Abstract
Strong reactivity for interferon-inducible protein 10 (IP-10), monokine induced by interferon gamma (Mig), and interferon-inducible T-cell alpha chemoattractant (I-TAC) was found in epithelial cells mainly localized to the medulla of postnatal human thymus. The CXC chemokine receptor common to the 3 chemokines (CXCR3) was also preferentially expressed in medullary areas of the same thymuses and appeared to be a property of 4 distinct populations: CD3+T-cell receptor (TCR) αβ+CD8+ single-positive (SP) T cells, TCRγδ+ T cells, natural killer (NK)–type cells, and a small subset of CD3+(low)CD4+CD8+TCRαβ+double-positive (DP) T cells. IP-10, Mig, and I-TAC showed chemoattractant activity for TCRαβ+CD8+ SP T cells, TCRγδ+ T cells, and NK-type cells, suggesting their role in the migration of different subsets of mature thymocytes during human thymus lymphopoiesis.
Introduction
Developing thymocytes pass through a complex differentiative program that results at the end in selection for mature T cells with receptors (TCRs) that recognize antigenic peptides bound to self–major histocompatability complex (MHC) molecules. The early thymic precursor cells are pluripotent in that they can give rise to both TCRαβ+ and TCRγδ+ T cells, low numbers of natural killer (NK) cells, and dendritic cells (DCs).1 Further differentiation commits the majority of cells to the T-cell lineage, thus curtailing the production of NK and dendritic cells. Early during the intrathymic developmental process, pre-T cells that have succeeded in productive rearrangements at the TCRβ locus are rescued from apoptotic cell death and selected for further maturation.1 The expression of a functional TCRβ chain is associated with the acquisition of the CD4+CD8+ double-positive (DP) phenotype. DP T cells then undergo positive selection that occurs in the cortex and reflects recognition of MHC-peptide complexes expressed on cortical epithelial cells. Positively selected cells proceed toward the medulla and undergo negative selection, which is followed by the migration of the few surviving CD4+CD8− or CD4−CD8+ single-positive (SP) mature T cells to the circulation and their colonization into secondary lymphoid organs.1
Chemokines have been shown to control the migratory behavior of several cell types, including lymphocytes, and have, therefore, the potential to regulate differentiation-dependent thymocyte migration.2-5 Many chemokines, including the following, are indeed constitutively expressed in the thymus: stromal-derived factor 1 (SDF-1),6,7 thymus and activation-regulated chemokine (TARC),8 thymus-expressed chemokine (TECK),9,10 pulmonary and activation-regulated chemokine (PARC),11 interferon (IFN)–inducible protein 10 (IP-10),12-14 IFN-inducible T-cell alpha chemoattractant (I-TAC),15 macrophage-derived chemokine (MDC),16,17 EBI1-ligand chemokine (ELC),18and secondary lymphoid tissue chemokine (SLC).19 Moreover, several chemokine receptors are also expressed in the thymus: CXC chemokine receptor 3 (CXCR3),20,21CXCR4,7,22-27 CC chemokine receptor 3 (CCR3),28 CCR4,17,27 CCR5,7CCR7,22,27-29 CCR8,30 and CCR9.10 31-34
SDF-1 seems to attract prevalently both CD4−CD8− double-negative (DN) and CD4+CD8+ DP immature thymocytes, thus favoring their migration at premedullar stage (cortex) before and during positive selection.22,24,25,27 By contrast, ELC and SLC prevalently attract mature SP (CD4+CD8− or CD4−CD8+) thymocytes, which suggests their role in migration from the thymus to the circulation of cells that have already passed both positive and negative selection.22,27-29 TECK efficaciously attracts both DP and SP human thymocytes.10,27,31,32 Recently we17,35 and others27 have shown that MDC attracts a small population of CD3+CD4+CD8+(low) thymocytes, which corresponds to cells transitional between cortex and medulla and in early medullary stages.
In this study we examined the expression of IP-10, monokine induced by interferon gamma (Mig), and I-TAC as well as their receptor, CXCR3, in human thymus. IP-10, Mig, and I-TAC messenger RNA (mRNA) and protein were mainly localized to medullary epithelial cells. Mig was strongly expressed in Hassall corpuscles, whereas IP-10 was prevalently expressed by epithelial cells scattered in the medulla and subcapsular areas. CXCR3 in the same thymus was also prevalently expressed in both medullary and subcapsular cortical areas by at least 4 subsets of thymocytes, 3 of which were CD3+, whereas the other was CD3−. CXCR3-expressing CD3+ thymocytes comprised TCRαβ+CD8+ SP T cells, TCRγδ+ T cells, and a small subset of CD1a+CD4+CD8+ T cells, suggesting their nature of immature T cells, whereas CXCR3 expression by CD3− thymocytes was a property of cells showing natural killer (NK)–type phenotype (CD8−CD56+ or CD8+CD56+). The chemotactic activity of IP-10, Mig, and I-TAC on total or fractionated thymocyte cell suspensions was also assessed. Of note, all 3 chemokines caused the selective accumulation of TCRαβ+CD8+ SP T cells, TCRγδ+ T cells, and NK-type cells, which suggests that the chemokines may play a role in the migration of distinct subsets of thymocytes during lymphopoiesis in human thymus.
Materials and methods
Reagents
The following reagents were used in the study: unconjugated and fluorescein isothiocyanate (FITC)–, phycoerythrin (PE)–, allophycocyanin (APC)–, or peridin chlorophyll protein (PerCP)–conjugated anti-CD3 (SK7), -CD4 (SK3), -CD8 (SK1), -CD45RA (L48), -CD45RO (UCHL-1), -CD56 (NCAM16.2), -CD16 (B73.1), -TCRαβ (WT31), and -TCRγδ11F2 monoclonal antibodies (mAbs) (Becton Dickinson, Mountain View, CA); unconjugated or FITC-conjugated anti-CD3 (UCHT1), -CD94 (HP-3D9), and -CD83 (HB15e) mAbs (Pharmingen, San Diego, CA); anti-CD68 (EBM11) and -CD20 mAbs (Dako, Glostrup, Denmark); anti–pan-cytokeratin (CK) (C11) (Sigma Immunochemicals, Milan, Italy); anti-CXCR349801.111 mAbs and IP-10 and Mig human recombinant chemokines (R&D Systems, Minneapolis, MN); conjugated and unconjugated isotype-matched control antibodies (Abs) (Southern Biotechnology, Birmingham, AL); and antihuman IP-10, Mig, and I-TAC rabbit polyclonal Abs (pAbs) and I-TAC human recombinant chemokines (Peprotech, London, England).
