Abstract
Vitamin A metabolites are potent differentiation-inducing agents for myelomonocytic cell lines in vitro and are successfully used for the treatment of patients with acute promyelocytic leukemia. However, little is known about the effects of vitamin A on normal hematopoietic cells. Therefore, we investigated the effect of vitamin A on differentiation and activation of human blood monocytes (MO). Culturing MO for up to 4 days with 9-cis retinoic acid (RA) and all-trans RA but not retinol reduced MO survival, with the remaining cells being morphologically comparable to control cells. Because macrophage colony-stimulating factor (M-CSF) is a well-known survival factor for MO, we measured the M-CSF content of MO culture supernatants using enzyme-linked immunosorbent assay and found that RA suppressed the constitutive secretion of M-CSF. Northern analysis showed that the M-CSF mRNA expression was only slightly reduced by RA treatment, suggesting regulation on the posttranscriptional level. In contrast to MO, M-CSF secretion by MO-derived macrophages (MAC) was not altered by RA, suggesting a differentiation-dependent switch in the responsiveness of MO/MAC to RA. Because M-CSF is not only a survival-promoting but also a differentiation-promoting factor for myeloid cells, we analyzed the effect of RA on MO to MAC maturation. RA suppressed the expression of the maturation-associated antigen carboxypeptidase M (CPM)/MAX.1 at both the protein and mRNA levels and modulated the lipopolysaccharide-stimulated cytokine secretion of MO/MAC. The addition of exogenous M-CSF to RA-containing MO cultures fails to overcome the RA-induced inhibition of MO differentiation. However, the survival rate was improved by exogenous M-CSF. We conclude that RA acts via two different mechanisms on monocyte survival and differentiation: posttranscriptionally by controlling M-CSF secretion, which decreases MO survival, and transcriptionally regulating the expression of differentiation-associated genes. The regulation of M-CSF production may contribute to the antileukemic effect of RA in vivo by reducing autocrine M-CSF production by leukemic cells.
RETINOIC ACID (RA) exerts pleiotropic effects on cellular growth and differentiation. It induces the differentiation of promyelocytic HL-60 cells and primary human leukemia cells into granulocytes1,2 and induces complete remission in patients with acute promyelocytic leukemia.3 The biological effects of RA are mediated through intracellular RA receptors, which are members of the nuclear receptor superfamily, including receptors for steroid hormones, thyroid hormones, and retinoids. They function as ligand-inducible transcription factors and activate target genes after binding as homodimers or heterodimers.4,5 Suppression of the endogenous RA receptor in a multipotent hematopoietic cell line by a dominant negative RA receptor blocked neutrophil differentiation at the promyelocyte stage, suggesting a crucial role for RA/RA receptors in the terminal differentiation of neutrophils.6 Primary human bone marrow or embryonic fetal liver cells as hematopoietic precursors show a shift to granulocytic differentiation when cultured in the presence of RA.7,8 Conflicting reports have been published regarding the effects of RA on monocytic differentiation. In the promyelocytic cell line HL-60, RA inhibited monocytic differentiation,9whereas, in the monoblastic cell line U937, RA was shown to modulate macrophage differentiation either positive or negative.10,11 We were interested in the effects of RA on primary human blood monocytes (MO) and MO-derived macrophages (MAC). MO differentiation into MAC in vitro is a well-established model system for a similar differentiation process in vivo.12,13 When MO are cultured in the presence of serum, characteristic changes in the morphology, antigenic phenotype, and functional competence of MO occur. Maturation-associated antigens, such as carboxypeptidase M (CPM)/MAX.1, CD16, CD51, or CD71, can serve as differentiation markers to define successful maturation in primary cultures of human MO.14-18 The signals inducing MO to MAC differentiation in vitro and in vivo are not well characterized. However, macrophage colony-stimulating factor (M-CSF) is an important cofactor for the survival and differentiation of MO and hematopoietic precursor cells.19 20
We investigated whether RA would influence human MO to MAC maturation and found that RA inhibited the MO maturation process, followed by the expression of differentiation-associated markers such as CPM or CD71. In addition, the survival rate of MO was reduced, which could be explained by a reduction of the constitutive M-CSF production. Cell survival but not differentiation could be rescued by the addition of exogenous M-CSF, suggesting that RA acts not only by the suppression of endogenous M-CSF.
