Abstract
The immune system of patients infected with human immunodeficiency virus (HIV) is in a state of chronic activation; however, the nature of HIV-related immune activation is unknown. As normal T-cell activation involves early tyrosine phosphorylation induced by the T-cell antigen receptor-associated src-family protein tyrosine kinase p59fyn(T) (Fyn), we examined a potential role for this kinase in HIV-related immune dysfunction. We determined the relative specific kinase activity of Fyn in lysates of peripheral blood mononuclear cells from 47 normal control individuals tested negative for HIV-1 and -2, human T-cell lymphotropic virus Type I, hepatitis B virus (HBV), hepatitis C virus (HCV), and syphilis; 14 asymptomatic HIV-infected patients having near-normal CD4+ T-cell counts (350 to 980 CD4+ cells/μL); 4 patients with symptomatic acquired immunodeficiency syndrome (AIDS) (<30 CD4+ cells/μL); 13 patients having chronic infection with HBV (6 patients) or HCV (7 patients); and 6 patients with systemic lupus erythematosis (SLE). All patients with asymptomatic HIV disease were shown to have a profound increase (mean increase of 19-fold; range threefold to 56-fold increase; p = 1.33 × 10−9) in the relative specific kinase activity of Fyn compared to uninfected controls or patients with hepatitis or SLE. In contrast, patients with AIDS had an Fyn-specific kinase activity that was much less affected (mean increase of threefold; range onefold to sevenfold increase; p = 1.30 × 10−5). It was further shown that HIV infection affects the Fyn-specific kinase activity in CD8+-enriched cells, suggesting abnormal Fyn activity in both CD8+ as well as CD4+ T lymphocytes. Initial results implicate a role for the CSK protein tyrosine kinase as responsible for the abnormal Fyn kinase activity observed in HIV-infected patients. These data indicate early and chronic activation of Fyn as a unique HIV-related effect that has the potential to be diagnostic for early HIV infection and/or may serve as a prognostic indicator for advancement to full-blown AIDS. More importantly, sustained activation of the protein tyrosine kinase associated with T-cell antigen receptor function may result in, or contribute to, the immunopathogenic effects associated with HIV infection.
THE IMMUNE SYSTEM of patients infected with the human immunodeficiency virus (HIV) is in a state of chronic activation.1 Patients present with elevated levels of markers of lymphocyte and macrophage activation including neopterin, soluble cytokine receptors, and β2 -microglobulin.1-5 The biochemical nature of HIV-related chronic immune system activation is unknown.
T-cell activation initiates a specific set of metabolic alterations including immediate (within seconds) increases in tyrosine phosphorylation of a number of proteins. Two members of the src-family of protein tyrosine kinases (PTK), p56lck (Lck) and p59fyn(T) (Fyn), are proximal signaling molecules responsible for initiating the cascade of biochemical signals resulting in T-cell activation.6-12 Previous work has suggested that the HIV envelope protein gp12013 and gp120-derived peptides14 can modulate the activity of the CD4-associated Lck in resting T lymphocytes. We have shown that an octapeptide analogue of gp120 also can modulate the activity of Lck in activated peripherial blood lymphoblasts (PBL).15 In addition, we have shown that gp120 analogues can upregulate the kinase activity of the T-cell antigen receptor-associated Fyn kinase and, also, the cell-cycle–associated pp60c-src (Src) PTK in activated PBL.15 Taken together, these results suggest that HIV/gp120-related modulation of lymphocyte signal transduction and PTK activation may contribute to the immune dysfunction associated with HIV infection.
Evidence of a functional interaction between HIV and endogenous cellular PTK has been provided by observations that CD4+ cells lacking Lck do not produce syncytia when incubated with Jurkat T cells transfected with gp120.16 In addition, cells expressing CD4 truncated or point-mutated in the cytoplasmic domain to prevent the association between Lck and CD4 replicate HIV at a higher rate than do cells expressing wild-type CD4.17 Recently, Lck has been shown to be required for HIV-induced apoptosis.18 Thus, endogenous Lck appears to be an important protein regulating HIV-induced synytium formation and apoptosis of CD4+ cells.
While normal, endogenous PTK activity may regulate certain aspects of HIV infection, evidence supporting a reciprocal effect of HIV infection on PTK activity is limited. Infection with HIV in vitro enhances the degree of PTK-mediated phosphotyrosine (ptyr) expression,19 suggesting that HIV or its gene products might activate PTK possibly contributing to HIV-related chronic immune system activation. Since we have demonstrated that gp120-derived peptides enhance the kinase activity of Fyn (15), and have, recently, also shown that infection in vitro of a human CD4+ T-cell line with HIV IIIB enhances the kinase activity of the T-cell–associated src-family PTK Lck, Src, and to a greater extent Fyn,20 we reasoned that infection with HIV in vivo would also modulate the activity of Fyn. Therefore, in this study we examined whether Fyn kinase activity is affected in patients with asymptomatic HIV or acquired immunodeficiency syndrome (AIDS) when compared with uninfected control subjects. The dysregulation of this critical T-cell activation pathway may represent an important step in immune system abnormalities associated with HIV infection.
MATERIALS AND METHODS
Patients
HIV patients. All patients were HIV antibody-positive, male homosexuals with either asymptomatic HIV infection or symptomatic AIDS (Table 1). Asymptomatic patients were free of any prescribed, HIV-related therapy or prophylaxis against opportunistic infections, and had not suffered any AIDS-defining illness. Mild oral candidiasis was not an exclusion criterion for asymptomatic patients. AIDS patients had suffered at least one AIDS-defining illness and were permitted to use antiviral therapy and opportunistic infection prophylaxis ad libitum. Exclusion criteria for all patients included use of immunosuppressive therapy (steroids), immunomodulators (cytokines, dinitrochlorobenzene), and the presence of preexisting autoimmune or neoplastic disease. CD4+ cell enumeration was performed by patient physician requisition and was provided following informed consent. CD4+ cell enumeration was performed within 4 months of blood sampling. All patients where CD8+ cells were enumerated showed a reversed ratio of CD4+:CD8+ T cells. Viral load testing was not performed. This study was approved by the Ethics Committee of The Toronto Hospital and venous blood was obtained following informed consent. Patient venous blood was drawn into sodium heparin and peripheral blood mononuclear cells (PBMC) separated immediately.
Hepatitis patients. All patients suffered from chronic hepatitis B virus (HBV) or hepatitis C virus (HCV) infection, were viremic, and were recruited from the Hepatitis Clinic of The Toronto Hospital, Western Division. Venous blood was drawn into sodium heparin and PBMC separated immediately. Blood was obtained after informed consent.
Systemic lupus erythematosis patients. Peripheral blood from five female and one male patient were examined. All but one patient had a positive anti-nuclear antibody test (ANA+) and 3 of 6 patients had additional autoantibodies detected (DNA, RNP). Three patients had anemia, one had renal disease, and one had pulmonary fibrosis. Venous blood was drawn into sodium heparin and PBMC separated immediately. Blood was obtained following informed consent.
Control subjects. All controls were confirmed HIV-1 and HIV-2 antibody negative by either enzyme-linked immunosorbent assay (ELISA) and/or Western blot. In addition, all controls were tested negative for human T-cell lymphotropic virus type I (HTLV-I), HBV, HCV, and syphilis. Selected donors were also tested for prior exposure to cytomegalovirus (CMV). Venous blood was drawn into either sodium heparin or into Red Cross (citrate-based) anticoagulant after informed consent.
