Abstract
The malarial protein Plasmodium falciparum erythrocyte membrane protein 1 (PfEMP1) is a parasite protein that is exported to the surface of the infected erythrocyte, where it is inserted into the red cell cytoskeleton in the second half of the parasite life cycle. The surface expression of PfEMP1 coincides with the occurrence of the adhesion of infected erythrocytes to vascular endothelium. This protein has been shown to interact with CD36, intercellular adhesion molecule-1 (ICAM-1) and chondroitin sulfate A (CSA). In this study, it is demonstrated by affinity purification and western blot analysis that PfEMP1 also functions as a cell surface ligand for P-selectin, an adhesion molecule that has been shown to mediate the rolling of infected erythrocytes under physiologic flow conditions, leading to a significant increase in adhesion to CD36 on activated platelets and microvascular endothelium.
Introduction
There has been increasing recognition thatPlasmodium falciparum–infected erythrocytes interact with a number of adhesion molecules on microvascular endothelium in a synergistic fashion under flow conditions.1-5 Infected erythrocytes tether and roll on CD36, intercellular adhesion molecule-1 (ICAM-1), P-selectin, and vascular cell adhesion molecule-1 (VCAM-1), whereas adhesion is mainly to CD36. In both in vitro and in vivo models, there is strong evidence that the rolling interaction enhances subsequent adhesion, either by increasing the arrest of rolling cells (ICAM-1 and VCAM-1),4,5 or by actually increasing the number of infected erythrocytes recruited from the bloodstream to vascular endothelium (P-selectin).4 These findings suggest that P-selectin may play an important role in the recruitment of infected erythrocytes in the pathogenesis of severe falciparum malaria.
P-selectin is a 140 kd glycoprotein that has a critical role in leukocyte recruitment in mediating the initial tethering of leukocytes to endothelium through interaction with its natural ligand P-selectin glycoprotein ligand-1 (PSGL-1) on leukocytes.6,7 The extracellular domain of P-selectin consists of a lectin domain, EGF domain, and 9 short consensus repeats. In previous studies, we have shown that as with leukocytes, infected erythrocyte rolling on immobilized P-selectin is Ca++-dependent, and neuraminidase- and trypsin-sensitive.3Furthermore, rolling can be competitively inhibited by the sulfated fucose polymer fucoidin and the tetrasaccharide sialyl Lewis x (sLex). These findings suggest that infected erythrocytes may interact with the lectin domain of P-selectin via a sialylated glycoprotein expressed on the surface of infected erythrocytes.
The parasite protein Plasmodium falciparum erythrocyte membrane protein 1 (PfEMP1) is expressed on the surface of infected erythrocytes in the second half of the parasite life cycle.8 It is a variant protein both in terms of antigenicity9 and molecular size.8 The protein is encoded by a large family of vargenes.10,11 The molecule consists of 2 to 7 Duffy-binding like (DBL) domains, each containing 5 consensus motifs rich in cysteine residues. There is a single transmembrane domain, followed by an intracytoplasmic domain. Different domains on PfEMP1 have been shown to bind CD36,12 ICAM-1,13 and chondroitin sulfate A.14 In the present report, we investigated whether PfEMP1 binds P-selectin.
Materials and methods
Materials
Unless otherwise stated, all reagents for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and blots were obtained from Sigma-Aldrich (Oakville, ON, Canada) or Bio-rad Laboratories (Hercules, CA). Recombinant human P-selectin (rhP-selectin) was purchased from R&D Systems (Minneapolis, MN). An anti-protease cocktail (Complete) was purchased from Boehringer Mannheim Biochemicals (Indianapolis, IN). Molecular mass standards and enhanced chemoluminescence (ECL) western blotting detection reagents were obtained from Amersham Pharmacia Biotech (Piscataway, NJ). Immobilon-P (polyvinylidene fluoride [PVDF] membrane) was purchased from Millipore (Bedford, MA). Neuraminidase fromVibrio cholerae was obtained from Calbiochem (La Jolla, CA). sLex was a kind gift of Dr R. Larsen, Glycomed, Alameda, CA.