MATERIALS AND METHODS
Cell separation and culture.
Peripheral blood mononuclear cells (MNC) were obtained by leukapheresis of healthy donors, followed by density gradient centrifugation over Ficoll/Hypaque. MO were isolated from MNC by counter-current elutriation (J6M-E Beckmann centrifuge) using a large-volume chamber (50 mL), a JE-5 rotor at 2,500 rpm, and a flow rate of 110 mL/min in Hank's balanced salt solution supplemented with 6% autologous human plasma.17 Elutriated MO were greater than 90% pure as determined by morphology and expression of the MO antigen CD14. Purified MO were cultured on teflon foils (Biofolie 25; Heraeus, Hanau, Germany) for up to 7 days at a cell density of 106 cells/mL in RPMI 1640 (Biochrom, Berlin, Germany) supplemented with antibiotics (50 U/mL penicillin and 50 mg/mL streptomycin; GIBCO, Berlin, Germany), L-glutamine (2 mmol/L; GIBCO), and 2% pooled human AB-group serum with or without 9-cis-RA (kindly provided by Hoffmann-La Roche, Basel, Switzerland), all-trans RA, 13-cis-RA, or retinol (Sigma, St Louis, MO). Cultures containing RA were fed with half of the initial RA dose after 2 days. At the indicated time periods, cells were harvested and viable cells were counted by trypan blue exclusion. Supernatants of these cultures were also harvested, sterile filtered, and stored at −20°C for M-CSF determination.
Supernatants of MO/MAC.
Either freshly isolated MO or MAC cultured for 7 days on teflon foils were cultured at a cell density of 106 cells/mL on petri dishes for 48 hours plus serum with or without RA. Supernatants were harvested, sterile filtered, and stored at −20°C for M-CSF determination.
Detection of M-CSF, interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α).
Cytokines were measured by commercially available enzyme-linked immunosorbent assay (ELISA) kits (Biermann, Bad Nauheim, Germany).
Flow cytometry.
MAC were harvested on day 4 and washed twice with cold phosphate-buffered saline (PBS) containing 0.1% sodium azide and 0.6 mg/mL Ig. Cells (5 × 105) were incubated with saturating amounts of specific monoclonal antibodies or IgG isotype control for 30 minutes at 4°C. The following antibodies were used: CD11c, CD14, CD51, CD54, and CD71 from Immunotech (Hamburg, Germany) and CPM/MAX.1 from our own laboratory. After two further washes, cells were incubated with saturating concentrations of fluorescein isothiocyanate-conjugated goat antimouse IgG for 30 minutes at 4°C. After two more washes, cells were fixed with 1% paraformaldehyde in PBS. Analysis was performed using a FACScan (Becton Dickinson, Mountain View, CA). Cell populations were gated according to their forward and side scattering. The same instrumental setting (FL1 426) was used for MAC cultured with or without RA.
RNA extraction and Northern analysis.
MAC were harvested from the teflon foils and lysed with guanidine thiocyanate solution. RNA extraction was performed according to Chomczynski and Sacchi.21 Ten micrograms of total RNA was run in 1% agarose-formaldehyde gels and transferred to nylon membranes (NT membranes; MSI, Westborough, MA). M-CSF mRNA was detected by hybridization of the membranes with an oligonucleotide complementary to bp 275-311 of the published sequence.22This probe was labeled with T4 polynucleotide kinase and γ-(32P)-ATP (3,000 Ci/mmol; Amersham, Buckinghamshire, UK). Hybridization conditions were 500 mmol/L sodium phosphate, pH 7.2, 7% sodium dodecyl sulfate (SDS), 1 mmol/L EDTA, and 150 mg/mL tRNA at 56°C overnight. CPM mRNA was detected by hybridization with a32P-labeled cDNA probe (random primed labeling kit; Boehringer Mannheim, Germany) of the EcoRI restriction fragments of the cloned CPM polymerase chain reaction (PCR) fragment.23 To provide an internal control, membranes were reprobed with an oligonucleotide against 18 S rRNA labeled by T4 kinase.
RESULTS
Effect of RA on MO survival.