Cells and Antibodies
PBMC were isolated from anticoagulated venous blood by centrifugation over Ficoll Hypaque (Pharmacia, Uppsala, Sweden). Activated peripheral blood lymphoblasts (PBL) were prepared by culturing PBMC in the presence of phytohemagglutinin (PHA) (10 μg/mL) for 2 days followed by interleukin-2 (IL-2) (100 U/mL) for 4 days. In selected experiments PBMC were depleted of CD4+ cells by two rounds of magnetic bead depletion (DynaBeads; Dynal, Great Neck, NY) from individual donors, one each with asymptomatic HIV infection, AIDS, and an uninfected control subject. In independent experiments using normal PBMC, the composition, using flow cytometric analysis, of the CD4+-depleted cell preparations was 76.7% ± 9.0% CD8+, 6.2% ± 5.6% CD14+, and 11.9% ± 7.3% CD19+, indicating the efficacy of the immunomagnetic bead negative selection method to enrich for CD8+ T cells. The human Jurkat T-cell line was obtained from the American Type Culture Collection (ATCC; Rockville, MD) and used as a positive control for intracellular Fyn. The HTLV-I–derived human cell lines C8 and S1T (a kind gift of Dr G.B. Mills, MD Anderson Cancer Center, Houston, TX) lack Fyn and Lck protein and mRNA21 and express relatively high levels of Src22 and p62c-yes (Yes).23 Polyclonal rabbit anti-Fyn (No. 428) was a kind gift of Dr A. Veillette (McGill University, Montreal, Quebec, Canada).8 This antibody is specific for mouse and human Fyn and has minimal crossreactivity with other src-family members in physiologic systems8 (A. Veillette, personal communication, May 1997). The specificity of the anti-Fyn (428) was confirmed for kinase assay using immunoprecipitation of lysates prepared from S1T and C8 T cells, and the specificity of the Western immunoblot confirmed using immunoprecipitation of lysates prepared from normal resting PBMC followed by specific Western immunoblotting. Antibodies used for Western immunoblotting were monoclonal anti-Fyn and anti-Lck (Transduction Laboratories, Lexington, KY), monoclonal anti-Src (Upstate Biotechnology, Inc [UBI], Lake Placid, NY), and rabbit polyclonal anti-c-Yes (Santa Cruz Biotechnology, Santa Cruz, CA). Monoclonal anti-p50csk (Csk; Transduction Laboratories) for immunoprecipitation was cross-linked to Protein G-Sepharose beads (Pharmacia Biotech, Uppsala, Sweden) before immunoprecipitation as previously described.24 Briefly, anti-Csk was added to Protein G-Sepharose beads and rotated overnight at 4°C. The immune complexes were then washed once in 0.2 mol/L borate buffer, pH 9.0, and then rotated for 1 hour in borate buffer containing 5.2 mg of the cross-linking agent dimethylpimelimidate (Aldrich Chemical, Milwaukee, WI). The reaction was stopped by washing the complexes with 0.2 mol/L ethanolamine, pH 8.0, followed by washing using lysis buffer (see below). Rabbit polyclonal anti-Csk for Western immunoblotting was a gift from Dr A. Veillette. Monoclonal anti-CD45 was purchased from Transduction Laboratories. Goat-antimouse or antirabbit IgG conjugated to horseradish peroxidase were purchased from BioRad (Hercules, CA). Enhanced chemiluminescence (ECL; Amersham, Little Chalfont, UK) was performed according to the manufacturer. c-Src C-terminal peptide was purchased from BIOMOL (Plymouth Meeting, PA). Monoclonal antiphosphotyrosine 4G10 was purchased from UBI.
Kinase Assays and Western Immunoblots
Kinase assays and Western immunoblots were performed as described15,22 after immunoprecipitation of cellular lysates with rabbit anti-Fyn antiserum No. 428. Briefly, up to 2 × 107 PBMC were centrifuged, pellets lysed in 1.0 mL RIPA (1% NP-40, 0.1% sodium dodecyl sulfate [SDS], 0.1% Na3 deoxycholate, 50 mmol/L HEPES, pH 7.3, 150 mmol/L NaCl, 1 mmol/L sodium orthovanadate, 50 μmol/L ZnCl2 , 2 mmol/L EDTA, 2 mmol/L phenylmethyl sulfonyl fluoride [PMSF ]) lysis buffer and 5 × 106 cell equivalents immunoprecipitated with anti-Fyn conjugated to protein A-Sepharose CL4B (Pharmacia). For kinase assays, immunoprecipitates were washed and incubated with 5 μCi [γ32P]ATP (ICN Biomedicals, Irvine, CA) in kinase buffer (50 mmol/L HEPES, pH 7.23, 150 mmol/L NaCl, 1 mmol/L MgCl2 , 1 mmol/L MnCl2 , 0.5% NP-40) containing acid-treated enolase (E) (ICN; 1.0 μg/condition) as an exogenous substrate.25 Proteins were separated by reduced SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and electrotransferred to Immobilon-P membranes (Millipore Corp, Bedford, MA) which were then autoradiographed. For Western immunoblotting, the kinase blot from above was first blocked using 5% skim milk in Tris-buffered saline (TBS), then probed with mouse antihuman-Fyn monoclonal antibody (Transduction Labs) at a dilution of 1:250 followed by goat-antimouse IgG conjugated to horseradish peroxidase (BioRad). Western blots were developed by ECL (Amersham).
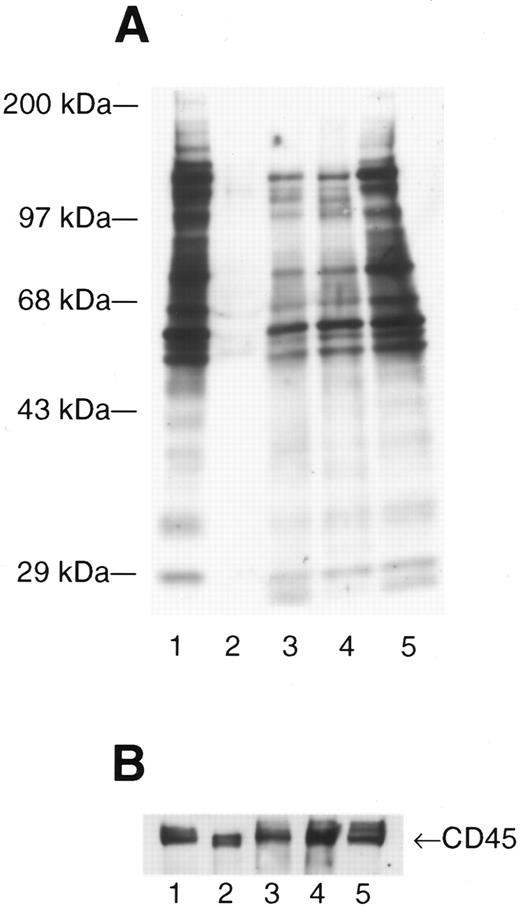
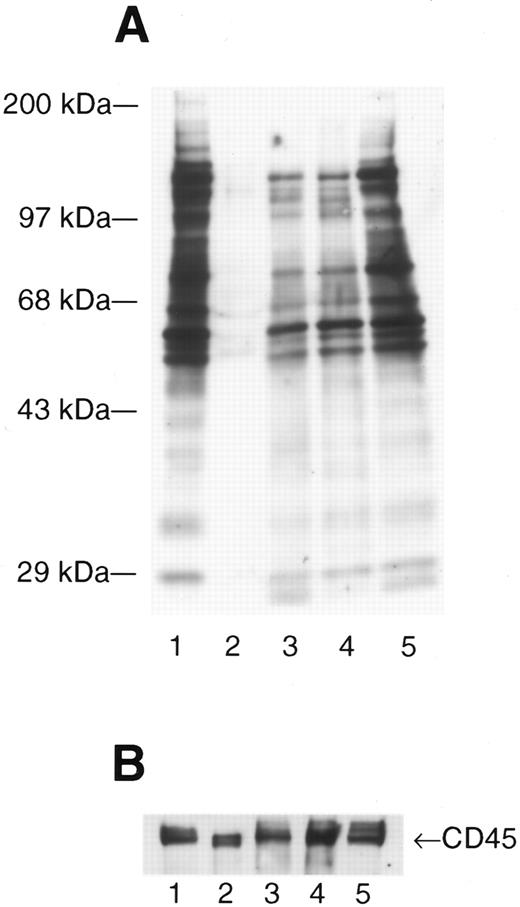
Infection with HIV produces enhanced and novel patterns of phosphotyrosine-containing proteins. Activated peripheral blood lymphocytes (PBL) (lane 1), resting PBL (lane 2), and resting PBL from three patients with asymptomatic HIV infection (lanes 3 through 5) were lysed in RIPA lysis buffer, the total protein measured (Pierce), and 10 μg of total protein loaded onto each lane prior to fractionation by SDS-PAGE. The separated proteins were electrotransferred to nitrocellulose membranes and expression of phosphotyrosine (ptyr)-containing proteins analyzed by Western immunoblot. (A) Blots were probed with mouse anti-ptyr followed by goat-antimouse IgG conjugated to horseradish peroxidase. Blots were developed by enhanced chemiluminescence. Molecular mass standards are indicated on the left. (B) The blot was stripped and reprobed with anti-CD45 to confirm equal loading.
