Abstract
Renal ossicles are ossified structures developed after the implantation of a bone marrow (BM) plug beneath the kidney capsule. The authors have investigated the origin of the hematopoietic cells in murine renal ossicles by conducting sex-mismatched implants into Ly-5 congenic mice. BM plugs from transgenic mice provided additional genotypic tracers. Flow cytometry analyses on nonadherent cells from long-term cultures established with ossicles excised at 17 to 40 weeks postimplantation evidenced the presence of 5% to 70% of donor-derived myeloid cells. The genetic analysis of the day 12 colony-forming unit (CFU-S12) population in ossicles excised at 10 to 40 weeks postimplantation revealed that 16% to 93% of the colonies were of donor origin. Moreover, we describe for the first time the presence of long-term repopulating cells of donor origin in ossicles excised at 10 to 19 weeks postimplantation.
Introduction
The implantation of bone marrow (BM) plugs beneath the murine renal capsule results in the generation of ossicles with active hematopoiesis.1-3 The origin of the hematopoietic and stromal cells in renal ossicles and in subcutaneously implanted BM plugs and femurs has been investigated by means of chromosomal markers2,4-8 and immunological techniques.8-10These studies evidenced the donor origin of the stromal cells in these transplantation models.8-10 On the other hand, the pluripotent progenitors and differentiated cells lodged in these ectopic hematopoietic foci have been shown to be predominantly of host origin.2,7-9 In some instances, donor hematopoietic cells have been detected longer than 6 months postimplantation,4,7,8 with an increasing ratio of donor versus host-derived contribution as the analysis period was shortened.8
We have further investigated the nature of the hematopoietic progenitors and repopulating cells (RCs) present in renal ossicles by establishing long-term cultures (LTCs) and conducting spleen colony-forming unit (CFU-S) and repopulating assays. The origin of these precursors was unequivocally identified by the use of several genetic (zfy-1 male and neomycin phosphotransferase [neor] gene sequences) and phenotypic (panleukocyte Ly-5 antigen) markers.
Study design
Mice
We used 12- to 15-week-old C57Bl/6 (Ly-5.2) and C57Bl/6-Ly 5.1-Pep3b (Ly-5.1) mice. Silent neorgene sequences of 624 base pairs (bp) and 1200 bp were used to generate the N1 and N2 transgenic Ly-5.2 mouse lines (Ly-5.2/neo) according to standard procedures.11
Long-term hematopoietic cultures
Long-term BM cultures12 were initiated by flushing the marrow of one hind limb into a 25-cm2 culture flask (Nunc, Roskilde, Denmark) with 10 mL Fischer's medium (GIBCO, Grand Island, NY) supplemented with 20% horse serum (Flow, ICN Biomedicals, Costa Mesa, CA) and 10−5mol/L hydrocortisone sodium succinate (Sigma, St Louis, MO). To establish LTCs from renal ossicles, a single bony ossicle was directly crushed inside the flask. Cultures were grown at 33°C with 5% CO2 in air, and half of the media was replaced weekly.
CFU-S and repopulating assays
Cell suspensions from ground ossicles were intravenously injected into x-ray–conditioned recipients receiving a 24-hour split dose of 9.5 Gy for the CFU-S assay13 and 10 Gy for the repopulating assay. Irradiation was delivered at 1.03 Gy/min, 300 kV, and 12.8 mA (Philips MG324 equipment, Hamburg, Germany) (half-value layer: 3.2 mm Cu).
Flow cytometry
Samples were analyzed on an Epics ELITE ESP flow cytometer (Coulter, Hialeah, FL) after erythrocyte lysis with ammonium chloride and exclusion of nonviable cells by propidium iodide. The monoclonal antibodies (PharMingen, San Diego, CA) used included the phycoerythrin-coupled anti-CD45.1 (anti–Ly-5.1) and the fluorescein-conjugated anti–Ly-6G (anti–Gr-1).
