Abstract
Acute thrombocytopenia is a recognized complication of treatment with GPIIb/IIIa inhibitors whose cause is not yet known. We studied 9 patients who developed severe thrombocytopenia (platelets less than 25 × 109/L) within several hours of treatment with the GPIIb/IIIa inhibitors tirofiban (4 patients) and eptifibatide (5 patients). In each patient, acute-phase serum contained a high titer (range, 1:80-1:20 000) IgG antibody that reacted with the glycoprotein IIb/IIIa complex only in the presence of the drug used in treatment. Four patients had been previously treated with the same drug, but 5 had no known prior exposure. Pretreatment serum samples from 2 of the latter patients contained drug-dependent antibodies similar to those identified after treatment. No tirofiban- or eptifibatide-dependent antibodies were found in any of 100 randomly selected healthy blood donors, and only 2 of 23 patients receiving tirofiban or eptifibatide who did not experience significant thrombocytopenia had extremely weak (titer, 1:2) tirofiban-dependent antibodies. In preliminary studies, evidence was obtained that the 9 antibodies recognize multiple target epitopes on GPIIb/IIIa complexed with the inhibitor to which the patient was sensitive, indicating that they cannot all be specific for the drug-binding site. The findings indicate that acute thrombocytopenia after the administration of tirofiban or eptifibatide can be caused by drug-dependent antibodies that are “naturally occurring” or are induced by prior exposure to drug. These antibodies may be human analogs of mouse monoclonal antibodies that recognize ligand-induced binding sites (LIBS) induced in the GPIIb/IIIa heterodimer when it reacts with a ligand-mimetic drug.
Introduction
The glycoprotein IIb/IIIa (GPIIb/IIIa, αIIbβ3 integrin) inhibitors are a new class of antithrombotic agents that act competitively to inhibit the binding of fibrinogen to the platelet fibrinogen receptor, thereby inhibiting platelet–platelet interaction essential for the formation of platelet thrombi.1-3 In clinical trials, these agents have been proved effective in reducing secondary complications following percutaneous transluminal coronary angioplasty.4-6 Three GPIIb/IIIa inhibitors—abciximab,1tirofiban,7 and eptifibatide8—are approved in the United States for specific clinical indications. In each clinical trial of these agents, a subset of treated patients experienced acute, severe thrombocytopenia, often with bleeding symptoms, during or shortly after treatment.9-11 Severe thrombocytopenia in a patient whose remaining platelets are functionally compromised by a GPIIb/IIIa inhibitor increases the risk for serious bleeding, and it is important that its etiology be clarified. We studied 9 patients who experienced severe thrombocytopenia (platelets less than 25 × 109/L) after receiving tirofiban or eptifibatide and obtained evidence that platelet destruction was caused by drug-dependent IgG antibodies that can be naturally occurring. It may be possible to identify patients at risk for this complication by screening for such antibodies before treatment.
Materials and methods
Reagents
Bovine serum albumin (BSA), Tris, diethanolamine, Tween 20, HEPES (N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid), and PGE1 were purchased from Sigma Chemical (St Louis, MO). Para-nitro phenyl phosphate (pNPP) was from Zymed Laboratories (South San Francisco, CA). Tirofiban (Aggrastat) and eptifibatide (Integrelin) were purchased from a pharmacy. Abciximab was supplied by Centocor (Malvern, PA). Orbofiban and xemilofiban were from G.D Searle (Skokie, IL). Fluorescein isothiocyanate (FITC)–conjugated, affinity-purified F(ab')2 goat anti-human IgG heavy- and light-chain–specific (H&L), FITC-conjugated, affinity-purified F(ab')2 goat anti-human IgG Fc, FITC-conjugated F(ab')2 donkey anti-human IgM Fc, and alkaline phosphatase (AP)–conjugated, affinity-purified F(ab')2 goat anti-human IgG (H&L) were from Jackson Immunoresearch Laboratory (West Grove, PA). Monoclonal antibody AP3 (anti-GPIIIa) was from Dr Peter J. Newman (Blood Center of Southeastern Wisconsin, Milwaukee).
Isolation of platelets
Normal donor platelets were isolated from blood anticoagulated with acid citrate dextrose as previously described,12washed in Ringer citrate dextrose, pH 6.5, and, unless otherwise indicated, suspended in HEPES buffer (10 mM HEPES, 0.145 M NaCl, 1 mM CaCl2, 1% BSA, pH 7.05) for serologic studies.