Antibodies and immunoblotting
The acidic terminal segment (ATS) is a very conserved region of the putative intracellular domain of PfEMP1. An affinity-purified anti-PfEMP1 polyclonal antibody directed against a fusion protein glutathione s-transferase (GST)–ATS was prepared and extensively characterized as described.15 A commercial affinity-purified polyclonal anti–P-selectin antibody was purchased from Pharmingen (San Diego, CA). SDS-PAGE and western blotting with anti-ATS and rhP-selectin was performed as described.7 15
PfEMP1 extraction
SDS soluble PfEMP1 extracts were prepared from a clinical isolate of P falciparum (P98-23) as previously described.16 A membrane extract of infected erythrocytes from a laboratory parasite line D10, which did not roll on immobilized rhP-selectin in a flow chamber assay,3 was prepared by the same protocol and used as a control. An extract from normal group O erythrocytes was also prepared by ultracentrifugation (100 000g) at all steps of the procedure.
Preparation of anti-ATS–coated sepharose beads
Cyanogen bromide–activated sepharose beads (Pharmacia Biotech, Upsala, Sweden) coated with bovine serum albumin (BSA) or anti-ATS antibody were prepared by adding 50 mL of 1 mM HCl to 0.5 g beads and rocked for 15 minutes. The beads were spun and the supernatant removed. Three mL of anti-ATS at 0.4 mg/mL was added to 1.5 mL beads and mixed on a rotating wheel at 4°C for 2 hours. The supernatant was removed and measured at 280 nm in a spectrophotometer to ensure that 70% to 90% of the antibodies had bound to the beads. Five mL of 0.1 M Tris buffer, pH 8.0 was added to the antibody-coated beads and put on a rotating wheel for 2 hours at 4°C. The beads were spun and the supernatants removed. The beads were washed alternately with 5 mL 0.1 M Na acetate, pH 4, in 0.5 M NaCl and 5 mL 0.1 M Tris-HCl, pH 4.0, in 0.5 M NaCl for a total of 6 washes. Phosphate-buffered saline (PBS) was added at a 1-to-1 ratio and a final NaN3 concentration of 0.05%. Control beads coated with BSA at 5 mg/mL were prepared by the same protocol.
Affinity purification of P-selectin with immobilized PfEMP1 on anti-ATS sepharose beads
Twenty μL sepharose 4B beads coated with BSA were incubated with 100 μL of PfEMP1 extract diluted 1-to-10 in TPP (1% Triton X-100 in PBS with protease inhibitor “Complete”) for 2 hours at 4°C on a rotating device (Thermolyne, Labuquake-Barnstead, Dubuque, IA). This step was done to absorb out any nonspecific protein–protein interactions. Subsequently, the beads were pelleted and the supernatant was incubated with 20 μL of anti-ATS–sepharose beads for 2 hours at 4°C on a rotating device. This step was followed by pelleting the beads, washes with TPP, and incubation with 400 μL rhP-selectin at 2 μg/mL of a buffer containing 20 mM Tris, 150 mM NaCl, 1 mM CaCl2, pH 7.4,10 for 2 hours at 4°C. Beads were washed and 20 μL beads were loaded with an equal volume of 2x sample buffer in each lane of an SDS-PAGE gel. After transfer, the blot was probed with anti–P-selectin followed by peroxidase-labeled secondary antibody and ECL system.
Neuraminidase digestion
The interaction of P-selectin with its natural ligand PSGL-1 on leukocytes is sialic acid–dependent. To determine if P-selectin recognition of PfEMP1 also requires sialyl residues, immobilized PfEMP1 was digested at 37°C for 1 hour with neuraminidase from V cholerae at a final concentration of 0.5 U/mL in 50mM sodium acetate, pH 5.5, 154 mM NaCl, 9 mM CaCl2, and 25 μg/mL human serum albumin (HSA). A second aliquot of immobilized PfEMP-1 was treated with the buffer alone. After extensive washes to remove the enzyme, the beads were incubated with rhP-selectin and then probed with anti–P-selectin.
To determine if the neuraminidase treatment produced any change in the size or amount of PfEMP1 protein, enzyme-treated immobilized PfEMP1 on anti-ATS beads was run on an SDS-PAGE gel. A second lane in the gel contained an aliquot of immobilized PfEMP1 treated with the buffer alone. After transfer, the blot was probed with anti-ATS and developed with ECL. The bands were scanned and individual bands were quantified after background subtraction using the National Institutes of Health (NIH) Image 1.61 program.
Competitive inhibition of P-selectin binding by sLex and fucoidin
To further characterize the interaction between PfEMP1 and P-selectin, 10 μL anti-ATS beads with immobilized PfEMP1 were incubated with rhP-selectin (2 μg/mL) in the presence or absence of 2.5 mM sLex, or in the presence of fucoidin or dextran. The beads were then pelleted, washed, and analyzed by SDS-PAGE and immunoblotting.