Infection with HIV produces enhanced and novel patterns of phosphotyrosine-containing proteins. Activated peripheral blood lymphocytes (PBL) (lane 1), resting PBL (lane 2), and resting PBL from three patients with asymptomatic HIV infection (lanes 3 through 5) were lysed in RIPA lysis buffer, the total protein measured (Pierce), and 10 μg of total protein loaded onto each lane prior to fractionation by SDS-PAGE. The separated proteins were electrotransferred to nitrocellulose membranes and expression of phosphotyrosine (ptyr)-containing proteins analyzed by Western immunoblot. (A) Blots were probed with mouse anti-ptyr followed by goat-antimouse IgG conjugated to horseradish peroxidase. Blots were developed by enhanced chemiluminescence. Molecular mass standards are indicated on the left. (B) The blot was stripped and reprobed with anti-CD45 to confirm equal loading.
For Western immunoblots for total ptyr, 5 × 106 cells were lysed in 1.0 mL RIPA lysis buffer, the protein concentration was determined by means of the bicinchoninic acid (BCA) assay (Pierce, Rockford, IL), and equal amounts of total proteins separated on SDS-PAGE. The fractionated protein were electrotransferred to nitrocellulose membranes (Schleicher & Schuell, Inc, Keene, NH) and expression of ptyr analyzed by Western blot as described.15 Blots were probed with mouse anti-ptyr 4G10 (UBI) followed by goat-antimouse IgG conjugated to horseradish peroxidase (BioRad). Blots were developed by ECL (Amersham). The blot was stripped (2% SDS, 0.1 mol/L 2-mercaptoethanol [2-ME], 1 mol/L TRIS, pH 6.8 at 50°C for 30 minutes) and confirmation of equal protein loading onto the gel was done by Western immunoblotting for the hematopoietic cell surface protein CD45.
Fyn-Specific Kinase Activity
Determination of the relative specific kinase activity of Fyn was done as previously described for Src kinase26 using the exogenous substrate enolase. Briefly, autoradiographs (kinase activity) and chemiluminescence immunoblots (protein level) were analyzed by densitometry and the relative Fyn-specific kinase activity determined as the ratio of Fyn kinase activity (optical density of phosphorylated enolase) to total Fyn protein (optical density of Fyn Western blot). All measurements of the relative Fyn-specific activity were normalized to uninfected samples by dividing the calculated specific activity by the mean Fyn-specific kinase activity from at least four uninfected control samples run on each gel. Therefore, the values were expressed as a fraction of control. Thus, results from different gels can be compared because they are normalized to uninfected controls and the variation in uninfected controls can be assessed by dividing individual control values by the mean of all control values.
Csk Activity
Tyrosine kinase activity of Csk was assayed as described previously27 by immunoprecipitation of lysates as described above using anti-Csk (Transduction Laboratories). The immunoprecipitates were washed four times in kinase buffer lacking MnCl2 and the precipitated Csk incubated in 40 μL of kinase buffer containing 10 μCi of [γ32P]ATP (Amersham) and 5 μg of a peptide (Thr-Ser-Thr-Glu-Pro-Gln-Tyr-Gln-Pro-Gly-Glu-Asn-Leu) (BIOMOL, lot K1940) derived from the amino acid sequence (aa 524-536) surrounding the Src tyrosine kinase C-terminal negative regulatory site (Tyr 530). The mixture was incubated for 15 minutes at 37°C and then centrifuged. The supernatant (26 μL) was then blotted onto phosphocellulose paper (Whatman International Ltd, Maidstone, UK) and washed eight times with 10% phosphoric acid. The amount of γ32P incorporated into the c-Src peptide was determined by scintillation counting and compared to a control consisting of the immune complex without lysate.
Specificity of anti-Fyn (428). (A) Control Western immunoblot. Anti-Fyn (428) was used to immunoprecipitate resting PBMC (5 × 106 cell equivalents), the immunoprecipitated proteins fractionated by SDS-PAGE and Western immunoblotting used to check the specificity of the immunoprecipitating antibody to recognize src-family PTK. Total cell lysate was used to control for Fyn, Lck, c-Src, and c-Yes proteins. (BI) Control kinase assay. Lysate containing 25 × 106 cell equivalents of Fyn+ Jurkat cells (lane 2) or Fyn− C8 (lane 3) and S1T (lane 4) cell lines were immunoprecipitated with rabbit anti-Fyn and immune-complexes washed and incubated with 5 μCi [γ32P] in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography. Lane 1 is an immunoprecipitating antibody control that also serves as a control for background enolase phosphorylation. (BII) The Immobilon-P membrane in (BI) was blocked in 5% skim-milk, probed with a monoclonal anti-Fyn, followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The positions of the molecular weight standards are indicated and arrows show the location of fyn, enolase (E), and the immunoprecipitating antibody heavy chains [Ig(H)], respectively.
Specificity of anti-Fyn (428). (A) Control Western immunoblot. Anti-Fyn (428) was used to immunoprecipitate resting PBMC (5 × 106 cell equivalents), the immunoprecipitated proteins fractionated by SDS-PAGE and Western immunoblotting used to check the specificity of the immunoprecipitating antibody to recognize src-family PTK. Total cell lysate was used to control for Fyn, Lck, c-Src, and c-Yes proteins. (BI) Control kinase assay. Lysate containing 25 × 106 cell equivalents of Fyn+ Jurkat cells (lane 2) or Fyn− C8 (lane 3) and S1T (lane 4) cell lines were immunoprecipitated with rabbit anti-Fyn and immune-complexes washed and incubated with 5 μCi [γ32P] in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography. Lane 1 is an immunoprecipitating antibody control that also serves as a control for background enolase phosphorylation. (BII) The Immobilon-P membrane in (BI) was blocked in 5% skim-milk, probed with a monoclonal anti-Fyn, followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The positions of the molecular weight standards are indicated and arrows show the location of fyn, enolase (E), and the immunoprecipitating antibody heavy chains [Ig(H)], respectively.