Flow cytometric detection of drug-dependent antibodies
The assay has been described previously.12 13 Ten microliters patient serum was incubated with 5 × 106platelets suspended in 40 μL HEPES buffer containing tirofiban (1 μg/mL) or eptifibatide (2 μg/mL). After incubation for 1 hour at room temperature, the platelets were washed twice in HEPES buffer containing drug at the same concentration as in the primary mixture. The washed platelet button was suspended in 25 μL HEPES containing drug at twice the concentration used in the primary mixture, and 25 μL fluorescein-labeled anti-immunoglobulin antibody (H&L chain-specific), diluted 1:40, was added. After incubation in the dark for 45 minutes, an aliquot was diluted in 0.5 mL HEPES, and platelet-bound fluorescence was analyzed by flow cytometry (FACScan; Becton Dickinson). A positive reaction was defined as mean platelet fluorescence intensity at least twice that of platelets processed identically, except for the absence of drug. This value always exceeded the value obtained with normal serum plus drug or patient serum in the absence of drug by at least 3 standard deviations.
Antigen-capture ELISA
The method has been described previously.12,14 In brief, washed platelets were solubilized with 1% Triton X-100 detergent, and the GPIIb/IIIa complex was captured in wells containing 0.5 μg fixed monoclonal antibody AP3-specific for GPIIIa. AP3 has been shown not to interfere with the binding of ligands to GPIIb/IIIa.15 Binding of patient antibodies to the immobilized GPIIb/IIIa in the presence and absence of drug at 1 μg/mL (tirofiban) or 2 μg/mL (eptifibatide) was determined by enzyme-linked immunosorbent assay (ELISA) using alkaline phosphatase-conjugated goat F(ab')2 specific for human IgG (H&L) chains. As with the flow cytometric assay, drug was used in all wash solutions and subsequent reaction steps at the same concentration used in the primary reaction mixture.
Results
Clinical and routine laboratory findings
Clinical and laboratory findings in 9 patients who experienced thrombocytopenia within 24 hours of treatment with tirofiban or eptifibatide are summarized in Table 1. For 5 patients, this was the first exposure to the drug; for 4, it was the second or third exposure. Patient T1, who had received tirofiban once before, had an episode of severe hypotension requiring treatment with vasopressors within 30 minutes of the start of drug infusion and subsequently developed rigor and chills. Drug infusion was stopped, and he was found to have a platelet level 2 × 109/L approximately 1 hour later. None of the other patients had acute symptoms, but they were found to be thrombocytopenic 6 to 24 hours after drug exposure. Thrombocytopenia was severe in all patients (range, 1-25 × 109/L; median, 5 × 109/L). All patients had petechial hemorrhages, and 6 experienced serious bleeding that included oozing at sites of catheterization (patients T3 and E1-E5), gastrointestinal bleeding (patient E3), urinary tract bleeding (patient E4), and fatal intrapulmonary hemorrhage (patient T3). Eight of the 9 patients were transfused with platelets, and 3 were given intravenous gamma globulin. Posttransfusion platelet count information was inadequate to judge the effectiveness of platelet transfusions in all patients. Transfusions given to patients T3, T4, E2, and E5 failed to produce a significant increase in platelet levels when repeat counts were performed 2 to 6 hours later. However, a transfusion given to patient E5 produced a rise in platelets from 5 × 109 to 20 × 109/L. All patients achieved a platelet level in excess of 100 × 109/L within 2 to 6 days of the thrombocytopenic episode except patient T3, who died.