Results
Detection of PfEMP1 with anti-ATS
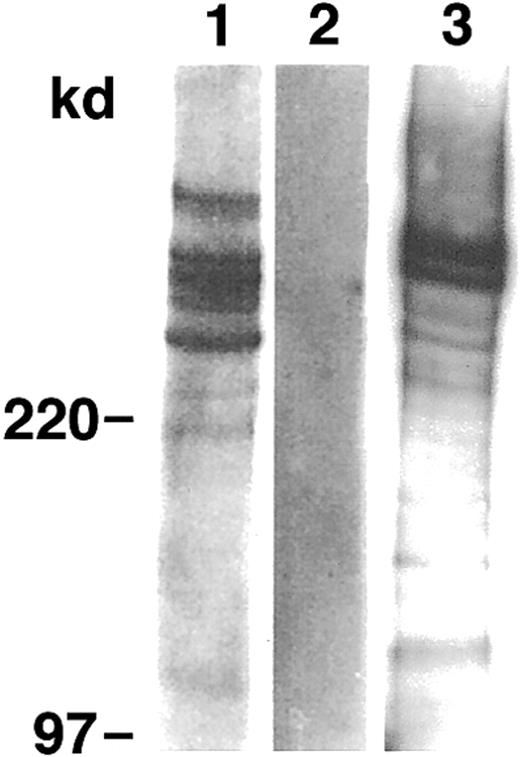
The PfEMP1 extracts from the parasite isolate P98-23 and parasite line D10 were electrophoretically separated by means of SDS-PAGE and transferred to immobilon-P. Probing with anti-ATS revealed a number of prominent bands above 220 kd (Figure 1). Weaker bands of approximately 220 kd were also seen. The bands were specific for parasite proteins, as none of them was present in the extract of noninfected erythrocytes.
PfEMP1 is present on membrane extracts of infected but not normal erythrocytes.
Western blot analysis of SDS extracts of P98-23 (lane 1), noninfected erythrocytes (lane 2), and D10 (lane 3) by anti-ATS. Bands of approximately 220 kd and above were seen only in the extracts from infected erythrocytes.
PfEMP1 is present on membrane extracts of infected but not normal erythrocytes.
Western blot analysis of SDS extracts of P98-23 (lane 1), noninfected erythrocytes (lane 2), and D10 (lane 3) by anti-ATS. Bands of approximately 220 kd and above were seen only in the extracts from infected erythrocytes.
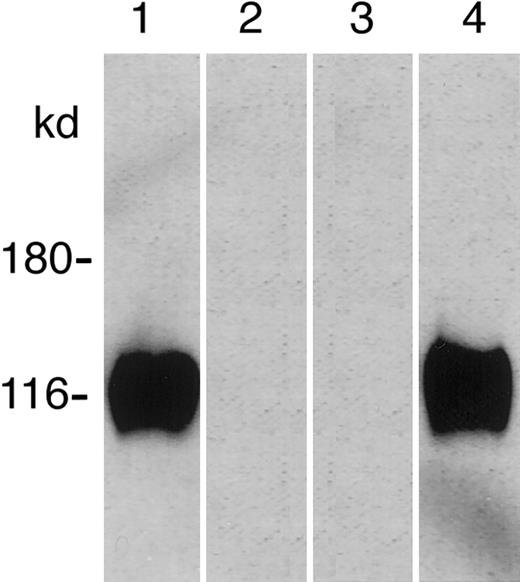
Affinity purification of P-selectin by PfEMP1
PfEMP1 bound to anti-ATS–coated beads were allowed to interact with rhP-selectin. As controls, extracts of infected eythrocytes from a noninteracting line D10 and noninfected erythrocytes were subjected to the same experimental procedure. During the subsequent SDS-PAGE, a protein of approximately 120 kd was solubilized from only the PfEMP1 beads from P98-23 which, on probing with anti–P-selectin, comigrated with rhP-selectin (Figure 2). No protein was solubilized from anti-ATS beads incubated with an extract of D10 or noninfected erythrocytes, although PfEMP1 was detectable in the D10 lane when probed with anti-ATS (data not shown).
Affinity purification of recombinant human P-selectin (rhP-selectin) with immobilized PfEMP1.
Soluble rhP-selectin was incubated with bead-immobilized PfEMP1. Binding of rhP-selectin was detected by western blotting (lane 1), and the solubilized protein comigrated with rhP-selectin (lane 4). No band was seen with the extracts from the noninteracting parasite line D10 (lane 2) or noninfected erythrocytes (lane 3).