Statistical Analysis
Data are presented as means ± SEM and statistical significance of the difference in means between HIV-infected patients with respect to uninfected controls was determined using a Student's t-test.
RESULTS
Effect of HIV Infection on Expression of Phosphotyrosine (ptyr)-Containing Proteins
The src-family PTK having molecular masses 54 kD to 62 kD28 can be activated in vitro to autophosphorylate on tyrosine residues and also phosphorylate a number of other protein substrates, as shown in Fig 1A, comparing resting lymphocytes (Fig 1A, lane 2) to lymphocytes activated with PHA and IL-2 (Fig 1A, lane 1). Similar to in vitro–activated lymphocytes (Fig 1A, lane 1) or Jurkat T cells infected with HIV,19 resting lymphocytes from HIV-infected patients have dramatically enhanced expression of ptyr-containing proteins (Fig 1A, lanes 3 through 5). Major ptyr-containing bands appear at 54 to 62 kD and at approximately 72 kD up to 150 kD (Fig 1A, lanes 3 through 5). Some patients had additional tyrosine phosphorylation of proteins having apparent molecular masses of 20 to 25 kD (Fig 1A, lanes 3 and 5) but, also, lacked some ptyr proteins when compared with PBMC activated in vitro. This suggests that the biochemical nature of HIV-induced lymphocyte activation may be distinct from that induced by in vitro immunologic stimuli. Western immunoblotting for CD45, a cell surface protein found on all hematopoietic cells, indicated that equal amounts of total protein were loaded in each lane (Fig 1B).
Levels of Fyn protein are low in asymptomatic HIV infection but relative specific kinase activity is elevated. Lysate containing 5 × 106 cell equivalents was immunoprecipitated with rabbit anti-Fyn and immune complexes washed and incubated with 5 μCi [γ32P]ATP in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography (Kinase Assay). Western immunoblotting was performed by probing the kinase blot with mouse anti-Fyn monoclonal antibody followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The relative Fyn specific kinase activity was estimated using the ratio of Fyn kinase activity (using tyrosine phosphorylation of enolase) to Fyn protein levels after densitometry analyses of the autoradiograph (Kinase Assay) and chemiluminescence blots (Western blot), respectively. The figure is representative of the results obtained with all patients and normal controls. Lanes 1 and 2 are patients with asymptomatic HIV infection, lanes 3 and 4 represent patients having AIDS, and lanes 5 and 6 are uninfected normal controls.
Levels of Fyn protein are low in asymptomatic HIV infection but relative specific kinase activity is elevated. Lysate containing 5 × 106 cell equivalents was immunoprecipitated with rabbit anti-Fyn and immune complexes washed and incubated with 5 μCi [γ32P]ATP in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography (Kinase Assay). Western immunoblotting was performed by probing the kinase blot with mouse anti-Fyn monoclonal antibody followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The relative Fyn specific kinase activity was estimated using the ratio of Fyn kinase activity (using tyrosine phosphorylation of enolase) to Fyn protein levels after densitometry analyses of the autoradiograph (Kinase Assay) and chemiluminescence blots (Western blot), respectively. The figure is representative of the results obtained with all patients and normal controls. Lanes 1 and 2 are patients with asymptomatic HIV infection, lanes 3 and 4 represent patients having AIDS, and lanes 5 and 6 are uninfected normal controls.
Specificity of Anti-Fyn (428)
The anti-Fyn antibody used in this study was assessed for its specificity for src-family PTK found in human T cells. Figure 2A shows that, by Western immunoblotting, the anti-Fyn (428) immunoprecipitated lysates from normal resting PBMC are specific for Fyn with no apparent crossreactivity for Lck, c-Src, or c-Yes under the conditions used. To further confirm the specificity of anti-Fyn in kinase assays, anti-Fyn (428) immunoprecipitates of lysates from S1T and C8 T cells, which lack Fyn but express c-Src and c-Yes,21-23 demonstrate no autophosphorylated bands or phosphorylation of exogenous enolase (Fig 2B).
Fyn Kinase Activity and Protein Levels in HIV-Infected Patients
Because the profound increase in tyrosine phosphorylated proteins seen in HIV infected patients (Fig 1A) could represent the action of activated src-family PTK, and as our previous in vitro studies20 suggested that the kinase activity of the src-family PTK Fyn is the most severely altered after infection with HIV, we examined Fyn activity in HIV-infected patients.
Figure 3 is representative of the kinase activity and amount of Fyn protein in PBMC from 2 patients with asymptomatic HIV infection, 2 patients with symptomatic AIDS, and 2 HIV-seronegative control individuals. Enolase was included as an exogenous substrate24,25 to measure tyrosine kinase activity of Fyn. Using enolase is a more sensitive indicator of kinase activity than autophosphorylation,25 particularly when the Fyn protein levels are very low. Indeed, patients with asymptomatic HIV infection had less Fyn protein compared to uninfected controls or patients with AIDS. The amount of Fyn protein (in arbitrary optical density units ± SEM) measured in a representative cohort of subjects was determined to be: 3.67 ± 1.38, asymptomatic (n = 9); 24.41 ± 5.40, AIDS (n = 4); and 36.83 ± 2.79, uninfected controls (n = 5).
Fyn-Specific Kinase Activity in HIV-Infected Patients
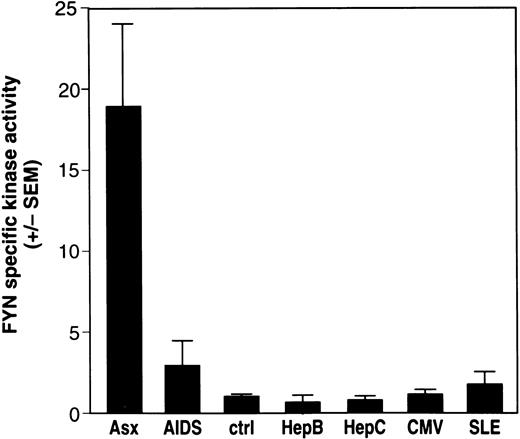
The relative Fyn-specific kinase activity, a ratio of kinase activity to the level of enzyme, was generated for Fyn using immunoprecipitated Fyn to phosphorylate enolase, an exogenous substrate,24 followed by Western blot analysis of the same kinase blot using chemiluminescence to determine the amount of Fyn protein responsible for the observed phosphorylation of enolase. As shown in Fig 4, all 14 patients with asymptomatic HIV infection had profoundly elevated levels of Fyn-specific kinase activity (mean increase, 18.87 ± 4.47; range, 3.16 to 56) compared with that of uninfected controls (mean, 1.28 ± 0.29; range, 0.29 to 2.26; p = 1.33 × 10−9). Patients with AIDS also showed a significantly elevated (p = 1.30 × 10−5) Fyn-specific kinase activity (mean increase, 3.36 ± 1.62; range, 1.07 to 7.06) compared with uninfected controls. However, the level of Fyn-specific kinase activity was increased much less than in asymptomatic patients. Furthermore, it is important to note that although the Fyn protein levels in lysates from patients with AIDS sometimes were very similar to Fyn protein levels found in patients with asymptomatic HIV infection, the Fyn-specific kinase activity was always lower (data not shown). This suggests that the HIV infection–related effect on the kinase activity of Fyn decreases as patients progress to symptomatic AIDS.