Human thymus specimens
Normal postnatal thymus specimens were obtained from 16 children (ages, 5 days to 3 years) who underwent corrective cardiac surgery at the Apuano Pediatric Hospital in Massa Carrara, Italy. The procedures followed in the study were in accordance with the ethical standards of the Regional Committee on Human Experimentation.
Cytofluorimetric analysis of thymocyte suspensions
Thymic tissue fragments were gently passed through a stainless-steel mesh to obtain single-cell suspensions from which mononuclear cells (MNCs) were separated by centrifugation on Lymphoprep (Nycomed Pharma, Oslo, Norway). Thymic MNCs were resuspended in phosphate-buffered saline (PBS) containing 0.5% bovine serum albumin (BSA) and 0.02% sodium azide and then incubated with fluorochrome-conjugated specific- or isotype-control mAbs. Cell surface marker analysis was performed on a fluorescence-activated cell sorter (FACSCalibur cytofluorimeter, Becton Dickinson) as previously described.35 36
Depletion of TCRαβ+ thymocytes
Depletion of TCRαβ+ thymocytes was performed by high-gradient magnetic cell sorting as described elsewhere.36 Briefly, thymic MNC suspensions were incubated for 20 minutes with anti-TCRαβ mAb, extensively washed, and then incubated for 20 additional minutes with goat antimouse pAb conjugated to colloidal superparamagnetic microbeads according to the magnetic cell sorter system (MACS, Milteny Biotec GmbH, Bergisch Gladbach, Germany). After washing, cells were inserted on a CS+ column and separated by a VarioMACS magnet.
Cloning and sequencing of the IP-10, Mig, and I-TAC probes
mRNA was extracted from human epithelial cells stimulated with tumor necrosis factor-α and IFN-γ and reversed to first-strand complimentary DNA (cDNA) by oligo dT primer using an M-MLV reverse transcriptase Kit (Life Technologies, Milano, Italy). The following primers were used: IP-10: forward, 5′-TGATTTGCTGCCTTATCTTTCTGA-3′; reverse, 5′-CAGCCTCTGTGTGGTCCATCCTTG-3′; Mig: forward, 5′-CAGCAGATGTGAAGGAACTG-3′; reverse, 5′-GCATGATGAAATTCAACTGG-3′; and I-TAC; forward, 5′-GCTATAGCCTTGGCTGTGATAT-3′; reverse, 5′-CAGGGCCTATGCAAAGACA-3′. Amplification of the first-strand products was carried out in a Thermal cycler (Mastercycler gradient; Eppendorf, Hamburg, Germany). The samples were subjected to 35 cycles of amplification using 400 nM of each primer and 2.5 units Taq DNA polymerase (Olymed, Florence, Italy). The DNA fragments of 335–base pair (bp) IP-10, 88-bp I-TAC, and 362-bp Mig amplified by polymerase chain reaction (PCR) were subcloned in pGEM-T (Promega, Madison, WI) according to the manufacturer's instructions. Sequencing of the amplified product was performed by the dideoxynucleotide chain-termination method17 by using sulfur 35 (35S) adenosine 5′-triphosphate (dATP) and sequenase enzyme (USB, Cleveland, Ohio).
In situ hybridization
In situ hybridization was performed on frozen thymus sections by using sense or antisense IP-10, Mig, and I-TAC RNA probes as detailed elsewhere.35 Briefly, the plasmid containing the cDNA was subcloned in PGEM-4Z then linearized with HindIII andBamHI (IP-10 and I-TAC) and EcoRI andHindIII (Mig) restricted enzymes followed by phenol-chloroform extraction and ethanol precipitation. Thereafter sense and antisense RNA probes were synthesized using SP6 or T7 RNA polymerases (Riboprobe Gemini System, Promega) in the presence of 48.1 × 102 MBq/mM (1300 mCi/mM)35S alpha-thio-UTP (uridine 5′-triphosphate) (NEN DuPont, Paris, France). Frozen thymus sections were mounted onto gelatin-coated slides and fixed with 4% paraformaldehyde for 20 minutes at room temperature. Sections were subsequently treated with 0.2 N hydrochloride (HCl) for 20 minutes, 0.125 mg/mL pronase for 10 minutes, 0.1 mol/L glycine for 30 seconds, and 4% paraformaldehyde for 20 minutes. Then the sections were rinsed with PBS, acetylated, and dehydrated in increasing ethanol concentrations. We applied 30 μL hybridization solution comprising 40% formamide, 4 × SSC (side scatter criteria), 10 mmol/L dithiothreitol, 1 times Denhardt solution, 10% dextran sulfate, 0.1 mg/mL sheared herring sperm DNA, and 1 mg/mL yeast transfer RNA (tRNA) and containing 8 × 105 cpm of35S-labeled human IP-10, Mig, and I-TAC RNA antisense probes to each section and covered it with parafilm. Hybridization was carried out at 52°C for 16 hours. Removal of the nonspecifically bound probe by ribonuclease (RNase) digestion and autoradiography was performed as detailed elsewhere.17 36 Sections were subsequently counterstained with Mayer hematoxylin and mounted with Kaiser glycerol gelatin. An average of 10 sections were analyzed for each tissue sample. Negative controls consisted of hybridization to sense RNA probes.
Immunohistochemistry
Immunohistochemical staining was performed on 10-μm cryostat sections or cultured cells fixed in 4% paraformaldehyde for 20 minutes or in acetone for 10 minutes. Sections were subsequently exposed to 0.3% hydrogen peroxide–methanol solution to quench endogenous peroxidase activity. After a 30-minute preincubation with normal horse serum (Vectastain ABC kit; Vector Laboratories, DBA, Milan, Italy), sections were layered for 30 minutes with 10 μg/mL anti–IP-10, 10 μg/mL anti-Mig, 10 μg/mL anti–I-TAC, 1 μg/mL anti-CD3, 3 μg/mL anti-CD68, 10 μg/mL anti-CD83, or 2 μg/mL anti-CK mAbs. This was followed by biotinylated horse antimouse or pAbs and goat antirabbit immunoglobulin G (IgG) Ab, respectively, and the avidin-biotin-peroxidase complex (Vectastain ABC kit) as previously described.17,36 We used 3-amino-9-ethylcarbazole (AEC) (Sigma) as a peroxidase substrate. Finally, sections were counterstained with Gill hematoxylin and mounted with Kaiser glycerol gelatin. All incubations were performed at room temperature. As a negative control, the primary Ab was replaced with an isotype-matched Ab with irrelevant specificity. Double immunostaining was performed by using the avidin-biotin-peroxidase system with 2 different substrates as previously described.17 36 To identify 2 different proteins on the same specimen, AEC (red) and the Vector SG (bluish gray) substrates were used, respectively. After double immunostaining, sections were counterstained with Gill hematoxylin and mounted with Kaiser glycerol gelatin.
Chemotactic assay
The chemotactic assay was performed according to the technique previously described.17 Briefly, chemokines were added at 3 different concentrations (10, 100, 1000 nM) in Roswell Park Memorial Institute medium (RPMI 1640) containing 0.5% BSA to the lower well of a transwell chamber (6 wells with a 3-μm pore size) (Costar, Corning, NY). We resuspended 5 × 106 freshly isolated thymocytes or their fractions in the same buffer and loaded them into the upper well. Cell migration was allowed to occur for 4 hours at 37°C, and cells migrating to the lower chamber were harvested and counted by FACSCalibur for 30 seconds both directly and after staining with fluorescent antibodies. Each experiment was performed in triplicate at least 3 times unless otherwise indicated. Cells that migrated in the presence of medium alone served as a negative control.
Results
IP-10, Mig, and I-TAC mRNA and protein are expressed by epithelial cells in human thymus
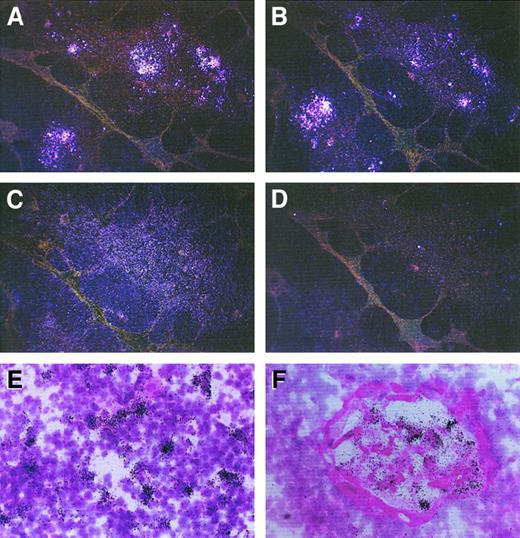
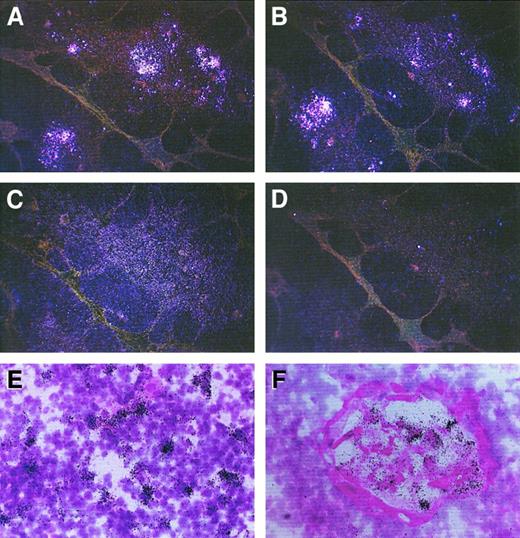
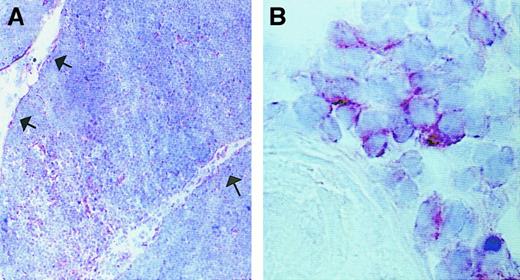
To investigate both the presence and the localization of IP-10–, Mig-, and I-TAC–producing cells in human thymus, 6 postnatal thymuses were assessed by in situ hybridization using sense and antisense probes on consecutive sections. There was a strong signal with both IP-10 and Mig antisense probes, which prevalently localized to the medullary areas, and rare IP-10+ cells were present even in the cortex (Figure 1A,B). A positive signal with I-TAC antisense probe was less intense and more spread (Figure1C), whereas there was virtually no signal with IP-10, Mig, or I-TAC sense RNA probes (Figure 1D). Of note, IP-10 mRNA was prevalently expressed by cells scattered in the medulla (Figure 1E), whereas Mig mRNA appeared to be mainly present at the level of the Hassall corpuscles (Figure 1F).
In situ hybridization showing expression of IP-10, Mig, and I-TAC mRNA in human thymus.
Consecutive thymic sections were hybridized with35S-labeled IP-10, Mig, or I-TAC antisense or sense probes. Positive signal with the IP-10 (A), Mig (B), and I-TAC (C) antisense probes (dark field, original magnification × 40) is visible mainly in the medullary areas, whereas virtually no signal is visible with the I-TAC sense probe (D) (dark field, original magnification × 40) as well as with the IP-10 or Mig sense probes (data not shown). The IP-10 signal is especially localized in single cells scattered in the medulla (E), whereas the Mig signal predominates at the level of Hassall corpuscles (F) (bright field, original magnification × 400).
In situ hybridization showing expression of IP-10, Mig, and I-TAC mRNA in human thymus.
Consecutive thymic sections were hybridized with35S-labeled IP-10, Mig, or I-TAC antisense or sense probes. Positive signal with the IP-10 (A), Mig (B), and I-TAC (C) antisense probes (dark field, original magnification × 40) is visible mainly in the medullary areas, whereas virtually no signal is visible with the I-TAC sense probe (D) (dark field, original magnification × 40) as well as with the IP-10 or Mig sense probes (data not shown). The IP-10 signal is especially localized in single cells scattered in the medulla (E), whereas the Mig signal predominates at the level of Hassall corpuscles (F) (bright field, original magnification × 400).
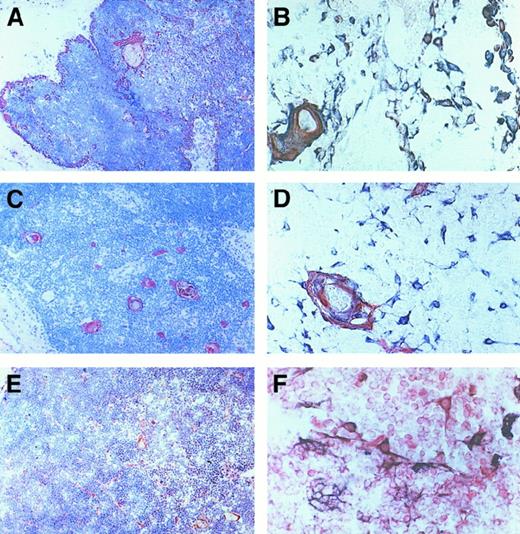
The expression of IP-10, Mig, and I-TAC protein in the same thymus was then analyzed by using immunohistochemistry. IP-10, Mig, and I-TAC expression showed a localization similar to that found by in situ hybridization (Figure 2A,C,E). The nature of IP-10–, Mig-, and I-TAC–expressing cells was then analyzed by using a double-immunostaining technique. Clear-cut separation in the staining between IP-10 and Mig from one side and CD3 (T cells), CD20 (B cells), CD68 (macrophages), and CD83 (dendritic cells) from the other side was observed. Separation in the staining between I-TAC and CD20, CD68, and CD83 was also observed, whereas several cells in both cortical and medullary areas costained for I-TAC and CD3 (data not shown). By contrast, all cells expressing IP-10 or Mig, as well as a proportion of I-TAC–expressing cells, showed costaining for CK, which provides a direct demonstration for the epithelial nature of most of these cells (Figure 2B,D,F).
Immunohistochemical analysis of IP-10, Mig, and I-TAC expression in human thymus.
Sections were immunostained with anti–IP-10, Mig or I-TAC (red) Ab, using the avidin-biotin-peroxidase method. IP-10 (A), Mig (C), and I-TAC (E) showed a localization similar to that found by in situ hybridization (Figure 1). Double immunostaining for CK (bluish gray) and IP-10 (B) or Mig (D) (red) reveals that the 2 chemokines are produced by thymic epithelial cells (purple brown), whereas I-TAC is constitutively produced not only by epithelial cells (purple brown), but also by several other thymocytes (F) (red). Original magnification × 400.
Immunohistochemical analysis of IP-10, Mig, and I-TAC expression in human thymus.
Sections were immunostained with anti–IP-10, Mig or I-TAC (red) Ab, using the avidin-biotin-peroxidase method. IP-10 (A), Mig (C), and I-TAC (E) showed a localization similar to that found by in situ hybridization (Figure 1). Double immunostaining for CK (bluish gray) and IP-10 (B) or Mig (D) (red) reveals that the 2 chemokines are produced by thymic epithelial cells (purple brown), whereas I-TAC is constitutively produced not only by epithelial cells (purple brown), but also by several other thymocytes (F) (red). Original magnification × 400.
CXCR3 localizes to both medullary and subcapsular areas and is expressed by different thymocyte populations
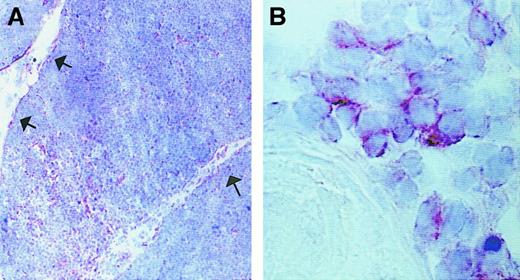
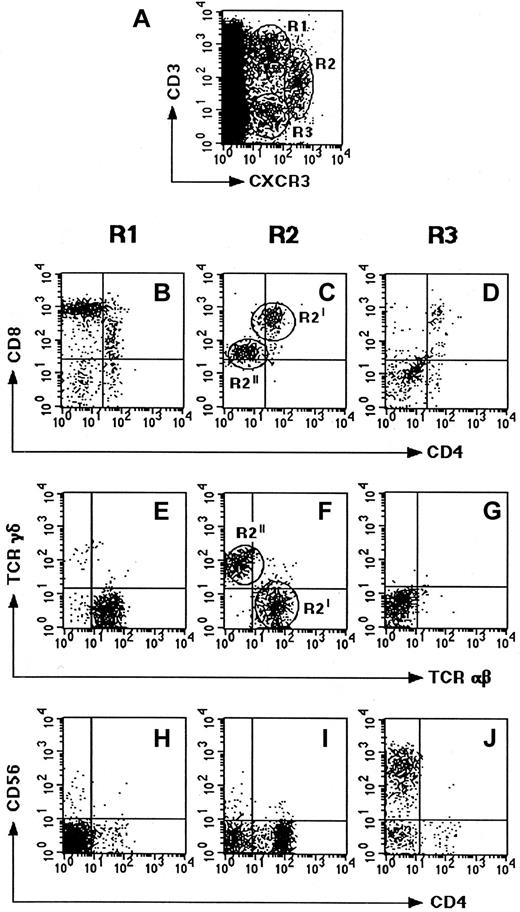
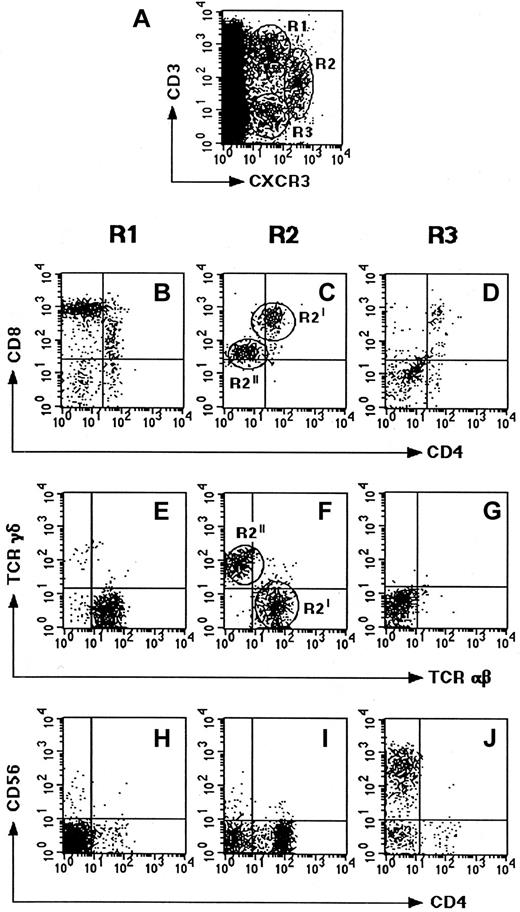
The localization and nature of cells expressing CXCR3, the receptor shared by IP-10, Mig, and I-TAC, was then examined. To this end, the expression of CXCR3 was assessed on 6 human thymuses by using both immunohistochemistry and flow cytometry. The immunohistochemical analysis showed that the majority of CXCR3+ cells localized to the medullary areas, but some of them were also found in the cortex and especially in the subcapsular areas (Figure3). The cytofluorimetric analysis revealed the existence of 3 small but clearly distinguishable populations of CXCR3+ cells, 2 of which (R1 and R2) also showed CD3 expression, whereas the third (R3) did not (Figure4A). When examined for their CD4 and CD8 expression, the most abundant CXCR3+CD3+population (R1) appeared to be composed of CD4−CD8+(high) T cells (Figure 4B), which were also TCRαβ+ (Figure 4E) and, in their majority, CD45RA+CD45RO− (data not shown). When the other CXCR3+CD3+ population (R2) was examined for CD4 and CD8 expression, 2 different subsets, one containing CD4+CD8+ T cells (R2I) and the second CD4−CD8+(low) T cells (R2II), were observed (Figure 4C). By contrast, the CXCR3+CD3− cells (R3) showed poor, if any, CD4 or CD8 expression (Figure 4D).
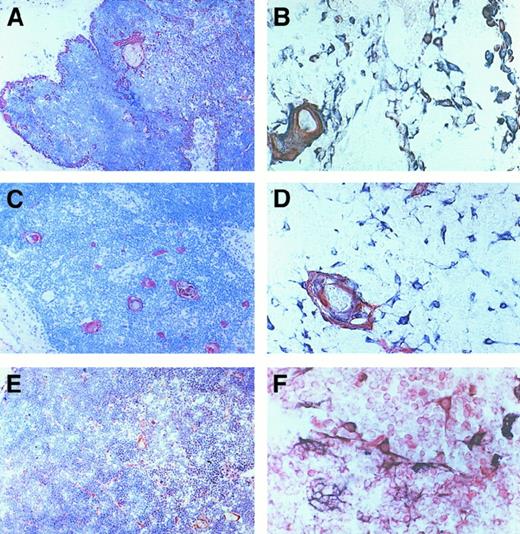
Localization of CXCR3-expressing cells in human thymus.
(A) CXCR3 expression in the medulla and in cells of subcapsular areas, as indicated by arrows (original magnification × 100). (B) Higher magnification of medullary CXCR3-expressing cells localized in proximity to a Hassall corpuscle (original magnification × 1000). Sections were immunostained with anti-CXCR3 Ab using the avidin-biotin-peroxidase method. One representative experiment is shown.
Localization of CXCR3-expressing cells in human thymus.
(A) CXCR3 expression in the medulla and in cells of subcapsular areas, as indicated by arrows (original magnification × 100). (B) Higher magnification of medullary CXCR3-expressing cells localized in proximity to a Hassall corpuscle (original magnification × 1000). Sections were immunostained with anti-CXCR3 Ab using the avidin-biotin-peroxidase method. One representative experiment is shown.
Phenotype of CXCR3-expressing cells in human thymus.
Freshly isolated thymocyte suspensions were assessed for different surface markers by 4-color FACS analysis. (A) Thymocyte costaining for CXCR3 and CD3 showing 2 CD3+ subsets (R1, 1.3%; and R2, 0.4%) and one CD3− subset (R3, 0.3%). (B-D) Costaining of the R1, R2, and R3 subsets for CD4 and CD8. (E-G) Costaining of the CXCR3+ subsets for TCRγδ and TCRαβ. (H-J) Staining of the CXCR3+ subsets for CD4 and CD56. Data shown are representative of 6 separate experiments.
Phenotype of CXCR3-expressing cells in human thymus.
Freshly isolated thymocyte suspensions were assessed for different surface markers by 4-color FACS analysis. (A) Thymocyte costaining for CXCR3 and CD3 showing 2 CD3+ subsets (R1, 1.3%; and R2, 0.4%) and one CD3− subset (R3, 0.3%). (B-D) Costaining of the R1, R2, and R3 subsets for CD4 and CD8. (E-G) Costaining of the CXCR3+ subsets for TCRγδ and TCRαβ. (H-J) Staining of the CXCR3+ subsets for CD4 and CD56. Data shown are representative of 6 separate experiments.
To better characterize the nature of the different CXCR3+ cell populations, their expression of TCRγδ and CD56 was also assessed. As expected, the CD3+CD4−CD8+TCRαβ+ subset (R1) showed neither TCRγδ nor CD56 expression (Figure 4E,H), which supports the concept that the subset was composed of CD8+ SP mature T cells. The cells of the CD3+TCRαβ+CD4+CD8+subset (R2I) were also TCRγδ− (Figure 4F) and CD56− (Figure 4I), suggesting that they were DP immature thymocytes. By contrast, the cells of the CD3+CD4+CD8+(low) subset (R2II) were TCRαβ−TCRγδ+CD56− (Figure4F,I), suggesting they were CD4−CD8+TCRγδ+ T cells. Finally, the cells of the CD3−CD4−CD8− subset (R3) were also TCRαβ− and TCRγδ− (Figure 4G) but CD56+ (Figure 4J), suggesting their NK cell nature. Accordingly, most of these cells also expressed CD94 and CD16 (data not shown).
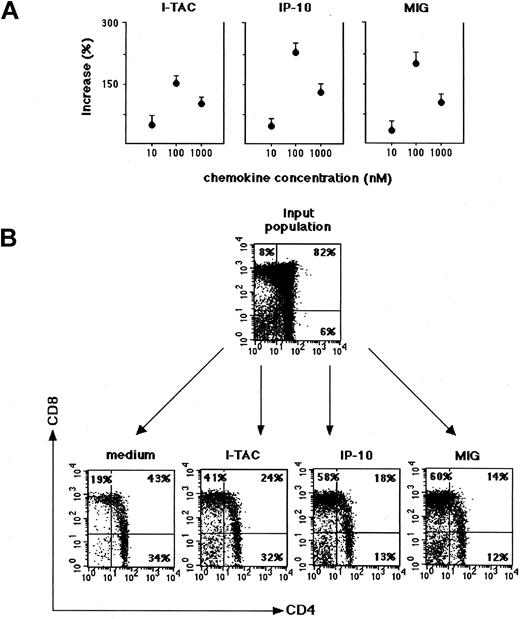
IP-10, Mig, and I-TAC in the human thymus act as chemoattractants for TCRαβ+CD8+ SP T cells, TCRγδ+ T cells, and NK-type cells
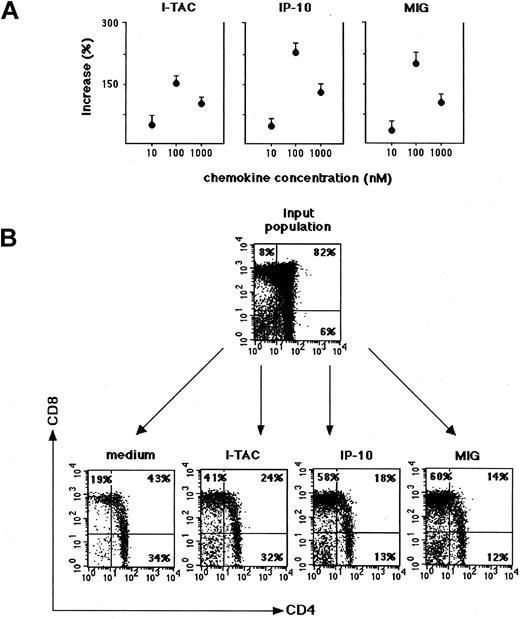
To establish whether CXCR3 expressed by either CD3+ or CD3− thymocyte populations was functional, the chemotactic activity of IP-10, Mig, and I-TAC on thymocyte suspensions from 4 postnatal human thymuses was assessed. Each chemokine was initially used at 3 different concentrations, and the concentration giving the maximal thymocyte migration into the lower chamber was considered as the optimal concentration to be used in subsequent experiments. Thymocytes were collected after chemotaxis to IP-10, Mig, and I-TAC, directly counted by FACScalibur for 30 seconds, and then analyzed by flow cytometry for the expression of CD4 and CD8 in comparison with the input thymocyte population as well as with the control population that had migrated in response to medium alone. The number of migrating cells was maximal at the 100 nM concentration with all 3 chemokines (Figure5A). There was a small number of cells migrating in response to control medium alone, which contained CD4+CD8+ DP T cells as well as CD4+CD8− and CD4−CD8+SP T cells. We noted a decrease in the proportion of CD4+CD8+ DP T cells and an increase in the proportion of both CD4+CD8− and CD4−CD8+ SP T cells in comparison with the input thymocyte population (Figure 5B). Percentage values of input cells that were responding to the chemoattractant activity of I-TAC, IP-10, and Mig are reported in Table 1. Thymocyte suspensions that migrated in response to the optimal concentration of IP-10 and Mig were largely enriched in CD4−CD8+ SP T cells as well as CD4−CD8− DN T cells. The proportions of CD4−CD8+ cells that migrated in response to I-TAC were lower, and CD4−CD8− DN T cells showed virtually no migration in response to I-TAC (Figure 5B, Table1).
Chemoattraction of thymocytes from human thymuses by I-TAC, IP-10, and Mig.
Freshly isolated thymocytes from thymuses of 5-day-old to 3-month-old children were assayed for chemotaxis in response to different concentrations of the 3 chemokines. (A) Values are expressed as percentage increase of cells migrated in response to I-TAC, IP-10, and Mig versus cells migrated in the presence of medium alone. Data represent mean values ± SE of 4 separate experiments. (B) Input and migrated thymocytes were costained for CD4 and CD8 and analyzed by FACSCalibur.
Chemoattraction of thymocytes from human thymuses by I-TAC, IP-10, and Mig.
Freshly isolated thymocytes from thymuses of 5-day-old to 3-month-old children were assayed for chemotaxis in response to different concentrations of the 3 chemokines. (A) Values are expressed as percentage increase of cells migrated in response to I-TAC, IP-10, and Mig versus cells migrated in the presence of medium alone. Data represent mean values ± SE of 4 separate experiments. (B) Input and migrated thymocytes were costained for CD4 and CD8 and analyzed by FACSCalibur.
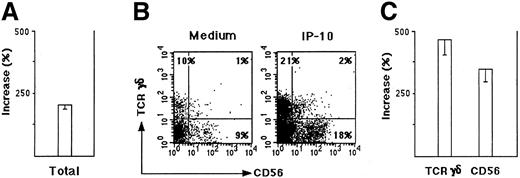
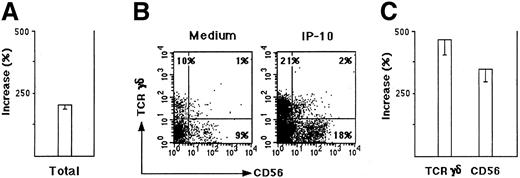
To determine if these populations migrated by chemotaxis, we performed a checkerboard analysis. The 2 above-mentioned populations transmigrated by chemotaxis because their migration occurred only when a gradient of IP-10 existed between bottom and top compartments, with a higher concentration in the bottom (data not shown).To establish whether CXCR3 expressed by TCRγδ+ T cells and by NK cells in the human thymus was functional, thymocyte suspensions were depleted from TCRαβ+ cells, and the chemotactic response to IP-10, Mig, and I-TAC of the remaining CXCR3+cells was examined. Figure 6A shows the percentage increase of cells responding to IP-10 in comparison with the migration of the same cell population in the presence of medium alone. Figure 6B shows that 2 populations of TCRαβ− cells migrated in response to IP-10, one of them composed of TCRγδ+CD56− and the other composed of TCRγδ−CD56+ cells. As shown in Figure 6C, the percentage increase of both TCRγδ+CD56− and TCRγδ−CD56+ cells was markedly higher than that observed in the whole migrated population. Similar results were obtained by using Mig or I-TAC (data not shown). Taken together, these data indicate that at least 3 thymocyte populations shown to possess the CXCR3 (TCRαβ+CD8+ SP T cells, TCRγδ+ T cells, and NK-type cells) can migrate in response to the CXCR3-binding chemokines IP-10, Mig, and I-TAC.
Chemoattraction of thymocyte suspensions depleted of TCRαβ+ cells.
Thymocyte suspensions were depleted of TCRαβ+ cells by the MACS system (TCRαβ−) as described in “Materials and methods.” (A) Migration was expressed as percentage increase of cells responding to 100 nM IP-10 versus cells migrated in the presence of medium alone. The mean values ± SE were obtained in 4 separate experiments. (B) Data from one representative experiment showing the expression of TCRγδ and CD56 by TCRαβ− thymocytes migrated in response to medium alone or to 100 nM IP-10. (C) Migration of TCRγδ+ and CD56+ in response to IP-10, expressed as percentage increase in comparison with the same cells migrated in response to medium alone. The mean values ± SE obtained in 4 separate experiments are reported.
Chemoattraction of thymocyte suspensions depleted of TCRαβ+ cells.
Thymocyte suspensions were depleted of TCRαβ+ cells by the MACS system (TCRαβ−) as described in “Materials and methods.” (A) Migration was expressed as percentage increase of cells responding to 100 nM IP-10 versus cells migrated in the presence of medium alone. The mean values ± SE were obtained in 4 separate experiments. (B) Data from one representative experiment showing the expression of TCRγδ and CD56 by TCRαβ− thymocytes migrated in response to medium alone or to 100 nM IP-10. (C) Migration of TCRγδ+ and CD56+ in response to IP-10, expressed as percentage increase in comparison with the same cells migrated in response to medium alone. The mean values ± SE obtained in 4 separate experiments are reported.
Discussion
Little is known about the expression of chemokines and/or their receptors in human thymus as well as their functional role on human thymocytes. SDF-1 has been shown to induce the migration of immature human thymocytes7; TECK efficaciously induced the chemotaxis of both immature DP and mature CD4+ and CD8+ SP cells31,32; eotaxin promoted the migration of all human thymocytes except the immature DN population28; MDC was found to be selectively produced by medullary epithelial cells and preferentially attracted a population of CD3+CD4+CD8+(low)thymocytes17,35; and CCR5 was detected on a small subset of human thymocytes, but apparently it did not exhibit chemotactic activity on the same cells.7
Although the presence of CXCR3 and/or its ligands IP-10, Mig, and I-TAC in murine thymus has already been described by using Northern blot analysis,13,14,20 21 their functional activity, as well as the nature of thymic cells responsible for their expression, has not yet been explored. Moreover, nothing is known about both the presence and functional activity of these chemokines in human thymus. Without doubt, our results provide evidence that all the 3 CXCR3-binding chemokines are expressed in human thymus and are produced by thymic epithelial cells (TECs) but not by B cells, macrophages, or mature DCs. I-TAC expression, but not IP-10 or Mig expression, by a proportion of cortical and medullary T cells was also observed. The results of this study also demonstrate that IP-10, Mig, and I-TAC are mainly detectable in the medullary areas, with some differences in regard to their localization. Both Mig mRNA and protein were strongly expressed at the level of Hassall corpuscles, whereas IP-10 mRNA and protein could be found in numerous TECs spread in the medulla and also in some cortical TECs, especially those localized to the subcapsular areas. I-TAC mRNA and protein were less intensely expressed in the medulla and appeared to be more spread throughout all the thymus due to their expression by both TEC and a proportion of T cells. Whether these differences in the distribution of IP-10–, Mig-, and I-TAC–producing cells really reflect distinct functional activities is still unclear. In agreement with the prevalent medullary localization of IP-10, Mig, and I-TAC, even their common receptor, CXCR3, was preferentially expressed in the thymic medulla as well as in cells present along the subcapsular areas. Surprisingly, when the nature of CXCR3+cells was examined, 4 distinct thymocyte populations were found to express CXCR3, and at least 3 of them appeared to be responsive to the chemoattractant activity of IP-10, Mig, and I-TAC.
The most abundant CXCR3+ population responsive, at least in vitro, to the chemoattractant activity of IP-10, Mig, and I-TAC was represented by CD8+ SP mature T cells because their phenotype was CD4−CD8+CD3+(high)TCRαβ+TCRγδ−CD45RA+CD45R0−CD−. CD8+ SP mature thymocytes have also been found to migrate in response to CCR7-binding ELC and SLC in both mice17,22,27 and humans.35 It is of interest, however, that both ELC and SLC acted as chemoattractants for both CD4+ and CD8+ SP mature thymocytes, whereas the CXCR3-binding chemokines did not exert any chemotactic activity on the CD4+ SP T-cell population, but it selectively attracted the CD8+ SP T-cell population. This finding is in keeping with the results reported by Soto et al20 showing CXCR3 mRNA expression by CD8+ murine thymocytes. It is unclear if the chemoattraction of IP-10, Mig, and I-TAC on this thymocyte population occurs before or after the process of negative selection. Indeed, the question of whether negative selection predominantly takes place in the cortex or in the medulla is still controversial.37,38 An obvious problem with the medulla as a site for negative selection is that the great majority of medullary T cells are SP cells and are thought to be beyond the stage of central tolerance induction.1
Recently, however, it has been shown that medullary TECs possess all the potential to directly mediate the process of negative selection because they can express different autoantigens39,40 as well MHC class II antigens41 and the costimulatory molecule CD80,42,43 which are essential for autoantigen recognition and T-cell activation, respectively. These findings, together with the observation here reported that CD8+ SP T cells attracted by IP-10, Mig, and I-TAC were strongly enriched in CD45RA+ cells in comparison with the input CD8+population, suggest that CXCR3+CD8+ SP T cells responsive to these chemokines are fully mature thymocytes that have already passed the process of negative selection in the medulla and are ready to migrate into the periphery. This finding is also in agreement with a recent report showing CXCR3 expression by both naive and activated circulating CD8+ T cells, whereas staining of naive CD4+ T cells for CXCR3 was found to be minimal.44
Another CXCR3+ population responsive in vitro to IP-10, Mig, and I-TAC in the human thymus showed the phenotype of CD3+TCRγδ+ T cells. This finding is apparently at variance with a previous report45 showing that neither resting TCRγδ+ T cells from normal peripheral blood nor activated human TCRγδ+ T-cell clones exhibited transendothelial migration in response to Mig. However, these authors45 neither assessed TCRγδ+ T cells for CXCR3 expression nor tested their response to the other CXCR3-binding chemokines, IP-10 and I-TAC. We have recently found that virtually all TCRγδ+ T cells in normal peripheral blood express CXCR3 and are attracted in vitro by the CXCR3-binding chemokines, especially by Mig (data not shown). Thus, it is likely that the apparent discrepancy between the results of this study and those reported by Roth et al45 mainly reflects differences in the chemotactic assay used (simple transwell versus transendothelial).
A third population of CXCR3+ cells in the human thymus consisted of NK-type cells in different stages of maturation. These cells were indeed CD3−CD56+, and a proportion of them expressed CD94 and CD16. CD56 seems to be one of the first NK-cell markers that appear during in vitro NK-cell development in response to interleukin (IL)-2 and IL-7 from fetal liver cell precursors followed by CD94 and CD16.46 The demonstration of CXCR3 expression on and responsiveness to IP-10 and Mig by the small population of NK cells present in the human thymus is in keeping with previous reports showing that IP-10 and Mig are potent NK-cell chemoattractants in vitro.47 48 This suggests that IP-10 and Mig are not only important for the accumulation of these cells in the inflamed tissues but also for their migration throughout the thymus and/or from the thymus to the periphery. By contrast, NK cells showed poor if any chemotactic response to I-TAC as shown by the lack of response of CD4−CD8− DN thymocytes. It remains to be established whether (1) this different activity of I-TAC reflects a lower affinity of CXCR3 present on NK cells for this chemokine or (2) the activity is due to the fact that I-TAC was largely expressed throughout the thymus and clearly detectable not only on medullary epithelial cells but also on thymic T cells, thus reducing the gradient during the chemotactic assay.
Finally, the results of this study demonstrate that CXCR3 can be expressed by a fourth population of human thymocytes that, because of their small number, could not be precisely characterized for either phenotype or chemokine responsiveness; however, at least on the basis of the expression of some surface markers, CD3+(low)CD4+CD8+TCRαβ+, it could be identified as an immature DP T-cell population. This population may be composed of the CXCR3+ cells found in the subcapsular areas, the same cells that likely respond to the chemoattractant activity of IP-10 produced by epithelial cells localized to the same areas. At present, however, this possibility remains unproven. The demonstration of CXCR3 expression in a small population of immature T cells in human thymus is also in keeping with the previous demonstration of CXCR3 mRNA expression by a subset of murine pro-T lymphocytes.19 However, the differences existing in the early stages of thymic T-cell development of mice and humans, predominantly in the timing of the appearance of CD4, CD8α, and CD8β,46,49 50 make it difficult to ascertain whether the expression of CXCR3 by a subset of immature T cells has the same functional meaning in the thymus development of the 2 species.
Despite this still unsolved question, the results of the present study provide the first evidence that IP-10, Mig, and I-TAC may play an important role in the transmigration of at least 3 distinct subsets of mature thymocytes during the process of lymphopoiesis in human thymus. On the one hand, the redundant expression of CXCR3-binding chemokines stresses their critical role in this function; on the other hand, their production in at least partially distinct thymic areas suggests that they may selectively act in vivo on distinct thymocyte subsets according to their different receptor affinity.
Supported by grants provided by Associosione Italiana Ricerca Cancro, Italian Ministry of Health (AIDS Project 2000), and Ministry of Education.
P.R. and F.A. contributed equally to this work.
Submitted July 7, 2000; accepted October 2, 2000.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
S. Romagnani, Dipartimento di Medicina Interna, Sezione di Immunoallergologia e Malattie dell'Apparato Respiratorio, Viale Morgagni 85, Firenze 50134, Italy; e-mail:s.romagnani@dfc.unifi.it.