Clinical and laboratory findings in patients with fiban-induced thrombocytopenia
| Drug . | Tirofiban . | Eptifibatide . | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Patient sample . | T1 . | T2 . | T3 . | T4 . | E1 . | E2 . | E3 . | E4 . | E5 . |
| Sex/age, y | M/78 | M/65 | M/64 | F/78 | M/73 | F/81 | F/79 | F/59 | M/65 |
| Prior drug exposure | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 2 |
| Pretreatment platelet level* | 212 | 200 | 192 | 200 | 195 | 220 | 225 | 196 | 90 |
| Platelet nadir* | 1 | 19 | 5 | 2 | 25 | 9 | 2 | 5 | 2 |
| Days to reach 100* | 4 | 3 | Died | > 5 | 4 | 3 | 4 | > 5 | > 5 |
| Platelet transfusion | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes |
| Antibody Ig class | IgG | IgG | IgG | IgG/M (w) | IgG | IgG | IgG | IgG/M (w) | IgG |
| Antibody titer | 1:1 280 | 1:80 | 1:1 000 | 1:10 000 | 1:20 000 | 1:5 000 | 1:1 280 | 1:5 000 | 1:320 |
| Drug . | Tirofiban . | Eptifibatide . | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Patient sample . | T1 . | T2 . | T3 . | T4 . | E1 . | E2 . | E3 . | E4 . | E5 . |
| Sex/age, y | M/78 | M/65 | M/64 | F/78 | M/73 | F/81 | F/79 | F/59 | M/65 |
| Prior drug exposure | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 2 |
| Pretreatment platelet level* | 212 | 200 | 192 | 200 | 195 | 220 | 225 | 196 | 90 |
| Platelet nadir* | 1 | 19 | 5 | 2 | 25 | 9 | 2 | 5 | 2 |
| Days to reach 100* | 4 | 3 | Died | > 5 | 4 | 3 | 4 | > 5 | > 5 |
| Platelet transfusion | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes |
| Antibody Ig class | IgG | IgG | IgG | IgG/M (w) | IgG | IgG | IgG | IgG/M (w) | IgG |
| Antibody titer | 1:1 280 | 1:80 | 1:1 000 | 1:10 000 | 1:20 000 | 1:5 000 | 1:1 280 | 1:5 000 | 1:320 |
Platelet level in thousands per microliter. w indicates weak.
Platelet-reactive, drug-dependent antibodies were identified in each patient
Samples tested for drug-dependent antibodies were obtained within 1 to 3 days of the onset of thrombocytopenia. Reactions of representative patient samples against normal target platelets with and without drug are illustrated in Figure 1. Each of the 9 patients had an IgG antibody that reacted with normal platelets in the presence, but not in the absence, of the GPIIb/IIIa inhibitor they were receiving when thrombocytopenia developed. Weak IgM drug-dependent antibodies were also detected in patients T4 and E4. The titer of the IgG drug-dependent antibodies in the flow cytometric assay ranged from 1:80 (patient T2) to 1:20 000 (patient E1) (Table 1). During treatment with eptifibatide and tirofiban, plasma levels of drug range from 0.4 to 2.2 μg/mL with eptifibatide and are approximately 0.05 μg/mL with tirofiban.16 Antibodies from the 9 patients could be detected at concentrations significantly lower than these values (0.2 μg/mL eptifibatide and 0.005 μg/mL tirofiban). Because each patient received unfractionated heparin, the patient samples were tested for heparin-induced antibodies by solid-phase PF4 ELISA.17 All gave negative results.
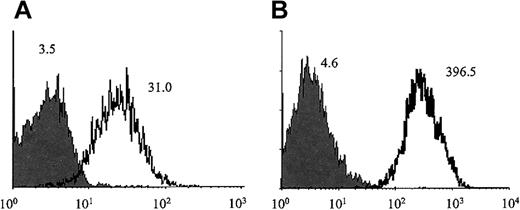
Patient plasma samples contained drug-dependent antibodies.
Plasma from patients T1 (A) and E1 (B) contained IgG antibodies that reacted with platelets only in the presence of the GPIIb/IIIa inhibitors tirofiban and eptifibatide, respectively (open histograms). No reaction was obtained in the absence of drug (closed histograms) or with normal serum in the presence or absence of drug (not shown). Serum from the other 7 patients gave reactions similar to those shown for patients T1 and E1.
Patient plasma samples contained drug-dependent antibodies.
Plasma from patients T1 (A) and E1 (B) contained IgG antibodies that reacted with platelets only in the presence of the GPIIb/IIIa inhibitors tirofiban and eptifibatide, respectively (open histograms). No reaction was obtained in the absence of drug (closed histograms) or with normal serum in the presence or absence of drug (not shown). Serum from the other 7 patients gave reactions similar to those shown for patients T1 and E1.
