The toxicity of preparative regimens render neonatal bone marrow transplantation (BMT) for progressive childhood diseases a controversial treatment. Ablative BMT in neonatal mice with or without the lysosomal storage disease mucopolysaccharidosis type VII (MPS VII) show high morbidity and developmental disruption of both brain and bone structure. In this investigation, BMT was performed with a high dose of congenic, normal bone marrow into nonablated newborn mice. Recipients had lifelong, multilineage, peripheral blood chimerism with the donor β-glucuronidase-positive (GUS+) cells that was both well tolerated and therapeutic. Three daily injections of normal adult marrow increased the average life span by at least 6 months and corrected the functional breeding deficits typical of the MPS VII mice. Twelve months after injection, several structural features of femurs were more like that of normal mice than of untreated MPS VII mice. Periosteal circumference and bone cortical thickness were significantly improved in males and cortical density did not differ significantly from values in normal females. Significant reduction of lysosomal glycosaminoglycan storage corresponded directly with GUS enzyme activity and percentage of histochemically GUS+ cells in visceral organs and hematopoietic tissues such as thymus, spleen, peripheral blood, and bone marrow. By all criteria tested, BMT into neonatal MPS VII mice in the absence of any preparative regimen is a successful therapy.
Introduction
A growing number of genetic disorders respond therapeutically to bone marrow transplantation (BMT).1,2BMT can restore immune function3 and stabilize multiple forms of anemia and hemoglobinopathies.4 For some metabolic diseases, myeloablative transplantation with normal hematopoietic tissues delivers cells carrying protein that, by cross-correction, stabilize cellular biochemistry and alleviate disease in surrounding tissues. A significant proportion of diseases that respond to BMT, such as the lysosomal storage diseases,5are highly progressive in nature and show improved response when BMT is performed early in development.6-9 When treatment occurs in the first few years of life, BMT halts progressive neuropsychological retardation10 and can reverse the hepatosplenomegaly and corneal clouding1 commonly observed in the lysosomal storage diseases. Currently, most transplantation protocols use chemical ablative preparatory regimens to obtain high levels of engraftment. These techniques are highly toxic in the neonatal period and can cause developmental malformations as well as increased mortality.8 11 Optimization of the transplantation regimen as well as donor cell type (both ontologically and phenotypically) are clearly paramount to the successful treatment of young children with life-threatening genetic disorders.
The homing and engraftment of donor hematopoietic stem cells (HSCs) in the rapidly expanding and developing neonatal environment is not well understood. Early experiments in murine adult BMT led to the hypothesis that HSC reside in marrow “niches” that require “opening” with irradiation or cytoreductive reagents.12 This idea was supported by experiments performed without marrow ablation that showed poor engraftment of donor cells.13-15 The concept of the HSC “niche” was put into question when Brecher and colleagues16 successfully engrafted male donor cells into nonmyeloablated female recipients using intravenous injection with very high cell doses. Subsequently, results in the nonablated adult transplant setting have been further improved by increasing the number of cells injected and performing multiple injections.17 18These results are significant because of the severe morbidity and mortality associated with lethally ablative BMT. Thus far, these experiments have not been replicated in the neonatal setting or performed in a model for genetic disease where therapeutic efficacy can be fully evaluated.
The mucopolysaccharidosis type VII (MPS VII) mouse19,20 has a lysosomal storage disease that is a model for developing therapies for Sly disease (human MPS VII)21 and other lysosomal enzyme deficiencies. These animals, as in humans, lack the lysosomal glycosaminoglycan (GAG)-degradative enzyme β-glucuronidase (GUS). MPS VII is a rapidly progressive genetic disorder causing premature death. It is manifested by the accumulation of dermatan sulfate, heparan sulfate, and chondroitin 4- and 6-sulfates and the simultaneous increase of other enzymes such as β-hexosaminidase and α-galactosidase in the lysosomes of most cell types.5These distended “foamy” lysosomes significantly disrupt tissue structure and function.19 Phenotypic changes include shortened and thickened long bones, reduced mobility, hearing loss, cognitive dysfunction, corneal clouding, hepatosplenomegaly, and inability to breed or maintain offspring to weaning.19,20,22 23
Successful alleviation of visceral storage disease in murine MPS VII has been accomplished by BMT into myeloablated adults24 and newborns.8 In the adult, the dysmorphology and progressive neurologic anomalies are already well established and are not corrected. In the newborn, the toxicity of the preparative regimen disrupts brain architecture and retards long bone growth. Enzyme replacement therapy (ERT) from birth to 5 weeks of age25-28alleviates the growth anomalies, corrects the breeding deficit,23 and improves results of behavioral testing.29 However, the enzyme is a putative immunogen in the null recipient and is short-lived. Following cessation of enzyme treatment, lysosomal storage returns in all tissues.27 Our experiences suggest that early BMT without prior cytoablation may offer a more complete and lasting cure. In this report, we test the efficacy of high-dose marrow transplantation into MPS VII neonates that received no preparative treatment.
