression of PRMTs is strongly associated with poor prognosis of solid cancers and hematologic malignancies.
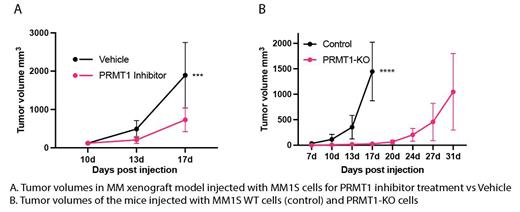
Recently, we identified PRMT1 as a novel therapeutic vulnerability in MM cells. Among PRMTs, PRMT1 is the highest expressed gene in MM cell lines and patient cells. The elevation of PRMT1 expression is strongly associated with relapsed/refractory patients and poor clinical outcomes. Deletion of PRMT1 and suppression of the PRMT1's enzymatic activity exhibited a strong anti-MM in vitro via suppressing cell cycle and elevating the apoptosis. We also found that inhibition of PRMT1 boosts the T cell activation in the co-culture setup, indicating a potential enhancement of immunotherapy. Using a subcutaneous xenograft model, we showed that suppressing PRTM1 in vivo by either genetic or pharmaceutical approaches robustly inhibits MM tumor growth and extends survival of tumor-bearing mice. Our findings demonstrated that PRMT1 is indispensable in MM.
PRMT5, a PRMT type II, is prognostically relevant and a known druggable target in MM. PRMT1 and PRMT5 inversely regulate post-transcription and have been implicated in human cancers. Recent studies in solid tumor showed a strong anti-tumor effect in vivo using the combinational inhibition of PRMT1 and PRMT5. We hypothesize that PRMT1 and PRMT5 might have a complementary function in MM cells. The combinational inhibition PRMT1 and PRTM5 will exhibit a synergistic anti-MM effect. To address this hypothesis, we treated MM cell lines with two clinical trial inhibitors: GSK3368715 (GSK) and EPZ 015666 (EPZ), suppressing PRMT type I and type II inhibitor, respectively.
We first performed dose-dependent cell viability inhibition and selected effective doses for drug combination treatments in three different MM cell lines. The combination of GSK and EPZ demonstrated a strong synergistic effect in suppressing the viability of MM cells. Using the SynergyFinder algorithm, we found that the synergistic indexes (>10, synergism) for 3 MM cell lines JJN3, MM1S, and KMS11 were 33.5, 32.8, and 14.3, respectively. We next assessed the effects of combined treatment on the cell cycle and apoptosis of MM cells. We found that combination of GSK and EPZ strikingly elevated the apoptosis and suppressed cell cycle of MM cell lines compared single treatment and untreated control. Intriguingly, the combination treatment of two inhibitors significantly decreased MMA, SDMA, and ADMA, suggesting a robust suppression of global arginine methylation. Of note, we found a similar anti-MM effect when treating bortezomib-resistant MM cells with GSK and EPZ, indicating that PRMT type I and type II inhibitors potentially have a substantial clinical impact on relapsed MM. Of note, the same dose of GSK and EPZ that exhibits the synergistic anti-MM effect, didn't suppress the viability of healthy peripheral blood cells.
In summary, our preliminary data suggest that suppression of both PRMT type I and type II might provide a potential effective combination treatment for both newly diagnosed and relapsed MM patients. We're currently investigating the underling mechanisms of how PRMTs regulate MM pathology and validating the in vivo efficacy of combination treatment using patient-derived xenograft.
Disclosures
Walker:Bristol Myers Squibb: Research Funding; Genentech: Research Funding; Abbvie: Speakers Bureau.