Patients T2 and E1 had drug-dependent antibodies in pretreatment samples
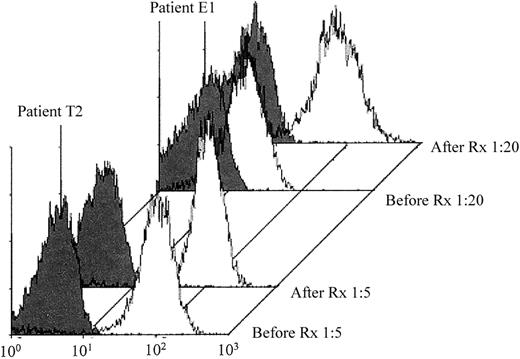
Because patients T1, T4, E4, and E5 experienced thrombocytopenia after their second or third exposure to a GPIIb/IIIa inhibitor, it seemed likely that their antibodies were induced by prior exposure to the drug. However, the remaining 5 patients had no known previous exposure to this class of drugs. Serum samples obtained before treatment were available from patients T2 and E1. As shown in Figure2, drug-dependent antibodies, comparable in strength to those identified after thrombocytopenia developed, were present in each of the pretreatment samples.
Fiban-dependent antibodies were identified in pretreatment serum samples from patients T2 and E1.
Reactions of pretreatment (Before Rx) and posttreatment (After Rx) serum with platelets in the presence of drug are shown by open histograms. Negative reactions in the absence of drug are shown by the closed histograms.
Fiban-dependent antibodies were identified in pretreatment serum samples from patients T2 and E1.
Reactions of pretreatment (Before Rx) and posttreatment (After Rx) serum with platelets in the presence of drug are shown by open histograms. Negative reactions in the absence of drug are shown by the closed histograms.
Antibodies were specific for the GPIIb/IIIa complex
Each of the 9 patient antibodies reacted in the presence of drug, but not in its absence, with GPIIb/IIIa complexes immobilized in microtiter wells by capture with the GPIIIa-specific monoclonal antibody AP3 (Figure 3). Antibody titers in the solid-phase assay were comparable to those obtained against intact platelets by flow cytometry (Table 1). The reactions were abolished when the target GPIIb/IIIa complex was dissociated by the prior addition of ethylenediaminetetraacetic acid (EDTA) to the wells. Specificity for GPIIb/IIIa was further documented by failure of the antibodies to react in the presence of drug with platelets from a patient with type 1 Glanzmann thrombasthenia (data not shown).
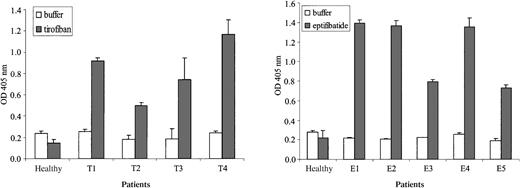
Antibodies recognized isolated GPIIb/IIIa in the presence of drug.
Monoclonal antibody AP3, specific for GPIIIa, was used to capture the GPIIb/IIIa complex from platelet lysate, and serum was incubated with this target in the presence (shaded bars) and absence (open bars) of drug. Brackets indicate 1 SD of triplicate experiments.
Antibodies recognized isolated GPIIb/IIIa in the presence of drug.
Monoclonal antibody AP3, specific for GPIIIa, was used to capture the GPIIb/IIIa complex from platelet lysate, and serum was incubated with this target in the presence (shaded bars) and absence (open bars) of drug. Brackets indicate 1 SD of triplicate experiments.
Only 1 of 9 antibodies cross-reacted with other GPIIb/IIIa inhibitors
Tirofiban-dependent antibodies from patients T1 to T4 failed to react with intact platelets in the presence of eptifibatide, and the eptifibatide-dependent antibodies from patients E1, E2, E3, and E5 were negative with tirofiban. However, the eptifibatide-dependent antibody from patient E4 gave weak but reproducible reactions against platelets pretreated with the GPIIb/IIIa inhibitors xemilofiban,18orbofiban,19 and tirofiban. None of the antibodies reacted with abciximab1-treated platelets (data not shown).
Antibodies comparable to those detected in the 9 patients were not found in any of 100 healthy controls or in patients treated with tirofiban or eptifibatide who did not acquire thrombocytopenia
Serum samples obtained from 100 unselected blood donors (45 men, 55 women) ranging in age from 19 to 64 years (mean, 41 years) were tested for tirofiban- and eptifibatide-dependent antibodies using immobilized GPIIb/IIIa and intact platelets as targets. Drug-dependent antibodies of the IgG or IgM classes were not detected in this population. Samples from 23 patients treated with tirofiban or eptifibatide who did not become thrombocytopenic following exposure to drug were similarly studied. Two of the 23 gave weak reactions (titer, 1:2) in the presence of tirofiban but were negative with eptifibatide.