Materials and methods
Treatment of mutant mice
Animals were maintained and handled using protocols reviewed by the Animal Care and Use Committee at The Jackson Laboratory under guidelines set forth by the Public Health Service. MPS VII mice (gusmps/gusmps) were obtained by brother-sister mating of B6.C-H2bm1/ByBir-+/mps (gus+/gusmps) animals.19MPS VII recipients were identified by biochemistry and polymerase chain reaction (PCR) soon after birth.23,30,31 Neonatal recipients of adult B6.C-H2bm1/ByBir-+/+(gus+/gus+) whole bone marrow were injected intravenously on 3 consecutive days (3xBMT) through the superficial temporal vein.32 Each recipient was injected with 100 μL containing 0.8 to 1 × 107bone marrow cells per day. Positive controls for functional restoration of breeding capabilities received 6 weekly injections of recombinant mouse GUS (6xERT) as previously described.25,27 33 Each dose contained 28 000 U enzyme, where units are nanomoles of 4-methylumbelliferyl β-d-glucuronide substrate hydrolyzed per milligram protein per hour.
Repopulation analysis
Peripheral blood (PB) was collected from adult transplant recipients in heparinized hematocrit tubes by retro-orbital puncture and transferred to 0.5 mL phosphate-buffered saline (PBS) with 1 mM EDTA on ice. Red blood cells (RBCs) were removed by 2 washes in ACK lysis buffer (150 mM NH4Cl, 1 mM KHCO3, and 0.1 mM EDTA). Each lysis was for 5 minutes at room temperature followed by a wash with Hanks balanced salt solution (HBBS) containing 2% fetal bovine serum (FBS) and 2 mM EDTA (HBSS+). Cells from each recipient were resuspended in 1 mL HBSS+ containing 10 mM HEPES pH 7.4 and 100 μM of the GUS substrate Imagene Green C12FDGlcU (lipophilic analog of fluorescein di-β-d-glucuronic acid, Molecular Probes, Eugene, OR). Cells were incubated at 37°C for 1 hour and 15 minutes. DNase I (Sigma Chemical, St Louis, MO) was then added to a final concentration of 15 μg/mL and incubation was continued 15 minutes further. Cells were washed and labeled with either Mac-1 (CD11b)/PE (M1/70), Gr-1/PE (RB6-8C5), CD45R/PE (RA3-6B2), or αβTcR/PE (H57-597) and examined by flow cytometry for expression of GUS in both myeloid and lymphoid lineages. At necropsy, bone marrow was collected from femur and tibia by flushing with PBS. The filtered, single-cell suspension was washed one time with ACK lysis buffer and then stained with Imagene Green C12FDGlcU and both myeloid and lymphoid markers as described above. Reagents for cell labeling were purchased from Pharmingen (San Diego, CA).
Biochemistry and histopathology
Mice were necropsied and tissues were collected from 4 female and 3 male 3xBMT mice at 1 year after transplantation. Tissues were saved for biochemistry, histology, and histochemistry.20,24 Quantification of lysosomal enzyme activities (β-glucuronidase, β-hexosaminidase, α-galactosidase) were determined biochemically in tissue homogenates as described.31 Activities were determined using the fluorescent substrates 4-methylumbelliferyl-β-d-glucuronide, 4-methylumbelliferyl-β-d-glucosaminide, and 4-methylumbelliferyl-α-d-galactoside, respectively (Sigma Chemical). Final values for enzyme activity of tissues from treated animals were expressed as percent +/+ specific activity. Histochemical analysis for identification of GUS+ cells in individual tissues was performed on 10-μm frozen sections as described elsewhere.31 For histologic analysis, tissue fragments from the 3xBMT recipients and controls were fixed and examined by light microscopy for reduction of lysosomal storage.27
Morphologic analysis of long bones
Femurs were collected from 3xBMT MPS VII males and females at 1 year after injection. In addition, femurs were collected from untreated +/+ and untreated MPS VII males and females (3-5 animals per group). Bones were analyzed by peripheral quantitative computer tomography (pQCT) with a Stratec XCT 960M densitometer (Norland Medical Systems, Ft Atkinson, WI), as described.34 Sequential cross-sectional scans were taken at 2-mm intervals down the length of each bone and numerical values were assigned to total density, cortical density, cortical thickness, periosteal circumference, and femur length. Values for treated males and females were compared to the untreated controls by Student t test with SigmaStat statistical analysis software (Jandel Scientific, San Rafael, CA).P values were used to determine significant difference from controls.