Fyn-specific kinase activity is elevated in patients with asymptomatic HIV infection. The study population consists of patients with asymptomatic (Asx) HIV infection (n = 14), symptomatic AIDS (AIDS) (n = 4), normal, HIV-seronegative controls (ctrl) (n = 47), patients with chronic hepatitis B (HepB) infection (n = 6), patients with chronic hepatitis C (HepC) infection (n = 7), normal donors with prior exposure to CMV (n = 7), and patients with systemic lupus erythematosis (SLE, n = 6). PBMC were isolated from whole blood and lysate from 5 × 106 cells immunoprecipitated with antibodies specific to p59fyn(T). Immune-complex kinase assays, Western immunoblotting of Fyn protein, and calculation of the relative specific kinase activity of Fyn was done as described in Fig 3. The results represent the mean Fyn specific kinase activity ± SEM.
Fyn-specific kinase activity is elevated in patients with asymptomatic HIV infection. The study population consists of patients with asymptomatic (Asx) HIV infection (n = 14), symptomatic AIDS (AIDS) (n = 4), normal, HIV-seronegative controls (ctrl) (n = 47), patients with chronic hepatitis B (HepB) infection (n = 6), patients with chronic hepatitis C (HepC) infection (n = 7), normal donors with prior exposure to CMV (n = 7), and patients with systemic lupus erythematosis (SLE, n = 6). PBMC were isolated from whole blood and lysate from 5 × 106 cells immunoprecipitated with antibodies specific to p59fyn(T). Immune-complex kinase assays, Western immunoblotting of Fyn protein, and calculation of the relative specific kinase activity of Fyn was done as described in Fig 3. The results represent the mean Fyn specific kinase activity ± SEM.
Effect of Hepatitis Infection on Fyn-Specific Kinase Activity
As another viral disease that, like HIV, can be chronic with viremia, we examined blood from patients having hepatitis. Infection with hepatitis viruses does not enhance the relative specific kinase activity of Fyn. Six patients with HBV infection and 7 patients with HCV virus infection had a mean Fyn-specific kinase activity of 0.68 ± 0.33 and 0.74 ± 0.10, respectively, relative to uninfected controls (Fig 4). Although these data are not conclusive, they support a hypothesis that elevated Fyn-specific kinase activity is a response unique to infection with HIV.20
Effect of Prior Exposure to CMV on Fyn-Specific Kinase Activity
Whether normal donors had prior exposure to CMV was examined to represent a viral infection where, as with HIV, the virus is known to integrate into the genomic DNA. Seven CMV+ normal donors were identified from 20 donors tested. There was no significant variance of the relative Fyn-specific kinase activity of CMV+ donors compared with CMV− donors (Fig 4).
Relative Fyn-Specific Kinase Activity in Patients With Systemic Lupus Erythematosis (SLE)
To better assess the specificity of the increases in Fyn-specific activity in the cells from patients with HIV infection, we examined the Fyn kinase activity from cells of patients having a disease of polyclonal activation. Six patients with SLE were evaluated and showed a mean Fyn-specific kinase activity of 1.78 ± 0.66. Thus, the elevation of Fyn-specific kinase activity in HIV-infected individuals is a phenomenon that is distinct from a disease of chronic immune activation such as SLE.
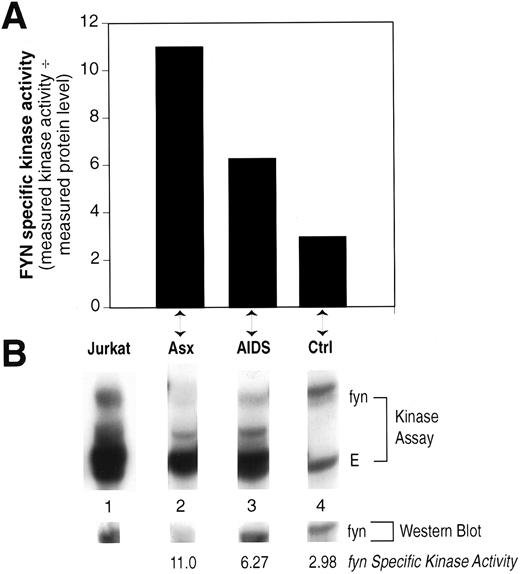
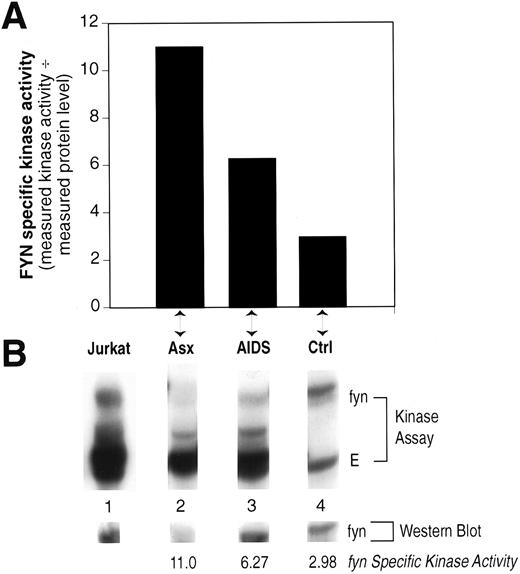
Fyn-Specific Kinase Activity in CD8+-Enriched Cells
The relative Fyn-specific kinase activity was increased in patients with AIDS, albeit not to the level seen in asymptomatic patients (Fig 4), even though these patients have very few CD4+ T lymphocytes (Table 1). Therefore, we examined whether this effect may be a result of abnormal Fyn activity in CD8+ T lymphocytes. Results shown in Fig 5 indicate that increased Fyn-specific kinase activity is, indeed, observed in PBMC that had been depleted of CD4+ cells (<0.5% CD4+ cells as determined by FACS analysis, data not shown). These results implicate abnormal Fyn kinase activity in both CD8+ and CD4+ T lymphocytes from HIV-infected patients. Furthermore, these results imply that this effect of HIV on Fyn activity is not a function of the gp120/CD4 interaction. This is consistent with our previous observations that the activation of src-family PTKs after HIV infection of CD4+ T-cell lines in vitro was independent of the interaction between gp120 and CD4.20
Peripheral blood lymphocytes enriched for CD8+ cells show increased Fyn specific kinase activity. The relative Fyn specific kinase activity was determined as described in Fig 3 from PBMC after depletion of CD4+ cells by two rounds of anti-CD4 magnetic beads. (A) The calculated Fyn-specific kinase activity from one each asymptomatic (Asx) HIV patient, AIDS patient, and normal, HIV seronegative control (Ctrl). (B) The Kinase assay and Western blot obtained for each of the individuals in (A) compared to a human T-cell line, Jurkat. The samples were run on the same gel, transferred to Immobilon-P, the kinase result visualized by autoradiography, and then the same blot probed with monoclonal anti-Fyn by Western immunoblotting and the results visualized by ECL. The lanes were cut from the original to align properly beneath each designated sample shown in (A).
Peripheral blood lymphocytes enriched for CD8+ cells show increased Fyn specific kinase activity. The relative Fyn specific kinase activity was determined as described in Fig 3 from PBMC after depletion of CD4+ cells by two rounds of anti-CD4 magnetic beads. (A) The calculated Fyn-specific kinase activity from one each asymptomatic (Asx) HIV patient, AIDS patient, and normal, HIV seronegative control (Ctrl). (B) The Kinase assay and Western blot obtained for each of the individuals in (A) compared to a human T-cell line, Jurkat. The samples were run on the same gel, transferred to Immobilon-P, the kinase result visualized by autoradiography, and then the same blot probed with monoclonal anti-Fyn by Western immunoblotting and the results visualized by ECL. The lanes were cut from the original to align properly beneath each designated sample shown in (A).