By several criteria, the antibodies appeared to recognize multiple epitopes on ligand-occupied GPIIb/IIIa
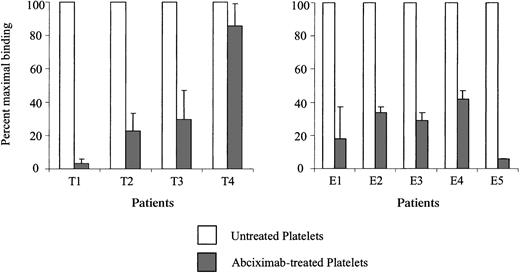
Tirofiban, eptifibatide, and other ligand-mimetic compounds react with a single site somewhere in the fibrinogen-binding pocket of the GPIIb/IIIa heterodimer.20-22 Occupancy of this site by a ligand-mimetic interferes with fibrinogen binding but does not block the binding of abciximab,22 a chimeric (human/mouse) Fab fragment derived from a murine monoclonal antibody (7E3) that binds with high affinity to a site on GPIIb/IIIa close to, but not identical with, the fibrinogen-binding site.1,23 24 We reasoned that antibodies recognizing the ligand-mimetic binding site or an epitope close to it would be blocked by abciximab, whereas those specific for a site elsewhere on GPIIb/IIIa might be unaffected. Accordingly, platelets treated with tirofiban or eptifibatide and then with a saturating quantity of abciximab were used as targets for the patients' antibodies. As shown in Figure4, the 9 antibodies differed in the extent to which they were inhibited by abciximab in that antibodies from patients T1 and E5 were blocked almost completely, those from patients T2, T3, and E1 to E4 were only partially blocked, and the tirofiban-dependent antibody from patient T4 was relatively unaffected.
Antibody binding was blocked in varying degrees by abciximab.
Healthy donor platelets were treated with tirofiban (A) or eptifibatide (B) and then with saturating quantities of abciximab. Antibody binding was then measured by flow cytometry. Values shown on the ordinate are fluorescence intensity obtained with abciximab-coated platelets (gray bars) expressed as a percentage of the signal obtained without abciximab (open bars) and represent the average of triplicate determinations. Brackets indicate 1 SD.
Antibody binding was blocked in varying degrees by abciximab.
Healthy donor platelets were treated with tirofiban (A) or eptifibatide (B) and then with saturating quantities of abciximab. Antibody binding was then measured by flow cytometry. Values shown on the ordinate are fluorescence intensity obtained with abciximab-coated platelets (gray bars) expressed as a percentage of the signal obtained without abciximab (open bars) and represent the average of triplicate determinations. Brackets indicate 1 SD.
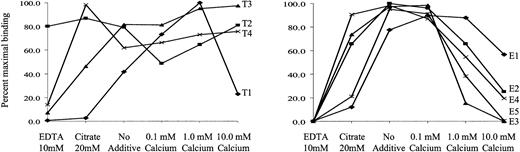
Antibodies were also heterogeneous in their reactions with platelets at different concentrations of ionized calcium. As shown in Figure5, antibody T1 reacted strongly with intact platelets at 1.0 mM Ca++ but was inhibited by 10 mM Ca++, reacted only weakly without added Ca++, and failed to react in the presence of the weak calcium chelator, citrate, or the strong chelator, EDTA. In contrast, antibody T2 reacted well at all Ca++ concentrations tested and even in the presence of EDTA. Antibodies T3 and T4 behaved like T2 except for their failure to react in EDTA. Eptifibatide-dependent antibodies differed from one another in that E1 and E5 reacted poorly in the presence of citrate and E2 to E5, but not E1, were inhibited at higher calcium concentrations. Under the conditions of these studies (room temperature, neutral pH), the GPIIb/IIIa complex remains intact even in the presence of EDTA,25,26 though it was altered structurally.27 28
Reactions of antibodies with intact platelets at various concentrations of Ca++.
Values shown (ordinate) are expressed as a percentage of the maximum fluorescence intensity obtained under the experimental conditions shown on the abscissa and represent the average of triplicate determinations that did not differ from each other by more than 15%. Reactions of tirofiban-dependent antibodies are shown in panel A, and eptifibatide dependent antibodies are shown in panel B. No reactions were obtained in the absence of drug under any of the experimental conditions (not shown).