Reproductive analysis
Foster mothers were generated by mating BALB/c adult mice 1 to 3 days before mating of treated MPS VII mice. To identify day 0 of pregnancy, females were examined for vaginal plugs and once identified, males were removed. Treated males and females (6xERT) were mated 1 week after their last enzyme injection.23 Similarly, 3xBMT recipients were mated at 7 weeks after marrow transplant. On day 15 to 17 of pregnancy, treated pregnant MPS VII females were paired with pregnant BALB/c females. Because mating was initiated earlier with the foster mothers, the BALB/c mothers gave birth first. The day that treated MPS VII mothers gave birth, all but 1 to 2 BALB/c pups were removed. These animals were easily identified by pink eye pigmentation versus black pigmentation in the MPS VII pups. Numbers of MPS VII offspring were monitored and recorded until weaning. After weaning, treated animals were mated a second time as described above.
Results
Nonmyeloablative neonatal transplantation of +/+ donor cells engraft long term
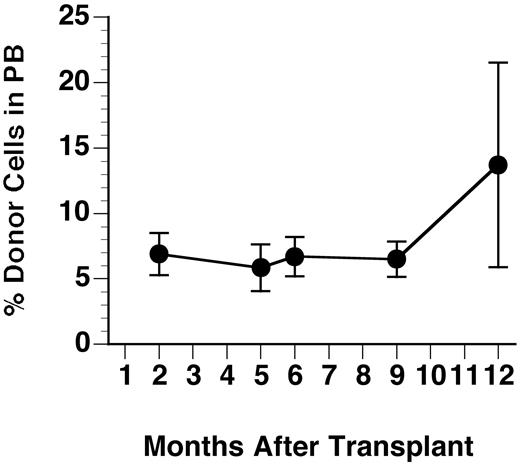
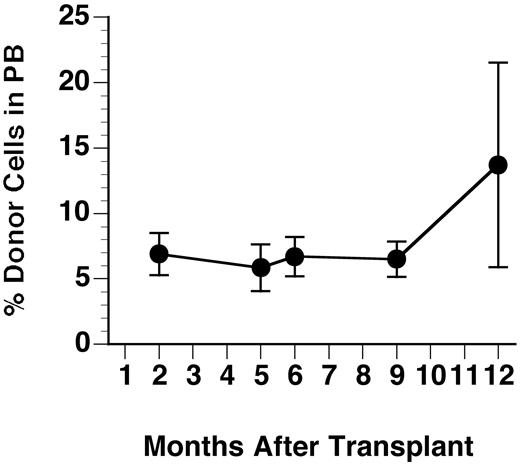
The GUS+ donor cells identified by the fluoresceinated GUS substrate FDGlcU were evaluated for percent repopulation in PB and bone marrow (BM) by flow cytometry. Even without myeloablation, 3xBMT MPS VII mice treated as neonates maintained engraftment until necropsy at 1 year of age (Figure 1). The mean percent donor cells (± SD) in PB were 6.9% ± 1.6%, 5.9% ± 1.8%, 6.7% ± 1.3%, 6.5% ± 1.3%, and 13.7% ± 7.8% at 2, 5, 6, 9, and 12 months, respectively. Figure2 compares the staining profile of bone marrow labeled for GUS expression and myeloid/lymphoid cell markers in a representative recipient and an untreated control at necropsy. Similar results were obtained during analysis of PB. Averages for percent GUS+, lineage-positive cells at 1 year of age are summarized in Table 1. Donor cells were present in all cell populations of the 7 recipients examined indicating multilineage engraftment by HSCs. There was no notable difference in levels of engraftment between males and females. The percent total GUS+ donor cells in BM at 1 year after transplantation averaged 14.1% ± 4.7%, similar to the values in the PB.
Percent repopulation of GUS+ donor cells in PB of transplant recipients.
Blood samples were lysed of RBCs and nucleated cells were incubated with the fluoresceinated GUS substrate Imagine Green FDGlcU followed by staining with lineage marker antibodies. Values shown are averages of both males and females and error bars show the SD (n = 7-12).
Percent repopulation of GUS+ donor cells in PB of transplant recipients.
Blood samples were lysed of RBCs and nucleated cells were incubated with the fluoresceinated GUS substrate Imagine Green FDGlcU followed by staining with lineage marker antibodies. Values shown are averages of both males and females and error bars show the SD (n = 7-12).
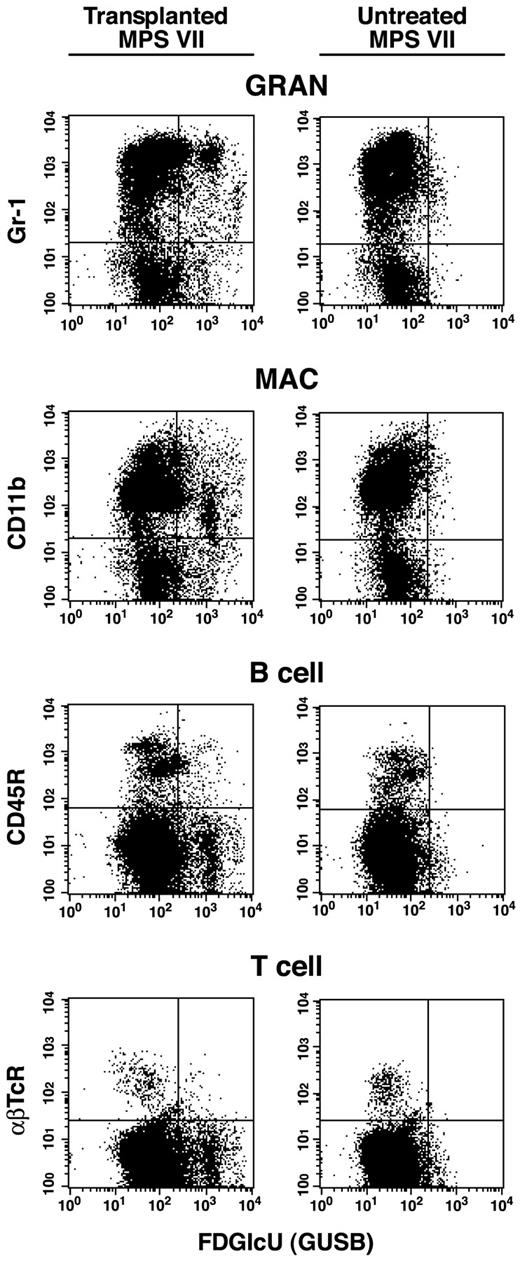
Flow cytometry.
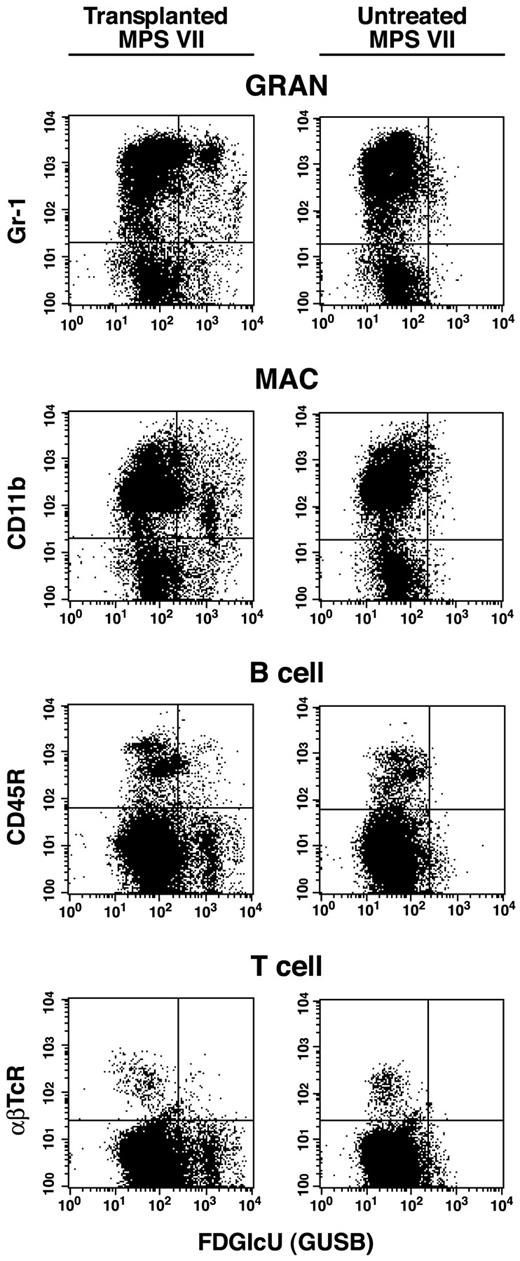
Flow cytometric analysis of bone marrow collected from a representative nonmyeloablated MPS VII neonatal transplant recipient of adult bone marrow at 1 year after engraftment (first column). Marrow was also collected from an untreated MPS VII control for comparison (second column). RBCs were removed by lysis and nucleated cells were incubated with the fluoresceinated GUS substrate Imagene Green FDGlcU followed by staining with phycoerythrin (PE)-conjugated lineage marker antibodies. These antibodies included Mac-1 (CD11b), Gr-1, B220 (CD45R), or αβTcR for macrophages, granulocytes, B cells, and T cells (αβ receptor), respectively. All transplant recipients showed a similar pattern of GUS+, lineage-positive cells demonstrating multilineage engraftment by +/+ donor HSC.
Flow cytometry.
Flow cytometric analysis of bone marrow collected from a representative nonmyeloablated MPS VII neonatal transplant recipient of adult bone marrow at 1 year after engraftment (first column). Marrow was also collected from an untreated MPS VII control for comparison (second column). RBCs were removed by lysis and nucleated cells were incubated with the fluoresceinated GUS substrate Imagene Green FDGlcU followed by staining with phycoerythrin (PE)-conjugated lineage marker antibodies. These antibodies included Mac-1 (CD11b), Gr-1, B220 (CD45R), or αβTcR for macrophages, granulocytes, B cells, and T cells (αβ receptor), respectively. All transplant recipients showed a similar pattern of GUS+, lineage-positive cells demonstrating multilineage engraftment by +/+ donor HSC.
Therapeutic levels of GUS are restored in multiple tissues
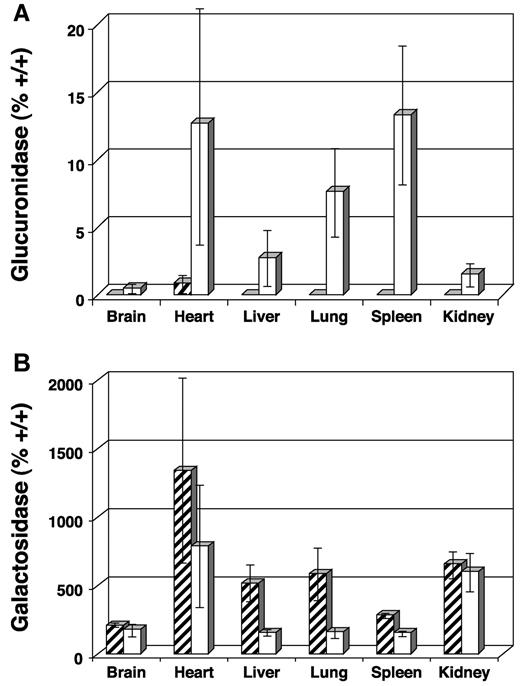
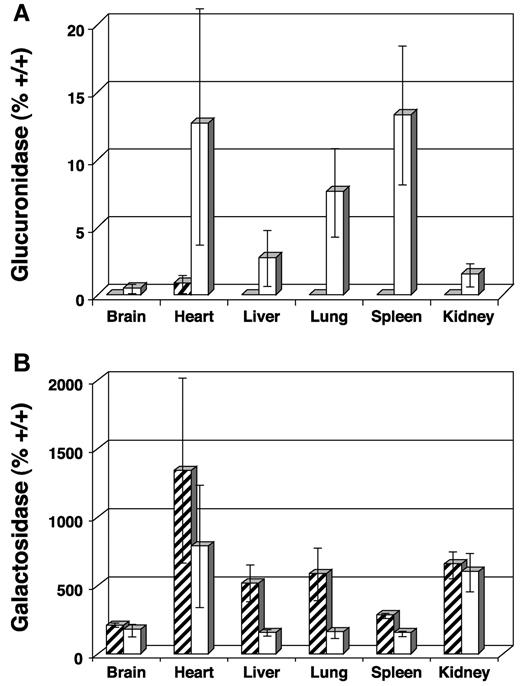
At necropsy, tissues were examined biochemically for the presence of GUS activity and reduction of activity in the secondary lysosomal enzymes α-galactosidase and β-hexosaminidase. The activity of these latter 2 enzymes are elevated when GUS activity is low and become normalized as GUS activity is restored. Average GUS activity expressed as percent +/+ was 0.3%, 12.3%, 2.6%, 7.4%, 13.1%, and 1.3% for brain, heart, liver, lung, spleen, and kidney, respectively (Figure3A). Values obtained for the α-galactosidase assay are presented in Figure 3B. A significant decrease in secondary enzyme levels was observed in most tissues with more than 2% GUS activity (decreased activity in heart was not significant due to high SD, P = .155). Values for brain and kidney, for example, were similar to untreated MPS VII controls (n = 3). Results from the β-hexosaminidase assay were similar (data not shown).
Biochemical analysis of tissue homogenates collected from transplanted MPS VII mice 1 year after engraftment and from untreated MPS VII controls.
Final values for enzyme-specific activity are expressed as average percent +/+ activity. (A) GUS activity showing increases in heart, liver, lung, and spleen and only modest increases in brain and kidney. (B) Decreased α-galactosidase activity was observed in tissues with the highest GUS activity, confirming lysosomal correction of GAG biochemistry. Levels of α-galactosidase activity remain high in brain and kidney, reflecting the low GUS activity seen in panel A. ▨ indicates untreated MPS VII; ■, 3x neonatal BMT.
Biochemical analysis of tissue homogenates collected from transplanted MPS VII mice 1 year after engraftment and from untreated MPS VII controls.
Final values for enzyme-specific activity are expressed as average percent +/+ activity. (A) GUS activity showing increases in heart, liver, lung, and spleen and only modest increases in brain and kidney. (B) Decreased α-galactosidase activity was observed in tissues with the highest GUS activity, confirming lysosomal correction of GAG biochemistry. Levels of α-galactosidase activity remain high in brain and kidney, reflecting the low GUS activity seen in panel A. ▨ indicates untreated MPS VII; ■, 3x neonatal BMT.
Microscopic analysis of tissues reveals the presence of donor-derived cells and reduction of lysosomal storage
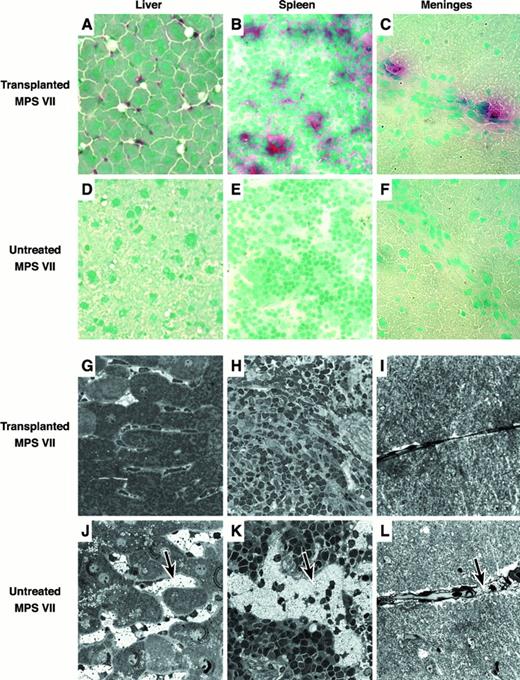
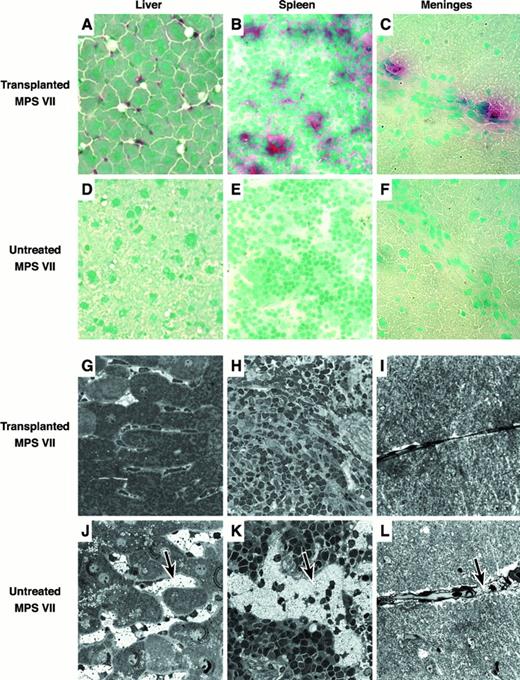
Liver, spleen, brain, kidney, lung, heart, thymus, and bone marrow were embedded in Tissue Tek, frozen, and sectioned for histochemical detection of GUS-expressing donor-derived cells (Figure4A-F). GUS+ red/pink donor cells were identified in all tissues examined. Tissues with the greatest number of positive cells included liver, spleen, lung, thymus, and bone marrow. The majority of GUS+ cells found in the liver were located in the sinusoids and are most likely Kupffer cells (Figure 4A). The spleen contained positive cells primarily in the red pulp (Figure 4B). In the brain, donor cells were concentrated along the meninges overlying the cortex with a few cells in the parenchyma (Figure 4C). The kidney contained very few GUS+ cells, located primarily in scattered glomerular mesangial regions. Heart muscle and lung had positive interstitial cells. Thymus contained donor-derived cells in both the medulla and cortex. Bone marrow had numerous positive cells throughout.
Histologic analysis of transplanted and untreated control MPS VII mice.
(A-F) Frozen tissue sections stained with naphthol AS-BI β-d-glucuronide and counterstained with methyl green. Donor-derived pink/red GUS+ cells can easily be seen in the tissues of the transplant recipients. (G-L) Microscopic analysis of fixed tissues showing the reduction in lysosomal GAG storage. Tissues from the transplant recipients have reduced amounts of distended lysosomal “foamy” structures as compared to the untreated controls (see arrows).
Histologic analysis of transplanted and untreated control MPS VII mice.
(A-F) Frozen tissue sections stained with naphthol AS-BI β-d-glucuronide and counterstained with methyl green. Donor-derived pink/red GUS+ cells can easily be seen in the tissues of the transplant recipients. (G-L) Microscopic analysis of fixed tissues showing the reduction in lysosomal GAG storage. Tissues from the transplant recipients have reduced amounts of distended lysosomal “foamy” structures as compared to the untreated controls (see arrows).
Histopathology was assessed by light microscopy in plastic embedded 1-μm thick sections (Figure 4G-L). Comparisons were made between 3xBMT recipients, a surviving untreated 6-month-old MPS VII mouse, and a normal 1-year-old mouse. Liver and spleen of the recipients showed marked reduction in the amount of lysosomal storage in the sinus lining cells (Figure 4G,H). Hepatocyte pericanalicular storage was also reduced. There was no consistent reduction in the lysosomal distention in the glomerular or renal tubular epithelial cells or in the connective tissue cells in the heart. In the brain, there was consistent reduction in the amount of storage in the meninges (Figure4I) and glial cells, but neurons in the neocortex, hippocampus, and cerebellum failed to show a response to therapy. Meganeurites were prominent in the hippocampus and neocortex in several of the treated MPS VII mice. In the ear, several of the treated mice had only mild otitis, but the ossicles were adherent to thickened tympanic membrane. The bone of the ear in the treated mice was abnormal but had less evidence of alterations due to lysosomal storage than in the untreated mouse (not shown).
The presence of GUS+ cells in the neonatal period attenuates the malformation of long bones
Femurs collected at necropsy from male and female 3xBMT recipients, from untreated normal mice, and from untreated MPS VII mice were evaluated by pQCT densitometry. Table2 shows the values (± SD) from various measurements as well as P values from comparisons between treatment groups and controls. Sex-dependent differences in bone parameters were noted between normal controls, so males and females were assessed separately. The criterion for existence of an aberrant bone parameter was a significant difference between untreated MPS VII and normal mice. The criterion for actual improvement was a significant difference between 3xBMT mice versus untreated MPS VII mice, but not versus normal mice. Using these criteria, effects of treatment on male cortical density and female cortical thickness could not be evaluated. Males showed some improvement in total density, and both cortical thickness and periosteal circumference were most like normal. Females showed modest improvement in total density, normalized cortical density, and no change in periosteal circumference. Although bone parameters were, on the whole, improved, normal femur length was not achieved in either 3xBMT males or females (data not shown).
Reproduction is improved in neonatal transplant recipients
Breeding has been used as a functional test of therapeutic improvement in MPS VII transplant recipients. We showed previously that 6xERT in 6 weekly doses from birth restores breeding in males and maintains females through a second, usually lethal, pregnancy.23 Here, we repeated the 6xERT treatment regimen and compared results with 3xBMT recipients. Because MPS VII mothers maintain few of their young, pregnant BALB/c females were added near birth as foster mothers. A summary of results from the breeding experiment is shown in Table 3. All treated animals were able to breed and mothers carried normal-sized litters to term. Unlike 6xERT males, 3xBMT males continued to breed for 6 months (data not shown). Three of 5 6xERT and 3 of 6 3xBMT females bred successfully a second time. The average percent survival to weaning of fostered pups was 80.0% from the 6xERT matings and 86.6% from the 3xBMT matings. Pup survival was much improved compared to the 35.9% MPS VII pups maintained solely by 6xERT females (without fostering) previously.23
Discussion
Our experiments show that transplantation of normal BM into nonablated neonatal MPS VII recipients alleviates many disease symptoms. Improvements include extension of the life span by at least 6 months, reduction of lysosomal storage, normalization of several bone parameters, restoration of breeding by males, and increased numbers of successful pregnancies by females. Improvements are dependent on the successful implantation and long-term production of multiple lineages of HSC derivatives. These include PB and BM macrophages, neutrophils, T cells and B cells; Kupffer cells in the liver; sinus lining cells in hematopoietic and hepatic tissues; interstitial cells in heart, lung, and kidney; and cells lining the meninges and perivascular regions within the cortex of the brain. GUS produced by the donor-derived cells clears storage products accumulated in host lysosomes by cross-correction in hepatocytes and other visceral tissues.