Affinity purification of recombinant human P-selectin (rhP-selectin) with immobilized PfEMP1.
Soluble rhP-selectin was incubated with bead-immobilized PfEMP1. Binding of rhP-selectin was detected by western blotting (lane 1), and the solubilized protein comigrated with rhP-selectin (lane 4). No band was seen with the extracts from the noninteracting parasite line D10 (lane 2) or noninfected erythrocytes (lane 3).
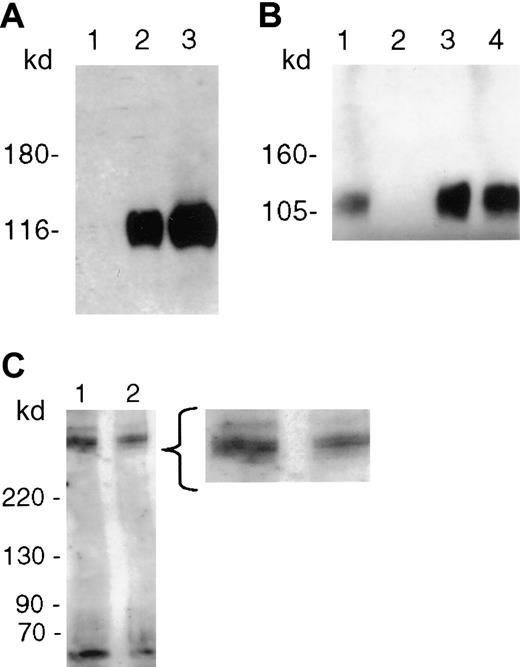
P-selectin recognizes a neuraminidase-sensitive determinant
Pretreatment of immobilized PfEMP1 with neuraminidase from V cholerae, which specifically cleaves both α2-3 and α2-6 linkages of sialic acids, totally abolished the binding of rhP-selectin (Figure 3A). Furthermore, immobilized PfEMP1 did not bind P-selectin in the presence of fucoidin (Figure 3B) or sLex(data not shown), but binding was not affected by an irrevelant sugar dextran. Neuraminidase-treated immobilized PfEMP1 separated on SDS-PAGE gel displayed the same bands as buffer-treated protein on probing with anti-ATS (Figure 3C). Densitometry readings of the major bands were 194.88 (doublet) and 206.12 in the enzyme-treated lane, and 213.31 (doublet) and 225.39 in the buffer-treated lane. The 9% difference after subtracting for background is within experimental variation in the amount of beads loaded in the 2 lanes.
PfEMP1 binding to recombinant human P-selectin (rhP-selectin) is inhibited by neuraminidase and fucoidin.
(A) PfEMP1 immobilized on anti-ATS beads was digested with neuraminidase (0.5 U/mL) from V cholerae for 1 hour at 37°C prior to incubation with the soluble receptor. The enzyme treatment completely abrogated its interaction with P-selectin (lane 1). Binding of rhP-selectin was seen when PfEMP1 was treated with the neuraminidase buffer alone (lane 2), and the solubilized receptor protein comigrated with rhP-selectin (lane 3). (B) PfEMP1 immobilized on ATS beads were incubated with rhP-selectin in the absence (lane 1) or presence (lane 2) of fucoidin 100 μg/mL. Fucoidin competitively inhibited the binding of rhP-selectin to PfEMP1. An unrelated sugar dextran had no effect on the binding of rhP-selectin (lane 3). The solubilized receptor protein comigrated with rhP-selectin (lane 4). (C) PfEMP1 immobilized on anti-ATS beads were incubated with 0.5 U/mL neuraminidase (lane 2) or with the buffer alone (lane 1) for 1 hour at 37°C. Treated beads were extensively washed and run on an SDS-PAGE gel for 30 minutes and blotted as described. The blot was probed using anti-ATS. There was no significant difference in the molecular size or thickness of the bands in the 2 lanes by densitometry.
PfEMP1 binding to recombinant human P-selectin (rhP-selectin) is inhibited by neuraminidase and fucoidin.