Results and discussion
Experimental model
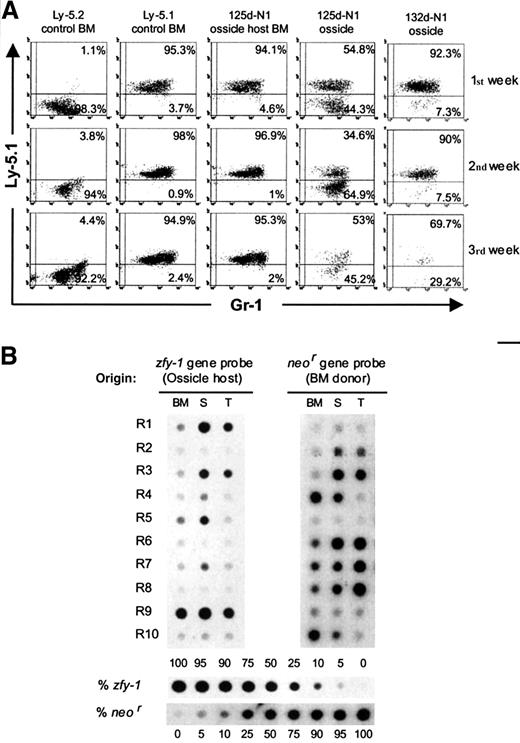
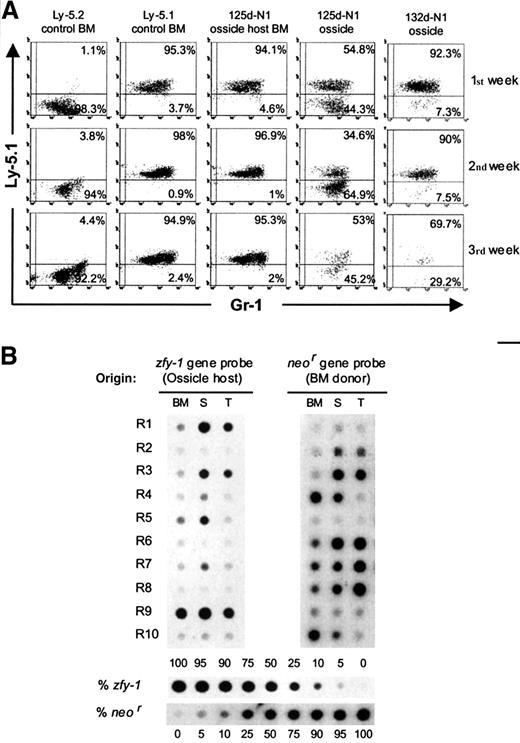
BM plugs from 2 femurs of transgenic Ly-5.2 female mice (F/Ly-5.2/neo) were implanted beneath the renal capsule of normal congenic male mice (M/Ly-5.1). The resulting ossicles were excised at periods longer than 10 weeks postimplantation. Each renal ossicle is coded with a number indicating the age (days postimplantation) followed by the transgenic strain (N1 or N2) used as BM donor. The origin of the hematopoietic progenitors lodged in renal ossicles was first investigated by anti–Ly-5.1 staining on supernatant cells from LTCs. These almost exclusively include myeloid cells (Gr-1+) in which the detection of Ly-5.1+ cells should result from ossicle-host–derived progenitors. On the other hand, the presence of Ly-5.1− cells in the supernatants should result from donor-derived progenitors. The origin of the CFU-S12 population and RCs was also investigated by transplanting ossicle marrow cells into irradiated female recipients (F/Ly-5.2). The spleen colonies and lymphohematopoietic organs from the reconstituted recipients were processed for DNA extraction and hybridization analysis.14 Cells derived from BM donors should give rise to DNA samples hybridizing with a probe for theneor gene but not with a male probe (zfy-1 gene probe). On the contrary, ossicle-host–derived cells should hybridize with the zfy-1 gene probe but not with the neor gene probe. Residual endogenous hematopoietic cells in the transplanted recipient should not contribute DNA hybridizing with either the neor or thezfy-1 gene probes.
Origin of nonadherent cells in LTCs established with renal ossicles
We used 125d-N1 and 132d-N1 ossicles to establish LTCs. LTCs were also initiated with BM from the 125d-N1 ossicle host and from Ly-5.1 and Ly-5.2 control mice (Figure 1A). As expected, most nonadherent myeloid cells from the Ly-5.1 and Ly-5.2 control cultures were Ly-5.1+ and Ly-5.1−, respectively. No significant numbers of Ly-5.1− cells (phenotype of the implanted BM) were detected in the LTC supernatants from the 125d-N1 ossicle host BM. However, ossicle-derived LTC supernatants always contained a significant proportion of myeloid cells from BM donor (7.3% to 64.9% of Ly-5.1− cells).
Analysis of the origin of ossicle-derived progenitors giving rise to LTCs and hematopoietic repopulation of transplanted mice.
(A) Cytometry profiles of nonadherent cells from LTCs established with renal ossicles and BM. Each week, supernatant cells were collected and stained with fluorescein-conjugated anti–Gr-1 and phycoerythrin-coupled anti–Ly-5.1. Control LTCs were established with BM from normal Ly-5.2 and Ly-5.1 mice. Test LTCs were established with 125d-N1 and 132d-N1 ossicles as well as with the 125d-N1 ossicle host BM. (B) Origin of the in vivo repopulating cells present in renal ossicles. A representative experiment in which 10 myeloablated Ly-5.2 female mice were intravenously transplanted with 2.5 × 105 ossicle marrow cells from an ossicle excised at 77 days postimplantation (77d-N2) is shown. The origin of the repopulating cells was determined by dot-blot hybridization analysis on DNA extracted from their lymphohematopoietic organs (bone marrow, BM; spleen, S; thymus, T) by means of the neor andzfy-1 gene probes. Recipient mice were killed at 92 days (R1), 120 days (R2-R3), and 195 days posttransplantation (R4-R10). Different proportions of normal male/N1 transgenic female splenic DNA were used as internal standards.
Analysis of the origin of ossicle-derived progenitors giving rise to LTCs and hematopoietic repopulation of transplanted mice.
(A) Cytometry profiles of nonadherent cells from LTCs established with renal ossicles and BM. Each week, supernatant cells were collected and stained with fluorescein-conjugated anti–Gr-1 and phycoerythrin-coupled anti–Ly-5.1. Control LTCs were established with BM from normal Ly-5.2 and Ly-5.1 mice. Test LTCs were established with 125d-N1 and 132d-N1 ossicles as well as with the 125d-N1 ossicle host BM. (B) Origin of the in vivo repopulating cells present in renal ossicles. A representative experiment in which 10 myeloablated Ly-5.2 female mice were intravenously transplanted with 2.5 × 105 ossicle marrow cells from an ossicle excised at 77 days postimplantation (77d-N2) is shown. The origin of the repopulating cells was determined by dot-blot hybridization analysis on DNA extracted from their lymphohematopoietic organs (bone marrow, BM; spleen, S; thymus, T) by means of the neor andzfy-1 gene probes. Recipient mice were killed at 92 days (R1), 120 days (R2-R3), and 195 days posttransplantation (R4-R10). Different proportions of normal male/N1 transgenic female splenic DNA were used as internal standards.
We analyzed 3 further ossicle-derived LTCs (198d-N1, 281d-N2, and 282d-N2 ossicles). In all instances, chimeric supernatants bearing 5% to 33% of Ly-5.1− cells were observed. As referred to above, fewer than 5% of Ly-5.1− cells were detected in the supernatants of the LTCs established with 198d-N1 and 281d-N2 ossicle host BMs (not shown). These results revealed the presence of BM-donor–derived progenitors in renal ossicles even at periods longer than 9 months postimplantation.
Orgin of the CFU-S12 population in renal ossicles
Hybridization analyses on CFU-S12 from 77d-N2, 133d-N1, 136d-N1, and 281d-N2 ossicles are summarized in Table1. This table also shows the proportion of hematopoietic cells from BM-donor and ossicle-host origin in the ossicle marrow as deduced by Ly-5.1 staining. Only 3 of 144 spleen colonies analyzed were of endogenous origin. Between 16% and 93% of the exogenous CFU-S12 in the ossicle marrow were of donor origin. Interestingly, even when the ossicle marrow cells were predominantly of host origin, a large proportion of donor-derived CFU-S12 was detected. We presume that the intense vascularization of the ossicles may account for this observation.1 Taken together, these data are consistent with studies detecting donor-derived CFU-S9 in subcutaneous femurs at 2 to 40 weeks postimplantation.5 8
Origin of the RCs in renal ossicles
Marrow cells from the 77d-N2 ossicle were transplanted into myeloablated F/Ly-5.2 mice. The origin of the RCs in the lymphohematopoietic organs from these recipients was investigated by means of hybridization analyses performed at the third (R1), fourth (R2-R3), and sixth month (R4-R10) posttransplantation (Figure 1B). Although the exogenous RCs in some recipients were predominantly from ossicle hosts (zfy-1+, ie, R1, R5, and R9), the remaining recipients were predominantly repopulated by BM-donor–derived cells (neo+). Similar results were obtained when marrow grafts from 78d-N1 and 133d-N1 ossicles were transplanted and recipients analyzed at 3 to 7 months posttransplantation (not shown).
Long-term RCs (LTRCs) had been previously reported in ossicles generated following the subcutaneous implantation of gelatin capsules containing recombinant human bone morphogenetic protein-2.18 Our study, however, reports for the first time the presence of LTRCs in mouse renal ossicles. Moreover, we demonstrate a significant repopulating contribution from donor origin. Given that we could not detect donor-derived progenitors in any ossicle host BM, we propose that donor RCs have been preserved at the implantation site. Nevertheless, we cannot rule out the possibility that donor RCs reached these organoids as a secondary process after the engraftment and subsequent mobilization from the host BM.
Our observations suggest the relevance of testing the origin of the hematopoietic populations in experimental models of heterotopic BM transplantation aiming to dissect the contribution of donor precursors to particular hematopoietic deficiencies.3 19 In addition, our data open new possibilities regarding the stable transplantation of donor hematopoietic stem cells in nonconditioned recipients.
Acknowledgments
The authors thank Thomas Graf for discussion on the manuscript, Antonio Bernad for collaboration to establish the N1 transgenic mouse line, Sergio Garcı́a for technical assistance, and J. Martı́nez for careful maintenance of the animals.
Departamento de Biologı́a Molecular y Celular y Terapia Génica, Centro de Investigaciones Energéticas, Medioambientales y Tecnológicas (CIEMAT), Madrid, Spain.
Submitted December 1, 1999; accepted May 25, 2000.
Supported by grant SAF 98-0008-C04-01 from the Comisión Interministerial de Ciencia y Tecnologı́a.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Florencio Varas, Centro de Investigaciones Energéticas, Medioambientales y Tecnológicas (CIEMAT), Departamento de Biologı́a Molecular y Celular y Terapia Génica, Edificio 7, Avenida Complutense 22, 28040-Madrid, Spain; e-mail: florencio.varas@ciemat.es.