Changes that are not as extensive as expected are in the architecture of the bones and the lysosomal storage in the brain. Presumably, the structural improvements observed in bone were due to donor-derived osteoclasts or osteoblasts.35 Complete normalization of bone was most likely not achieved because of the predominance of host-derived cells remaining in the marrow. With regard to the brain, it is widely accepted that microglial cells are hematopoietically derived36 and may repopulate the brain after transplantation. In mice transplanted as adults with lethal irradiation, donor cells derived from the monocytic lineage are found primarily in perivascular and leptomeningial sites, with very rare donor cells found in the parenchyma.37-39 The migration of donor cells into the central nervous system (CNS) is apparently quite slow and may take as long as a year.40 41 Similar results were obtained in our experiments without irradiation indicating that irradiation increases hematopoietic engraftment, but not necessarily migration into the brain.
Cognitive function has been evaluated after BMT in human patients with Hurler syndrome.9,10,42 When transplantation was performed early during onset of disease symptoms, therapeutic response was observed as arrest of further cognitive decline. It was only when BMT was initiated before onset of disease that patients demonstrated improved cognitive development. With regard to MPS VII, the amount of enzyme activity required for improvement in behavior and learning has not been determined. Neonatal murine MPS VII recipients ablated with 2 Gy and transplanted with adult bone marrow were shown to have 1.6% +/+ GUS activity.8 When these animals were tested for changes in spatial learning by the Morris Water-Maze test,43 no improvement was observed.44 This was thought to be due to irradiation-induced disruption of brain architecture during development, because identically treated normal controls showed the same defect in learning. MPS VII neonates treated with ERT attain about 10% +/+ GUS activity in the brain after 6 weekly injections, which subsequently declines after therapy ceases.27 When tested for cognitive function as above, these animals show acquisition and relearning similar to normal animals.29 Although enzyme activity was not present in the brain during testing, neurons of the cerebral cortex retained a significant reduction in lysosomal storage. The animals transplanted without myeloablation in the current study attained only 0.3% +/+ GUS activity in the brain, on average, and showed lysosomal storage similar to that in untreated animals. Therefore, the restored breeding observed in these mice was unlikely due to neurologic changes in the CNS and was most likely due to peripheral systemic improvements in function and mobility.
To obtain engraftment of donor hematopoietic cells without ablation, very high cell dosage must be used. The 3xBMT recipients were injected with an equivalent of 3 × 1010cells/kg, which translates to an average human neonate of 3.5 kg receiving 1.05 × 1011 cells total. When performing myeloablative murine adult BMT, 4-8 × 107 cells/kg easily provide 100% donor engraftment. To obtain high levels of engraftment in myeloablated children, 5 to 10 × 108cells/kg are required and higher doses in this setting lead to greater success.10 Although overall expansion of donor cells in the nonablated mouse neonates was modest, it may be possible to further increase expansion after engraftment. Donor fetal liver cells engraft and expand more rapidly in the neonatal setting, even when fewer cells are injected. These cells are a near equivalent to human cord blood cells and have greater repopulation potential than adult bone marrow.45 Experiments are currently underway to evaluate the therapeutic contribution of these cells as compared to adult bone marrow.
In addition to manipulation of cell numbers and ontologic source to improve nonablative transplantation, other techniques may be applicable. Recent studies demonstrate that sublethal ablative regimens that establish mixed chimerism can be manipulated to enhance engraftment by donor lymphocyte infusions (DLI).46 This technique not only improves outcome of allogeneic transplantation by reducing mortality, but it also allows a graft-versus-leukemia reaction concomitant with a reduction in graft-versus-host disease (GVHD).47 48 Patients with genetic disorders that respond to BMT therapy, as in the mucopolysaccharidoses, will often be treated with allogeneic marrow because of the difficulty in finding a compatible donor. Therefore, we wish to test nonablative neonatal transplantation of allogeneic cells in the MPS VII model, followed by DLI, to test for amplified engraftment and reduction of GAG storage without GVHD. If this treatment is successful by the current criteria, these animals will be tested for improved learning and behavioral modifications to demonstrate amelioration of cognitive defects after long-term engraftment.
We thank Dr Edward H. Leiter for critical review of the manuscript and Brenda Sprague for mouse colony maintenance. We thank Dr Wesley G. Beamer for helpful discussions and equipment training regarding bone morphologic analysis with pQCT. We also thank Dr William S. Sly for generously supplying recombinant murine β-glucuronidase.
Supported by National Research Service Award DK09570 to B.W.S., National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) RO1s DK41082, 27726, and 49525 to J.E.B., and Cancer Center support grant CA34196 to The Jackson Laboratory.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Brian W. Soper, The Jackson Laboratory, Box 95, 600 Main St, Bar Harbor, ME 04609; e-mail: bws@jax.org.