Mechanism of Increased Fyn Kinase Activity in HIV-Infected Patients
To address the possible mechanism of the increased relative specific kinase activity of Fyn in HIV-infected asymptomatic patients, we assessed the activity of the C-terminal Src Kinase (Csk), known to regulate src-family PTK.29,30 Csk functions to negatively regulate the kinase activity of src-family PTK by phosphorylating their C-terminal regulatory tyrosine.28-30 Thus, a decrease in Csk kinase activity may be associated with a concomitant increase in Fyn kinase activity.29 30 Figure 6 shows a representative experiment where we found that the activity of Csk was markedly decreased in asymptomatic HIV-infected patients compared to uninfected controls without differences in Csk protein levels. In three independent experiments the kinase activity of Csk isolated from HIV-infected asymptomatic patients was found to be decreased by 68% ± 16% compared with normal, uninfected controls.
The kinase activity of Csk is decreased in HIV-infected patients. (A) Csk protein tyrosine kinase was immunoprecipitated from lysates containing equal total protein as measured by the bicinchoninic acid method, from uninfected individuals and from asymptomatic HIV+ patients. The immune complexes were washed, incubated with c-Src C-terminal peptide [Thr-Ser-Thr-Glu-Pro-Gln-Tyr-Gln-Pro-Gly-Glu-Asn-Leu] and 10 μCi [γ32P]ATP at 37°C for 15 minutes, centrifuged, and the supernatant blotted onto phosphocellulose paper and washed with 10% phosphoric acid. After drying, the blotted phosphocellulose paper was tested for 32P-containing c-Src peptide by scintillation counting. The results represent the relative Csk kinase activity of uninfected control and HIV+ patient after subtracting the background control (immune-complex without lysate; 350 cpm). (B) Western immunoblotting of 50-kD Csk protein for (A). Antibody control consists of immune complex without lysate. These results are representative of three independent experiments.
The kinase activity of Csk is decreased in HIV-infected patients. (A) Csk protein tyrosine kinase was immunoprecipitated from lysates containing equal total protein as measured by the bicinchoninic acid method, from uninfected individuals and from asymptomatic HIV+ patients. The immune complexes were washed, incubated with c-Src C-terminal peptide [Thr-Ser-Thr-Glu-Pro-Gln-Tyr-Gln-Pro-Gly-Glu-Asn-Leu] and 10 μCi [γ32P]ATP at 37°C for 15 minutes, centrifuged, and the supernatant blotted onto phosphocellulose paper and washed with 10% phosphoric acid. After drying, the blotted phosphocellulose paper was tested for 32P-containing c-Src peptide by scintillation counting. The results represent the relative Csk kinase activity of uninfected control and HIV+ patient after subtracting the background control (immune-complex without lysate; 350 cpm). (B) Western immunoblotting of 50-kD Csk protein for (A). Antibody control consists of immune complex without lysate. These results are representative of three independent experiments.
DISCUSSION
The cytosolic protein tyrosine kinase Fyn is thought responsible for initiating the biochemical signals following engagement of the T-cell antigen receptor (TCR) present on both CD4+ and CD8+ T lymphocytes.6-8,11 Fyn can be co-immunoprecipitated as part of the TCR complex6 and TCR signaling and IL-2 production are augmented in Fyn transgenic mice7 and in in vitro transfection experiments.8,31 Moreover, mutant mice that do not express Fyn (by homologous recombination in embryonic stem cells resulting in a “knockout” of the gene expression) produce thymocytes and also peripheral blood T cells that show diminished responses to TCR agonists and abnormal IL-2 production.10,11 Thus, Fyn is a critical PTK involved in early signaling events leading to T-cell activation and normal T-cell function.32
In the report herein, we have shown that HIV, but not HBV, HCV, or prior exposure to CMV, alters the kinase activity and protein levels of Fyn in infected patients. The biochemical mechanism of alteration in Fyn protein and kinase activity in asymptomatic HIV-infected patients is unknown, but may be related to chronic cellular activation resulting in the loss of regulation of Fyn kinase activity, protein synthesis, or both. Even so, this effect of HIV appears to be a unique phenomenon associated with HIV infection as patients with SLE, a polyclonal activation disease, do not show similar alterations in Fyn kinase activity.
It is known that CD4+ lymphocytes from patients with AIDS turn over rapidly33,34 and it is logical to propose that cells from asymptomatic patients have longer life spans because of lower viral burden.35 Thus, cells from asymptomatic patients may be in a state of activation longer than those from patients with AIDS, leading to “exhaustion” of Fyn protein synthesis. Alternatively, the HIV-induced effect on Fyn may be a direct effect of HIV on molecules responsible for the regulation of the activity of this PTK. For example, protein tyrosine phosphatases can regulate the kinase activity of src-family PTK and a number of potential regulatory phosphatases have been described.36,37 Also, mouse cells that have been genetically altered to lack the src-family negative regulatory PTK, Csk, have chronically active Fyn kinase but reduced levels of Fyn protein.29,30 These Csk-deficient cells also have elevated expression of ptyr-containing proteins. Thus, Csk-deficient cells are remarkably similar to cells from patients with asymptomatic HIV infection (Figs 1 and 3). Indeed, our initial results implicate a defect in Csk kinase activity as likely responsible for the abnormal increase in Fyn kinase activity observed in HIV-infected patients (Fig 6). In the Csk knockout animal it has been postulated that effects on Fyn result from decreased phosphorylation on its COOH-terminal regulatory tyrosine and a feedback mechanism involving Csk.29 However, it remains for future studies to determine the extent to which viral effects on Csk-family proteins affects Fyn protein tyrosine kinase activity and the exact mechanism of this effect.
In our study, patients with asymptomatic HIV infection had very low levels of Fyn protein compared to uninfected control subjects, but greatly elevated Fyn kinase activity. Cayota et al38 have previously shown that the CD4+ lymphocytes of 18 of 25 patients with asymptomatic HIV infection had similar Fyn protein levels as uninfected controls whereas 7 of 25 patients had Fyn protein levels higher than uninfected controls. The differences between this report and our findings cannot be explained by differences in numbers of CD4+ T cells. Our patients had mean CD4 cell counts (572 cells/μL) higher than those studied by Cayota et al (395 cells/μL) yet had lower Fyn protein levels. As described above, our findings of low levels of Fyn protein having high Fyn kinase activity in the face of decreased Csk kinase activity correlates with similar findings in Csk knockout mice having dysregulation of Fyn kinase activity. However, we do find that the amount of Fyn protein in our patients approaches uninfected levels as HIV disease progresses, although significant exceptions were seen as shown in Fig 3. Thus, in our studies, it is likely the reason for the modulation of Fyn protein levels is similar to that found in the Csk-knockout animal and, as patients progress in their disease, the effect becomes less pronounced. Thus, as indicated by CD4+ cell counts, the patients studied by Cayota et al38 were more advanced in their disease; therefore, based on our data, these patients would be expected to have levels of Fyn protein more similar to uninfected levels.
As summarized in Table 1 and shown in Fig 3, the relative Fyn-specific kinase activity is elevated in patients with asymptomatic HIV infection and decreases in patients with AIDS; therefore, Fyn-specific kinase activity may be elevated shortly after infection in vivo and, thus, has the potential to be diagnostic of seronegative HIV infection. Indeed, in vitro infection experiments have shown that Fyn kinase activity increases within 30 minutes after HIV infection of the human CD4+ Jurkat T-cell line.20 Furthermore, comparing our results of the relative specific activity of Fyn isolated from patients with asymptomatic infection or AIDS suggests that monitoring of Fyn-specific kinase activity may be prognostic of the progression to symptomatic HIV disease.