MO were cultured for up to 4 days on teflon foils with or without 10−7 mol/L 9-cis-RA, harvested, and counted. Cell numbers are given as the percentage of cells of the starting population. We found that, compared with cells cultured without RA, the survival rate of cells cultured with RA was reduced 25% ± 7% (mean ± SEM; n = 10) on day 2 and 36% ± 5% (mean ± SEM; n = 17) on day 4 (Fig 1). However, the morphology of the remaining cells resembled MAC from cultures free of RA.
Comparative analysis of MO survival, secreted M-CSF protein, and M-CSF mRNA level after RA treatment. MO were cultured for 2 days and 4 days, respectively, on teflon foils with or without 10−7 mol/L RA. Cells were harvested, counted, and lysed with guanidine thiocyanate solution. Northern analysis was performed as described in the Materials and Methods. Densitometric analysis of the signals was performed. M-CSF content of the corresponding supernatants was determined by ELISA. All values are given as the mean percentage of inhibition by RA compared with the serum control (results are significantly different: for survival, day 2 [P < .02] and day 4 [P < .0001]; for M-CSF protein, day 2 [P < .03] and day 4 [P < .009]; for M-CSF mRNA, day 4 [P < .05] by two-tailed t-test for paired data). Results are not significantly different for day 2 mRNA data.
Comparative analysis of MO survival, secreted M-CSF protein, and M-CSF mRNA level after RA treatment. MO were cultured for 2 days and 4 days, respectively, on teflon foils with or without 10−7 mol/L RA. Cells were harvested, counted, and lysed with guanidine thiocyanate solution. Northern analysis was performed as described in the Materials and Methods. Densitometric analysis of the signals was performed. M-CSF content of the corresponding supernatants was determined by ELISA. All values are given as the mean percentage of inhibition by RA compared with the serum control (results are significantly different: for survival, day 2 [P < .02] and day 4 [P < .0001]; for M-CSF protein, day 2 [P < .03] and day 4 [P < .009]; for M-CSF mRNA, day 4 [P < .05] by two-tailed t-test for paired data). Results are not significantly different for day 2 mRNA data.
Regulation of M-CSF secretion by RA.
Because M-CSF is a potent survival and differentiation factor for human MO, we analyzed whether a reduction of M-CSF protein in the corresponding culture supernatants would explain the decreased survival rate of MO/MAC cultured with RA. Therefore, we measured the M-CSF content in the supernatant after 2 days and after 4 days and found an average reduction of M-CSF in the culture supernatant of 49% ± 9% (mean ± SEM; n = 11) on day 2 and 61% ± 7% (mean ± SEM; n = 17) on day 4 as compared with the control (Fig 1). Five experiments in which survival, M-CSF protein, and M-CSF mRNA were analyzed in parallel are shown in Table 1. A decrease in MO/MAC survival was always paralleled by a decrease in the M-CSF level of the corresponding supernatant; however, the absolute M-CSF value varied between different donors and did not correlate with the survival rate of MO/MAC. This indicates that other mechanisms, eg, at the M-CSF receptor level, may influence MO/MAC survival.
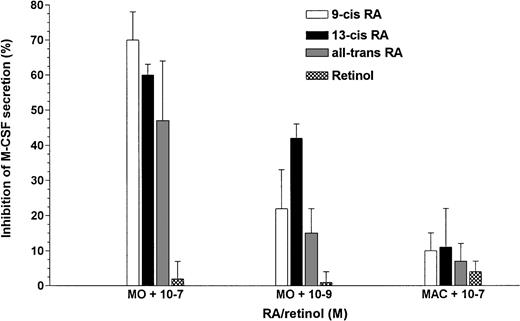
Because adherence is a strong inducer of M-CSF mRNA expression and protein synthesis, we were interested as to whether this downregulation would also take place on plastic surfaces, because teflon is only a weak adherence substrate for MO/MAC. Freshly isolated MO were cultured for 48 hours on petri dishes with or without different RA isomers. Control MO were incubated with retinol, the inactive precursor of RA. As shown in Fig 2, at a concentration of 10−7 mol/L, all RA metabolites could reduce M-CSF protein secretion, even on plastic surfaces, whereas retinol had no effect. RA at a concentration of 10−9 mol/L had a slight effect on the reduction of M-CSF protein in the supernatant; however, no effect was found on the survival rate or antigen expression (data not shown). Interestingly, M-CSF production of MO-derived MAC was not inhibited by RA, indicating a differentiation-dependent switch in the responsiveness of MO/MAC to RA. Addition of exogenous M-CSF to RA-containing cultures resulted in a cell survival rate comparable to that of the controls (data not shown).
M-CSF secretion is decreased by different RA metabolites in MO but not in MO-derived MAC. Either MO or MO-derived MAC were cultured for 48 hours with or without serum plus 10−7mol/L 9-cis-RA, all-trans-RA, 13-cis-RA, or retinol on petri dishes. The M-CSF content of these supernatants was analyzed by ELISA.
M-CSF secretion is decreased by different RA metabolites in MO but not in MO-derived MAC. Either MO or MO-derived MAC were cultured for 48 hours with or without serum plus 10−7mol/L 9-cis-RA, all-trans-RA, 13-cis-RA, or retinol on petri dishes. The M-CSF content of these supernatants was analyzed by ELISA.
Effect of RA on M-CSF mRNA expression.
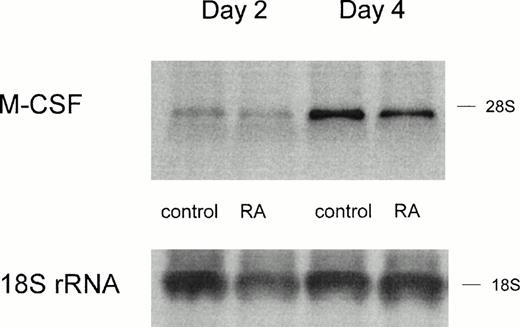
RA is known to regulate gene expression via binding to its intracellular receptors acting as transcription factors. Therefore, we investigated whether the inhibition of M-CSF production would correspond to a reduced expression of M-CSF mRNA. RNA was isolated from MO cultured for 2 days and 4 days with serum plus RA and analyzed by Northern analysis. Freshly isolated MO showed no expression of the M-CSF mRNA (data not shown), but after 2 and 4 days of adherence culture, a signal could be detected. RA did not reduce the expression of M-CSF mRNA in day-2 cultures, indicating a posttranscriptional regulation of M-CSF production (Fig 3); however, on day 4, the mRNA was slightly reduced by 26% (Fig 3 and Table 1, experiment no.1). In 4 of 8 experiments, we could detect a decrease of the M-CSF mRNA on day 4 (control v RA, different with P < .05); however, only 1 of 8 experiments showed a slight reduction of M-CSF mRNA on day 2 (no significant difference).
M-CSF expression on mRNA level is only slightly altered by RA treatment. RNA was isolated from MO cultured for 2 days or 4 days on teflon foils with or without 10−7 mol/L RA. Northern analysis was performed according to the Materials and Methods. Hybridization with an 18S rRNA oligonucleotide is shown as a loading control.
M-CSF expression on mRNA level is only slightly altered by RA treatment. RNA was isolated from MO cultured for 2 days or 4 days on teflon foils with or without 10−7 mol/L RA. Northern analysis was performed according to the Materials and Methods. Hybridization with an 18S rRNA oligonucleotide is shown as a loading control.
Maturation-associated antigen expression is altered by RA.
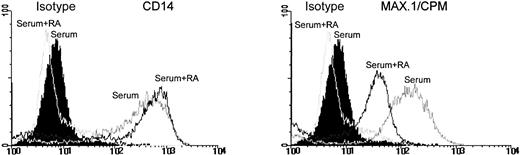
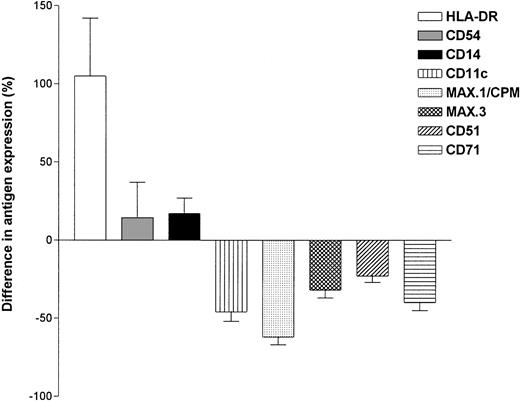
M-CSF is not only a survival but also a differentiation promoting factor for myeloid cells. Therefore, we analyzed whether RA would also modulate MO differentiation. MO were cultured for 4 days on teflon foils with or without 10−7 mol/L 9-cis-RA and the expression of maturation-associated antigens was followed by flow cytometry. Figure 4 shows a comparison of the antigen expression of MO-derived MAC cultured with or without RA regarding the expression of MAX.1/CPM and CD14. The expression of CPM, which is normally nearly undetectable on freshly isolated MO and upregulated during the differentiation of MO to MAC, remained low in RA-containing cultures, whereas the expression of CD14 was comparable to control cultures. Expression of other differentiation-dependent antigens such as CD51 and CD71 were also suppressed by RA. In contrast, HLA-DR expression was upregulated by RA (Fig 5). Because RA had no effect on the M-CSF mRNA level, despite a suppression of the protein level, we investigated whether this is also true for CPM expression. Similar to the protein level, CPM mRNA level declined in a dose-dependent manner after RA incubation (Fig 6).
RA suppresses CPM expression but not CD14 expression in MAC. MO were cultured on teflon foils in serum with or without 10−7 mol/L 9-cis-RA. After 4 days, cells were harvested and stained according to the Materials and Methods for flow cytometry.
RA suppresses CPM expression but not CD14 expression in MAC. MO were cultured on teflon foils in serum with or without 10−7 mol/L 9-cis-RA. After 4 days, cells were harvested and stained according to the Materials and Methods for flow cytometry.
Effect of RA on antigen expression analyzed by flow cytometry. MO were cultured on teflon foils with or without 10−7 mol/L 9-cis-RA. After 4 days, cells were harvested and stained according to the Materials and Methods. Data represent the difference in fluorescence intensity between cultures with or without RA (mean ± SEM for at least 3 experiments with different donors).
Effect of RA on antigen expression analyzed by flow cytometry. MO were cultured on teflon foils with or without 10−7 mol/L 9-cis-RA. After 4 days, cells were harvested and stained according to the Materials and Methods. Data represent the difference in fluorescence intensity between cultures with or without RA (mean ± SEM for at least 3 experiments with different donors).
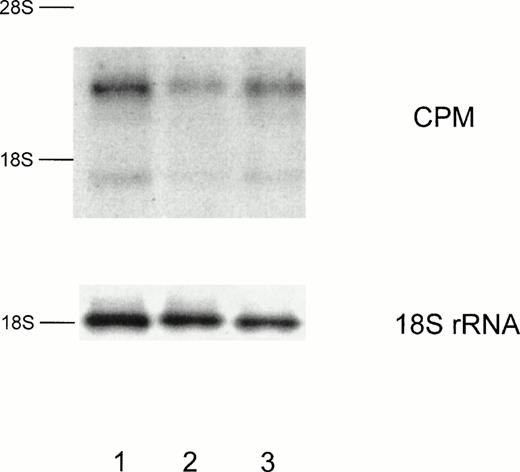
CPM expression is also suppressed on mRNA level by RA. Total RNA was isolated either from MO cultured on teflon foils with 10−7 mol/L 9-cis-RA (lane 2) or 10−9 mol/L 9-cis-RA (lane 3) or without RA (lane 1). After 4 days cells were harvested and Northern analysis was performed according to the Materials and Methods. Hybridization with an 18S rRNA oligonucleotide is shown as a loading control.
CPM expression is also suppressed on mRNA level by RA. Total RNA was isolated either from MO cultured on teflon foils with 10−7 mol/L 9-cis-RA (lane 2) or 10−9 mol/L 9-cis-RA (lane 3) or without RA (lane 1). After 4 days cells were harvested and Northern analysis was performed according to the Materials and Methods. Hybridization with an 18S rRNA oligonucleotide is shown as a loading control.
Cytokine secretion is modulated by RA.
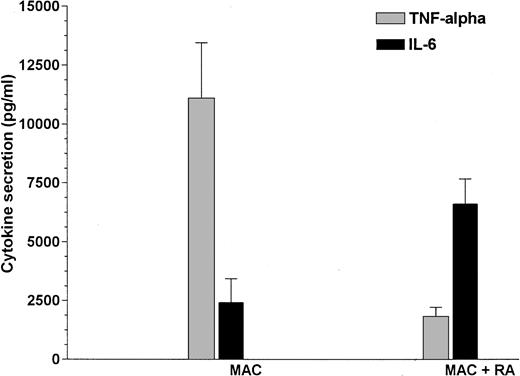
Besides the morphology and antigen-expression, the functional activity of MO changes during the differentiation into MAC. Especially the lipopolysaccharide (LPS)-induced cytokine production can serve as a maturation marker for MO-derived MAC. We therefore analyzed whether MO cultured with or without RA would differ in their ability to produce IL-6 and TNF-α after LPS stimulation. Control MAC cultured in the presence of serum showed a normal MAC LPS response, ie, a high secretion of TNF-α and a low secretion of IL-6. However, MAC derived from RA-containing cultures showed an inverse pattern of cytokine secretion after LPS stimulation (Fig 7) that is a typical feature of freshly isolated MO, indicating that MO do not differentiate into functionally normal MAC in the presence of RA.
MAC cytokine pattern is modulated by RA. MO were cultured on teflon foils with or without 10−7 mol/L 9-cis-RA. After 4 days, cells were harvested, seeded in a 6-well plate at a density of 5 × 105/mL, and stimulated for another 24 hours with 100 ng/mL LPS. Cytokines were determined by ELISA.
MAC cytokine pattern is modulated by RA. MO were cultured on teflon foils with or without 10−7 mol/L 9-cis-RA. After 4 days, cells were harvested, seeded in a 6-well plate at a density of 5 × 105/mL, and stimulated for another 24 hours with 100 ng/mL LPS. Cytokines were determined by ELISA.
DISCUSSION
RA is a well-known inducer of the differentiation of promyelocytic cell lines1 and primary leukemic cells in vitro.2,24However, little is known about the effect of RA on the differentiation and activation of normal human hematopoietic cells. For the first part of MO differentiation, the development of MO from hematopoietic precursor cells, RA has been shown to shift the differentiation to the granulocytic pathway.7,8 In this study, we were interested whether RA modulates the next step of MO maturation, the differentiation of normal human blood MO into MAC. To analyze the effects of RA on MO/MAC maturation, MO were cultured with serum as the usual inducer of MO differentiation in vitro with or without RA. Morphology of MAC was not influenced by RA treatment, but the functional activity and the expression of maturation-associated antigens, such as MAX.1/CPM, CD51, and CD71, were suppressed. Accordingly, Northern analysis showed a reduction of the CPM mRNA level. The effect of RA on MO/MAC differentiation has also been studied by others, including the differentiation of the human monoblastic cell line U937 and the differentiation of osteoclasts in chicken.11,25 Both studies found an antagonistic effect of 1,25-dihydroxyvitamin D3[1,25(OH)2D3] and RA on the differentiation process, with RA being dominant over 1,25(OH)2D3. 1,25(OH)2D3, which is a potent inducer of MO differentiation,26,27 could also not overcome the differentiation block by RA in our system (data not shown). The functional antagonism of RA and 1,25(OH)2D3 on MAC differentiation may be explained by an interaction of RA receptors (RAR and RXR) and 1,25(OH)2D3 receptors (VDR). Heterodimers of these receptors can act as transcription factors regulating the expression of different genes.28 The regulation is dependent on the receptor level and the ligand concentration. In vitro, it has been shown that an excess of 9-cis-RA, a ligand for the RAR and the RXR receptor, destabilizes the VDR/RXR heterodimer, leading to an inhibition of 1,25(OH)2D3-activated gene expression.29,30 In our system, not only 9-cis RA but also all-trans RA, which binds to another RA receptor, RAR, suppressed maturation, indicating additional mechanisms of action. Cao et al31 reported that VDR/RXR and RAR/RXR heterodimers can compete for a shared half site in the response element for the integrin αVβ3/CD51 (which was also suppressed during MO differentiation in the presence of RA). Co-addition of both ligands, 1,25(OH)2D3 and RA, completely inhibited the transactivational effect of 1,25(OH)2D3.31 But RA receptors do not only interact with just VDR but also with other transcription factors such as AP-1. The expression of the AP-1–responsive gene collagenase, which is also a maturation marker for MO/MAC, is downmodulated by the RA receptor RAR.32Therefore, we suggest that the interactions of RA receptors with other transcription factors that are involved in the regulation of differentiation may lead to a differentiation block in MO/MAC.
Besides the changes in functional activity and antigen expression, the addition of RA to MO cultures leads to a decrease in the survival rate of MO, which could be explained by a reduction of the M-CSF content in RA-containing cultures because M-CSF is a well-known survival factor for MO.19,20 Accordingly, survival was comparable to control cultures after supplementation of RA-containing cultures with exogenous M-CSF. In contrast to our findings, Nakajima et al33 reported an increase in the M-CSF production in primary cultures of human bone marrow stromal cells and in a human bone marrow stromal cell line cultured with RA.33 Similar results were found by Hamilton et al34 with synovial fibroblasts. This discrepancy may be due to the fact that different cell types were studied. In MAC, which are also a part of the bone marrow stroma, we were not able to find a downregulation of M-CSF production. The differences in the responsiveness of cells to RA can be explained by differences in the receptor level or concentrations of cellular RA binding proteins (CRABP) in the target cell. Preliminary results in our laboratory suggest that the unresponsiveness of MAC could be due to a high expression of CRABP II, which can inhibit RA action (data not shown). Freshly isolated MO showed no expression of the M-CSF mRNA on the mRNA level (and no protein was detected in the corresponding supernatant), but after 24 to 48 hours, a strong signal could be detected that was similar with or without RA, ie, the reduction in M-CSF protein levels was not paralleled by a decrease in the mRNA level. However, after 96 hours, a slight reduction was also found on mRNA level. Accordingly, Haskill et al35 found an induction of M-CSF mRNA by adherence without further stimulation. Regarding the regulation of the M-CSF mRNA, similar results have been published by Lee et al,36 who investigated the regulation of M-CSF by prostaglandin E2(PGE2) and found that PGE2 not only inhibited M-CSF protein levels but even upregulated the mRNA. Therefore, M-CSF seems, at least in part, to be regulated by posttranscriptional mechanisms.
RA is used in the treatment of acute promyelocytic leukemia,3 myelodysplastic syndrome,37 and juvenile chronic myelogenous leukemia.38 Elevated circulating levels of M-CSF are found in acute myeloid leukemia (AML) and other leukemias,39,40 ovarian adenocarcinoma,41 and myeloproliferative disease.42 In addition, the receptor for M-CSF, cfms, is expressed in AML and other malignancies.41 43 If M-CSF plays a role as an autocrine or paracrine growth factor in these diseases, the downregulation of M-CSF may be one explanation for the therapeutic effects of RA.
Supported by DFG.
Address reprint requests to Marina Kreutz, PhD, Department of Hematology and Oncology, University of Regensburg, Franz-Josef-Strauss-Allee 11, 93042 Regensburg, Germany; e-mail:Marina.Kreutz@klinik.uni-regensburg.de.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" is accordance with 18 U.S.C. section 1734 solely to indicate this fact.

![Fig. 1. Comparative analysis of MO survival, secreted M-CSF protein, and M-CSF mRNA level after RA treatment. MO were cultured for 2 days and 4 days, respectively, on teflon foils with or without 10−7 mol/L RA. Cells were harvested, counted, and lysed with guanidine thiocyanate solution. Northern analysis was performed as described in the Materials and Methods. Densitometric analysis of the signals was performed. M-CSF content of the corresponding supernatants was determined by ELISA. All values are given as the mean percentage of inhibition by RA compared with the serum control (results are significantly different: for survival, day 2 [P < .02] and day 4 [P < .0001]; for M-CSF protein, day 2 [P < .03] and day 4 [P < .009]; for M-CSF mRNA, day 4 [P < .05] by two-tailed t-test for paired data). Results are not significantly different for day 2 mRNA data.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/91/12/10.1182_blood.v91.12.4796/4/m_blod41239001x.jpeg?Expires=1769083521&Signature=OFFSOhOC~1tVEl4EjBufEjFl8ocQlJKNXqxNcH0waAY~ekE-PU-~c1qB2KqWVhgTa7M~jQ9oTcinetWiwnvZfgjN0j3fXjajJHc4Ermv5hmNUxy5gjTIvNu1B-EOHgZoDYp5IWeS06b-zmeysgzSiuryE4uJLoqA9E0-hW4yU78BMHFACRYRq1Ig~2svLD4FLBTdemWH5CfnhCD-400BUF8anlk3T1eUcQMZBHYMHN~Oc0owyI361Z8PI3NlcLRlzABeU9LQpgLWMhGllk74R2tZorpG39M8bjbR6ZXP4~greIq8IhvzZuUfu6~ZHT-tQHykn8ydN-RJ3nzB0BcaXg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)