Reactions of antibodies with intact platelets at various concentrations of Ca++.
Values shown (ordinate) are expressed as a percentage of the maximum fluorescence intensity obtained under the experimental conditions shown on the abscissa and represent the average of triplicate determinations that did not differ from each other by more than 15%. Reactions of tirofiban-dependent antibodies are shown in panel A, and eptifibatide dependent antibodies are shown in panel B. No reactions were obtained in the absence of drug under any of the experimental conditions (not shown).
Discussion
In clinical trials of tirofiban, the incidence of severe thrombocytopenia (platelets less than 50 × 109/L) ranged from 0.1% to 0.5%, approximately twice the incidence seen in patients not given the drug.5,11,29 Similar observations were made in a large-scale trial of eptifibatide.6 McClure et al30 performed a careful analysis of patients participating in the PURSUIT6 trial of eptifibatide and concluded that, though morbidity was greater in patients who acquired thrombocytopenia, the incidence of this complication was approximately the same in patients given the study drug as in patients given placebo. However, a small subset of patients given eptifibatide had profound, unexplained thrombocytopenia within a few hours of receiving the drug,30 consistent with the possibility that platelet destruction in those patients could have an immunologic basis.
The 9 patients described in this report experienced severe thrombocytopenia within a few hours of tirofiban or eptifibatide administration and, with the exception of patient T3, recovered within 2 to 6 days after the drug was discontinued. Each patient had a potent antibody that reacted with normal target platelets at a concentration of drug significantly less than that achieved in vivo during treatment at the recommended dosage. No antibodies of this type were found in any of 100 healthy subjects. Of 23 patients treated with tirofiban or eptifibatide who did not acquire thrombocytopenia (platelets less than 100 × 109/L), 21 had no detectable antibodies, and 2 had extremely weak tirofiban-dependent antibodies. One of the latter 2 patients was given tirofiban and experienced a decrease in platelet level to 110 × 109/L at 24 hours, followed by a rapid return to the normal range. The other patient received eptifibatide and maintained a normal platelet count.
Although all patients received heparin, failure to detect heparin-dependent antibodies in a highly sensitive solid-phase assay argues strongly against the possibility that heparin sensitivity affected platelets in any of the patients studied. The presence of strong drug-dependent antibodies in the patients who experienced acute thrombocytopenia after treatment with tirofiban or eptifibatide and their absence in healthy subjects and in patients treated with these drugs who maintained normal or near normal platelet levels provide evidence that platelet destruction in the 9 patients studied was antibody mediated. A preliminary report of studies in patients who developed thrombocytopenia during clinical trials of the oral ligand-mimetic drugs xemilofiban and orbofiban provides further evidence that severe thrombocytopenia can be caused by antibodies dependent on ligand-mimetic drugs for their reactions with platelets.31
Not surprisingly, antibodies from the patients studied reacted with the GPIIb/IIIa complex, to which tirofiban and eptifibatide bind specifically. We carried out preliminary studies to characterize the epitopes recognized. Drug-dependent binding of antibodies associated with quinine-induced immune thrombocytopenia fixes the drug in place on the target protein, suggesting that a trimolecular complex is formed that consists of antibody, drug, and a domain on the target.32 RGD-containing peptides and ligand-mimetic compounds appear to inhibit fibrinogen binding by reacting specifically with GPIIb/IIIa.24,33,34 Although the exact binding site has not yet been identified, it is probably the same for all ligand-mimetic GPIIb/IIIa inhibitors in view of their structural and functional similarities and the fact that they compete with each other for binding.22 A likely possibility, therefore, was that antibodies from the patients studied recognize the ligand-mimetic drug for which they are specific at its binding site on GPIIb/IIIa.
For 2 reasons, this seems unlikely. First, the antibodies differed in the extent to which their binding to GPIIb/IIIa could be blocked by abciximab (Figure 4). Antibodies from patients T1 and E5 were completely inhibited by this treatment, those from patients T2, T3, and E1 to E4 were only partially blocked, and the antibody from patient T4 was only minimally affected. Abciximab, which appears to recognize a disulfide-bonded peptide loop in GPIIIa comprised of amino acid residues 177 to 184,23 24 inhibits fibrinogen binding by steric hindrance and would be expected to block the binding of antibodies that recognize the binding site for RGD ligand-mimetic drugs. The variable effect of abciximab on binding of the 9 antibodies studied indicates they cannot all be specific for a single epitope at or near the ligand-binding site.