Tremblay et al17 have suggested that high PTK activity would result in low viral replication. Thus, elevated Fyn-specific kinase activity may play a role in maintaining the low viral load characteristic of the asymptomatic state.33 It is more likely, however, that chronic elevation of Fyn kinase activity is responsible, at least in part, for the failure and eventual exhaustion of the immune response.1 Thus, chronic activation of Fyn could result in an inability for affected T cells to respond to additional stimuli as a result of a defective signaling pathway.39 The increase in Fyn-specific kinase activity may act in a way to sabotage the immune system and allow HIV to evade immune recognition. Indeed, Cayota et al38 have shown that most asymptomatic HIV-infected patients have a profound downregulation of their immediate response to external activation through the T-cell antigen receptor. Furthermore, it is well documented that PBMC from HIV-infected individuals have decreased responses in vitro to activation by mitogens, recall antigens, and stimulation by anti-CD3, with or without costimulation with anti-CD28.38 40-42
Our findings may also extend to the mechanism of HIV-related apoptosis of CD4+ T cells43 as chronic activation of Fyn may provide an incomplete activation signal that may lead to an increased rate of programmed cell death in these cells,44 particularly upon reactivation.18,45 Our findings that CD4+ T-cell–depleted peripheral blood cells (Fig 4) also have increased Fyn-specific kinase activity suggests that this effect of HIV may play a role in the eventual exhaustion of the CD8+ CTL population of HIV-specific killer T cells previously described.46 Although recent reports implicate direct in vivo HIV infection of CD8+ T lymphocytes,47 this is controversial and may not be an explanation for our results with CD8+-enriched cells. It is possible that activation of Fyn occurs indirectly as a result of the chronic activation of the immune system during HIV infection1 and that other mediators produced during HIV infection may be responsible for the increased activation of Fyn.
Our novel findings of HIV inducing chronic activation of Fyn not only contribute to our understanding of asymptomatic HIV infection, but make predictions regarding the early detection and progression of the disease. Moreover, our results provide a novel rationale to explain the unusual immunologic abnormalities associated with the pathogenesis of HIV infection.
ACKNOWLEDGMENT
The willing informed cooperation of all subjects is gratefully appreciated. The authors thank Drs Rob Inman, Robert McMillan, David Tilley, Sharon Walmsley, Jenny Heathcote, Colina Yim, and Paula Watson for their cooperation in recruiting patients and obtaining blood samples for this study. We are grateful to John Piovesan and Meena Bali for technical assistance. We also express our gratitude to Alice Peters for advice and assistance in statistical analysis. We are grateful to Dr Gordon B. Mills for providing cells and Dr Andre Veillette for the gift of anti-Fyn (428) and anti-Csk used in these studies.
Supported by grants from The Canadian Red Cross Society Blood Services R&D Fund and CANFAR, the Canadian Foundation for AIDS Research, and the National Health Research and Development Program (NHRDP) of Canada. D.J.P. is the recipient of a Toronto Hospital HIV Research Fellowship award. S.Y. is a recipient of a Natural Sciences and Engineering Research Council (NSERC) of Canada Fellowship award.
Address reprint requests to Donald R. Branch, PhD, The Canadian Red Cross, 67 College St, Toronto, Ontario, Canada M5G 2M1.

![Fig. 2. Specificity of anti-Fyn (428). (A) Control Western immunoblot. Anti-Fyn (428) was used to immunoprecipitate resting PBMC (5 × 106 cell equivalents), the immunoprecipitated proteins fractionated by SDS-PAGE and Western immunoblotting used to check the specificity of the immunoprecipitating antibody to recognize src-family PTK. Total cell lysate was used to control for Fyn, Lck, c-Src, and c-Yes proteins. (BI) Control kinase assay. Lysate containing 25 × 106 cell equivalents of Fyn+ Jurkat cells (lane 2) or Fyn− C8 (lane 3) and S1T (lane 4) cell lines were immunoprecipitated with rabbit anti-Fyn and immune-complexes washed and incubated with 5 μCi [γ32P] in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography. Lane 1 is an immunoprecipitating antibody control that also serves as a control for background enolase phosphorylation. (BII) The Immobilon-P membrane in (BI) was blocked in 5% skim-milk, probed with a monoclonal anti-Fyn, followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The positions of the molecular weight standards are indicated and arrows show the location of fyn, enolase (E), and the immunoprecipitating antibody heavy chains [Ig(H)], respectively.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/9/10.1182_blood.v90.9.3603/4/m_bl_0043f2.jpeg?Expires=1763668683&Signature=vqtFL3B99yRYtnvJlo-GKcTwzVGZL48ImRknv3X~VdZe6KZVTJpoqEarKceugEUOIXWtUsBv4A257hSWbALPYXi3~PqVzQeA0QKIEcwTKgjva6Kd5lgpk0mObCQACTqSEIz9xHRMO0AySKrsOeeCjYiCsR3KWPI3eroA03RSooJAoNJ5gZnz-0xw7YAnhw1IxoJvxigjf-vI1o626ARTBJ-Yjms6-fWEmvAkEbxLKzhk3ug5R0jDE2XRWRbAqacDYFxMddhwzixJGFbl5FPqqmG42bVKTfevPWKzOw1ldadgUlA4nfyAbj3LpQwgdBwTWO0D2ketG1Ip89bPiP~ZCw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 3. Levels of Fyn protein are low in asymptomatic HIV infection but relative specific kinase activity is elevated. Lysate containing 5 × 106 cell equivalents was immunoprecipitated with rabbit anti-Fyn and immune complexes washed and incubated with 5 μCi [γ32P]ATP in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography (Kinase Assay). Western immunoblotting was performed by probing the kinase blot with mouse anti-Fyn monoclonal antibody followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The relative Fyn specific kinase activity was estimated using the ratio of Fyn kinase activity (using tyrosine phosphorylation of enolase) to Fyn protein levels after densitometry analyses of the autoradiograph (Kinase Assay) and chemiluminescence blots (Western blot), respectively. The figure is representative of the results obtained with all patients and normal controls. Lanes 1 and 2 are patients with asymptomatic HIV infection, lanes 3 and 4 represent patients having AIDS, and lanes 5 and 6 are uninfected normal controls.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/9/10.1182_blood.v90.9.3603/4/m_bl_0043f3.jpeg?Expires=1763668683&Signature=hwFAmb5KQGNkCJbg-rmMkOrzbEmuKWnbEIWtg3J10R3hCzyu3RBGUSiUpBieoA3x2-5JXPdPvNWAftaKPmzUooE2~hEr5a8aNClwzTZtNuOOa7a2RQEtWTkp4Hi3nHxZ0Nq9zr6MUNh8BDu~jNuPyInypzpgCeBB8BPlXPIqiPfuu99K59C95NkUjD14h-5rgFf9GRaDXxWJVXza~w326lI2ImBx-oF5FmhNP90AvE~j3zipSwWxZV3rAtz2XlyxHrqLRXM4l6-k6aB-VXsTOI818VLFTEBBl6Uu~ORGROfbjuHEjdPRX9pd3XyCamLzKTAWU~J7kHpO-GzWyOaOug__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 6. The kinase activity of Csk is decreased in HIV-infected patients. (A) Csk protein tyrosine kinase was immunoprecipitated from lysates containing equal total protein as measured by the bicinchoninic acid method, from uninfected individuals and from asymptomatic HIV+ patients. The immune complexes were washed, incubated with c-Src C-terminal peptide [Thr-Ser-Thr-Glu-Pro-Gln-Tyr-Gln-Pro-Gly-Glu-Asn-Leu] and 10 μCi [γ32P]ATP at 37°C for 15 minutes, centrifuged, and the supernatant blotted onto phosphocellulose paper and washed with 10% phosphoric acid. After drying, the blotted phosphocellulose paper was tested for 32P-containing c-Src peptide by scintillation counting. The results represent the relative Csk kinase activity of uninfected control and HIV+ patient after subtracting the background control (immune-complex without lysate; 350 cpm). (B) Western immunoblotting of 50-kD Csk protein for (A). Antibody control consists of immune complex without lysate. These results are representative of three independent experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/9/10.1182_blood.v90.9.3603/4/m_bl_0043f6.jpeg?Expires=1763668683&Signature=kcLQxXEEzJTbxAIMBDEeb08MG6GbhzhmwoowGJIqJZKV8qyitNAxSyJMHDSztcKtFj8fRUdvXhmAg9yd-NF3lKNV71q6~xey-fIL107V5-IYjH-JbbOFD8IrIgqJHcQRu-Q9X8vCeH35QxOIm9NNYrBAEOEQB-8J--~yplx71Dy7lk3q-l7aGFbQKwts23oIIzuc0y1VHYdMJUf0GlXZuLTYEdF63rbgaCpZaolLbPeIAgTdHsOmYypi3p-ZTRHqwYTxVsvMstIz2ek58qsnxaRvycE3Mk9yG4a6zGyYACMNWre5-gdq5twoSUuzCZGI6sciKgFRCwhnLKp4fA28Kw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Fig. 2. Specificity of anti-Fyn (428). (A) Control Western immunoblot. Anti-Fyn (428) was used to immunoprecipitate resting PBMC (5 × 106 cell equivalents), the immunoprecipitated proteins fractionated by SDS-PAGE and Western immunoblotting used to check the specificity of the immunoprecipitating antibody to recognize src-family PTK. Total cell lysate was used to control for Fyn, Lck, c-Src, and c-Yes proteins. (BI) Control kinase assay. Lysate containing 25 × 106 cell equivalents of Fyn+ Jurkat cells (lane 2) or Fyn− C8 (lane 3) and S1T (lane 4) cell lines were immunoprecipitated with rabbit anti-Fyn and immune-complexes washed and incubated with 5 μCi [γ32P] in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography. Lane 1 is an immunoprecipitating antibody control that also serves as a control for background enolase phosphorylation. (BII) The Immobilon-P membrane in (BI) was blocked in 5% skim-milk, probed with a monoclonal anti-Fyn, followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The positions of the molecular weight standards are indicated and arrows show the location of fyn, enolase (E), and the immunoprecipitating antibody heavy chains [Ig(H)], respectively.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/9/10.1182_blood.v90.9.3603/4/m_bl_0043f2.jpeg?Expires=1763668684&Signature=NKTLE18oLirs9Ba1XjRSs3WKdHRAlkdLGpXILEHlt4aFCbcJOnCY9HrbDbpJBsQReY7KbpNwaxK2WYi9mlwxVdxq~Pla8Zmcf1b0XKEhgCOigMKV68Wih0sReY6xmdEamL12CwT8v4u~6xkyGpAgK9ErizBAZe65rAX7P5Yd851tAMarzqyVGhTDmdXj72QYKDq52k7tZ05KbjaoDPX~tFhUkcuinlCx5tYukSUBwrOV63Xz8jn1EV46S-I9TjDe5NeoBt0phe5pYIrmVUZf69fKhUFj0scJrlv8Rk7TDw3nhMs5hBSNptiNAuE0DcIus3xVR0Zc0l4r3-L~az2G4g__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Fig. 3. Levels of Fyn protein are low in asymptomatic HIV infection but relative specific kinase activity is elevated. Lysate containing 5 × 106 cell equivalents was immunoprecipitated with rabbit anti-Fyn and immune complexes washed and incubated with 5 μCi [γ32P]ATP in kinase buffer containing acid-treated enolase (1.0 μg/condition). Proteins were separated by reduced SDS-PAGE and electrotransferred to Immobilon-P membranes and tyrosine-phosphorylated proteins visualized by autoradiography (Kinase Assay). Western immunoblotting was performed by probing the kinase blot with mouse anti-Fyn monoclonal antibody followed by goat-antimouse IgG conjugated to horseradish peroxidase and enhanced chemiluminescence. The relative Fyn specific kinase activity was estimated using the ratio of Fyn kinase activity (using tyrosine phosphorylation of enolase) to Fyn protein levels after densitometry analyses of the autoradiograph (Kinase Assay) and chemiluminescence blots (Western blot), respectively. The figure is representative of the results obtained with all patients and normal controls. Lanes 1 and 2 are patients with asymptomatic HIV infection, lanes 3 and 4 represent patients having AIDS, and lanes 5 and 6 are uninfected normal controls.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/9/10.1182_blood.v90.9.3603/4/m_bl_0043f3.jpeg?Expires=1763668684&Signature=qYDdjPGXFyHsR1swFrVBf37I3HBpoCxX800rlgPPRqs5t1UmcantWhk~EalZAzRwy60xZSL16QWL~0h25dsW5U-mcFreq7Fh~ien9cXBoqdCNWAPDSaNfQcpF2FopsYpm236YHhx8u1KldsDQVRXLRr1MTiABo4ExmHzRDCvIGIsRb3jbbJkYbFF8gSC1QaKtfAqXMUPzIVnYJqWVMcKTmzvcXvwHLAv23nlouJKjmRLdcDjjcS4wV25iATMTfRLi6J3o4UU2JEUGOROw37IA1LeCHLtOGdmk89Z1JnFi3f~5EPR6hyBikqgvTN62CsKOwoqLYJqAqSOMcGqY4-SOw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

![Fig. 6. The kinase activity of Csk is decreased in HIV-infected patients. (A) Csk protein tyrosine kinase was immunoprecipitated from lysates containing equal total protein as measured by the bicinchoninic acid method, from uninfected individuals and from asymptomatic HIV+ patients. The immune complexes were washed, incubated with c-Src C-terminal peptide [Thr-Ser-Thr-Glu-Pro-Gln-Tyr-Gln-Pro-Gly-Glu-Asn-Leu] and 10 μCi [γ32P]ATP at 37°C for 15 minutes, centrifuged, and the supernatant blotted onto phosphocellulose paper and washed with 10% phosphoric acid. After drying, the blotted phosphocellulose paper was tested for 32P-containing c-Src peptide by scintillation counting. The results represent the relative Csk kinase activity of uninfected control and HIV+ patient after subtracting the background control (immune-complex without lysate; 350 cpm). (B) Western immunoblotting of 50-kD Csk protein for (A). Antibody control consists of immune complex without lysate. These results are representative of three independent experiments.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/90/9/10.1182_blood.v90.9.3603/4/m_bl_0043f6.jpeg?Expires=1763668684&Signature=lwGd-020~Th7TNdZFRNxTYPK6nPiFTyCE1p9xsm2JXw6NXf5TuuXVjVIJ7aiX-h6rLe3bRQWdevxhWHg~t5dQ28IzTbJYGgvCY6TSVNZTevzH7JQtbc~5WHCPxJP9winF0-QgzP8vl6GLgm6IiE3m5xZXe2rugsZKL-1nht6U-FTfup59MxCW4ejrIlkC7vp6rql33ZfJX4xXdGp~TVNo0Kdyr0JFkPG3VX1qxC4dB4wfP7JrhonLBalk4OneuC3jkMe1ZeXR4v0EFPnT1mNf~IW5TbniYrnTZljSbzUbxjIAeVC~bj290Wia6yrZDDcWjwbsv6SE9W8Bk8gY6Nalg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)