(A) PfEMP1 immobilized on anti-ATS beads was digested with neuraminidase (0.5 U/mL) from V cholerae for 1 hour at 37°C prior to incubation with the soluble receptor. The enzyme treatment completely abrogated its interaction with P-selectin (lane 1). Binding of rhP-selectin was seen when PfEMP1 was treated with the neuraminidase buffer alone (lane 2), and the solubilized receptor protein comigrated with rhP-selectin (lane 3). (B) PfEMP1 immobilized on ATS beads were incubated with rhP-selectin in the absence (lane 1) or presence (lane 2) of fucoidin 100 μg/mL. Fucoidin competitively inhibited the binding of rhP-selectin to PfEMP1. An unrelated sugar dextran had no effect on the binding of rhP-selectin (lane 3). The solubilized receptor protein comigrated with rhP-selectin (lane 4). (C) PfEMP1 immobilized on anti-ATS beads were incubated with 0.5 U/mL neuraminidase (lane 2) or with the buffer alone (lane 1) for 1 hour at 37°C. Treated beads were extensively washed and run on an SDS-PAGE gel for 30 minutes and blotted as described. The blot was probed using anti-ATS. There was no significant difference in the molecular size or thickness of the bands in the 2 lanes by densitometry.
Discussion
In this report, we demonstrated that PfEMP1 from a clinicalP falciparum isolate can function as a ligand for P-selectin. The specific affinity of the receptor molecule to the beads with immobilized PfEMP1 and the absence of any effect in all controls were clear and direct proof of the receptor–ligand interaction. This was further supported by corroborating evidence such as the abrogation of the P-selectin–PfEMP1 interaction by the sulfated glycoconjugate fucoidin, which inhibits the binding of PSGL-1 on leukocytes to the lectin domain of P-selectin.17
Our data also suggest that sialic acid residues may possibly be involved in the interaction between PfEMP1 and P-selectin. However, glycosylation by P falciparum remains controversial.18 Currently there is no evidence that the parasite either makes sialic acid or possesses trans-sialidase activity.19,20 Conclusive evidence for the involvement of sialic acid residues can be obtained only when a single PfEMP1 species that binds P-selectin is cloned and sialylation identified. In our experiments, we have not exhaustively excluded a nonspecific effect of neuraminidase on PfEMP1 or the binding domain for P-selectin on the protein. It is also conceivable that a sialoglycoprotein of host cell origin could have coprecipitated with the PfEMP1 in our experiments, and mediated the interaction with P-selectin. Host cell proteins by themselves were obviously insufficient to mediate binding of P-selectin, as a membrane extract of noninfected erythrocytes did not bind any receptor protein, and noninfected erythrocytes did not roll on immobilized P-selectin in a flow chamber assay.3Similarly, the membrane extract of the parasite line D10, which did not roll on immobilized P-selectin in the flow chamber assay, also did not bind P-selectin.
The dissociation constant (Kd) of P-selectin for sLex is in the millimolar range.21 Despite the low affinity and high on-off rate, this interaction is critical for the initial tethering and rolling of leukocytes on venular endothelium. The binding affinity of P-selectin for PfEMP1 is not known. However, the interaction between P falciparum–infected erythrocytes and P-selectin under physiologic flow conditions is also one of tethering and rolling.3 As in the case of leukocytes, these adhesive interactions are physiologically significant. On oncostatin M–stimulated human dermal microvascular endothelium, the induction of P-selectin expression resulted in an increase in the number of rolling cells in a parallel plate flow chamber, leading to an increase in the number of adherent cells to CD36.4 Recent investigations by intravital microscopy in a human/severe combined immunodeficiency (SCID) mouse chimera model confirmed that rolling and adhesion of infected erythrocytes do indeed occur in a human microvasculature in vivo, and that the rolling component can enhance subsequent adhesion.5 In leukocyte recruitment, the steps of the adhesive cascade are restricted, inasmuch as no adhesion occurs unless there is prior rolling on selectins22 or VCAM-1.23 In comparison, infected erythrocytes can roll on a number of adhesion molecules all of which can contribute to the eventual arrest of the cells on CD36. Understanding the molecular basis of the interaction between the parasite and each of the adhesion molecules will provide crucial information for the design of antiadhesive therapy.
The authors would like to thank Tim Byrne, Division of Infections and Immunity, The Walter and Eliza Hall of Medical Research, Melbourne, Australia, for technical assistance; and Dr Dror I. Baruch, Laboratory for Parasitic Diseases, National Institutes of Health, for helpful discussions.
Supported by the Canadian Institutes of Health Research and the Alberta Heritage Foundation for Medical Research, Alberta, Canada.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
May Ho, Department of Microbiology and Infectious Diseases, 3330 Hospital Dr NW, Calgary, Alberta, Canada T2N 4N1; e-mail: mho@ucalgary.ca.