Second, the antibodies differed greatly in their drug-dependent binding to intact platelets at different concentrations of calcium (Figure 5). Although the structural changes that take place in GPIIb/IIIa as cation concentration is varied are only partially understood at the molecular level,21,26-28 35-37 our findings suggest that the epitopes recognized by the 9 fiban-dependent antibodies studied are expressed differently on various calcium-dependent conformers and provide further evidence that the antibodies bind to different sites on the heterodimer.
The ability of RGD peptides and ligand-mimetic compounds to induce neo-epitopes (ligand-induced binding sites [LIBS]) on various domains of the GPIIb/IIIa α and β chains28,34,38-41 suggests their binding leads to widespread conformational changes in the heterodimer. The behavior of the 9 antibodies studied is consistent with the possibility that they are specific for LIBS determinants on GPIIb/IIIa induced by tirofiban (antibodies T1-T4) and eptifibatide (antibodies E1-E5) and represent human analogues of LIBS-specific murine monoclonal antibodies. Failure of the tirofiban-dependent antibodies to cross-react with eptifibatide, and vice versa, does not rule out LIBS specificity because the set of LIBS epitopes induced in GPIIb/IIIa can vary from one ligand-mimetic compound to another.42 Even the antibodies from patients T1 and E5, which were completely blocked by abciximab (Figure 4), could be LIBS-specific because abciximab is known to block binding of a well-characterized, LIBS-specific monoclonal antibody, D3.41 Studies to determine whether these antibodies do, in fact, have LIBS specificity are in progress.
Another unique aspect of the fiban-dependent antibodies studied is that they can be naturally occurring, as illustrated by their presence in pretreatment serum samples from patients T2 and E1 (Figure 2). Bednar et al,43 in evaluating several experimental ligand-mimetic compounds in a primate model, identified 2 animals among a group of more than 100 that developed acute thrombocytopenia when challenged for the first time with drugs designated A1-L and B-P1. In serologic studies, they found that pretreatment serum from both animals contained IgG antibodies that recognized GPIIb/IIIa on human and primate platelets in the presence of the drug that provoked thrombocytopenia. The same workers screened serum samples from 1024 healthy blood donors with A1-L, B-P1, and 2 other ligand-mimetic compounds and found that 18 (1.7%) had antibodies that reacted with GPIIb/IIIa in the presence of at least 1 of the 4 drugs. Although the strength of the reactions given by the so-called normal antibodies detected was not specified, this report and our identification of strong drug-dependent antibodies in pretreatment serum from 2 patients suggest that a small subset of healthy persons has naturally occurring antibodies that recognize GPIIb/IIIa complexed with certain ligand-mimetic compounds. Our findings indicate that in some persons these antibodies can be sufficiently strong to cause thrombocytopenia on challenge with the appropriate ligand-mimetic compound. Experience with patients T1, T4, E4, and E5, and possibly with patients T3 and E3, is consistent with the possibility that first exposure to a ligand-mimetic drug can convert a weak antibody to a potent one and place a patient at risk for thrombocytopenia if challenged a second time with the same drug.
Numerous GPIIb/IIIa inhibitors are in various stages of experimental development,1-3 and it seems possible that drug-induced thrombocytopenia will be seen more often as this class of compounds becomes used more widely. Because drug-dependent antibodies specific for GPIIb/IIIa inhibitors can be readily detected in a solid-phase assay (Figure 3), it may be possible to identify patients at risk for thrombocytopenia if given a particular GPIIb/IIIa inhibitor by pretreatment screening.
Supported by grants HL44612 and HL13629 for the National Heart, Lung and Blood Institute.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Note added in proof
Recently Seiffert et al44 described 2 patients who developed severe thrombocytopenia during treatment with the experimental GPIIb/IIIa inhibitor roxifiban and provided evidence that drug-dependent antibodies were causative.
Author notes
Daniel W. Bougie, Blood Research Institute, Blood Center of Southeastern Wisconsin, PO Box 2178, Milwaukee WI 53201-2178; e-mail: dwbougie@bcsew.edu.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal