From 1994 to 2000, 154 adults with Philadelphia chromosome–positive (Ph+) and/orBCR-ABL+ acute lymphoblastic leukemia (ALL) were treated according to a prospective trial (median follow-up, 4.5 years) with the aim to study the prognostic value of early response to therapy and the role of stem cell transplantation (SCT) in first complete remission (CR). All patients received a standard induction course followed by a course of mitoxantrone and intermediate-dose cytarabine (HAM). After each course, minimal residual disease was tested by specific reverse transcriptase–polymerase chain reaction (RT-PCR) (median sensitivity, 10−5). Allogeneic SCT (if a donor) or autologous SCT (if not) was planned at 3 months in all patients in CR after HAM. CR rates after induction, after HAM, and at 3 months were 53%, 67%, and 62%, respectively. High leukocyte count and m-bcr subtype were the 2 identified bad-prognosis factors for CR at 3 months, both superseded by a poor early response assessed at day 8 of the induction course. HAM-associated salvage rate was higher in patients with M-bcr than in those with m-bcr ALL (55% vs 30%;P = .05). In the 103 patients eligible for SCT, the existence of a donor and the negative BCR-ABL status after HAM were independently predictive of remission duration (P < .001 and .01, respectively) and survival (P = .02 and .01, respectively). Relapse was the most common cause of treatment failure in all patient groups. Allogeneic SCT in first CR is the current best treatment option in adults with the disease. New strategies must be tested during early phases of therapy to increase the rate of BCR-ABL− remissions.
Introduction
The Philadelphia chromosome (Ph) is a shortened chromosome 22 generated by a balanced t(9;22) chromosomal translocation. Originally thought to be associated with chronic myelogenous leukemia (CML) only, this translocation has since been identified in other hematologic malignancies, including de novo acute lymphoblastic leukemia (ALL). The translocation results in a p210BCR-ABL fusion protein when the protooncogeneABL moves from chromosome 9 to the major breakpoint cluster region (M-bcr) within the BCR gene on chromosome 22 or in a shorter p190 BCR-ABL fusion protein when it moves to the minor breakpoint cluster region (m-bcr) within the same BCRlocus.1 The p210 protein is the usual finding in patients with CML, whereas the p190 protein is found in approximately two thirds of adults and more than 90% of children with Ph+ ALL.2,3 Both p210 and p190 transcripts can be detected by specific BCR-ABL reverse transcriptase–polymerase chain reaction (RT-PCR) assays.4
In patients with ALL, the incidence of Ph+ and/orBCR-ABL+ ALL is increasing with age from less than 5% in children to 40% in adults aged 40 years or more.5,6 In adults as well as in children with ALL, Ph+ and/or BCR-ABL+ ALL is the subgroup associated with the worst prognosis.7,8 Long-term survival rates range from 35% to 40% in children to less than 20% in adults.7-10 It has been recently suggested that the disease can be controlled by chemotherapy alone in a subgroup of young children with a low leukocyte count at diagnosis.9 In adults with the disease, it is generally thought that the only curative option is allogeneic hematopoietic stem cell transplantation (HSCT), even if this opinion is only based on small retrospective studies.10-12
Because an increased incidence of myeloid antigen expression has been observed in Ph+ and/or BCR-ABL+ALL,1,6,10 it has been suggested that cytarabine-based chemotherapy regimens might be associated with high response rates in patients with the disease. More recently, promising results have been reported in refractory and relapsing patients treated with STI571, an inhibitor of the protein-tyrosine kinase associated withBCR-ABL.13
Initial response to therapy, as assessed by early bone marrow evaluation or by the measurement of minimal residual disease during the initial phases of therapy,14-16 has been associated with outcome in prospective studies dealing with childhood ALL in general. The detection of persistent BCR-ABL fusion transcripts in patients with Ph+ and/or BCR-ABL+ALL achieving a complete remission after initial chemotherapy might thus provide useful information to stratify their prognosis.
In the present prospective multicenter study of 154 adults with Ph+ and/or BCR-ABL+ ALL, our main objectives were (1) to study the prognostic value of initial response to therapy as assessed by early bone marrow examination at day 8 of the induction course and minimal BCR-ABL residual disease evaluation after 1 and 2 courses of chemotherapy in responding patients; (2) to evaluate the value of a cytarabine-based course of consolidation/salvage; and (3) to evaluate the role of allogeneic and autologous HSCT on an intention-to-treat basis.
Patients and methods
Patient enrollment
With the exception of patients with a previous history of malignant disease including myeloproliferative syndromes, all adult patients aged 15 to 55 years with newly diagnosed, previously untreated ALL (mature B-cell ALL not included) were eligible for randomization in the LALA-94 trial. After initial randomization between idarubicin and daunorubicin for the first course of chemotherapy, they all received a common 4-drug induction course. All of them diagnosed with a Ph+ and/or BCR-ABL+ ALL within the 35 days following initial randomization were enrolled in the present study. Response and outcomes were evaluated from initial randomization in the LALA-94 trial. The study was approved by the Ethics Committee, Hospices Civils de Lyon (Lyon, France), and all patients gave signed informed consent.
Diagnosis criteria for Ph+ and/or BCR-ABL+ ALL
The diagnosis of Ph+ and/orBCR-ABL+ ALL was based on standard cytogenetic and/or on molecular analysis at baseline. Chromosomal analysis was performed using short unstimulated bone marrow and/or peripheral blood cultures. A minimum of 20 analyzed mitoses were required in each patient designated as having a normal karyotype. Standard criteria to define a clone were applied, and chromosomal abnormalities were classified according to the International System for Human Cytogenetic Nomenclature.17 Patients were also classified according to the presence or the absence of normal metaphases (NN/AN/AA) and on the basis of modal number. Patients with a modal number between 51 and 60 chromosomes were classified as hyperploid. RT-PCR specific for theBCR-ABL transcripts was performed on bone marrow and/or on peripheral blood samples at baseline as part of a systematic molecular screening for fusion transcripts (BCR-ABL, E2A-PBX1, and MLL-AF4). The RT-PCR reaction was carried out following a common protocol using already publishedBCR-ABL primers,1,18 as described.19 More recently, the standardized BIOMED-1 primers were adopted by most laboratories.20 Any sample with no amplification of the control genes (ABL orPBGD) was discarded from further analysis. The sensitivity of the reaction was assessed during each experiment using a dilution series of Tom1 (m-bcr) or K562 (M-bcr) RNA. It ranged from 10−4 to 10−6 with a median of 10−5. Each positive sample with no concordance with the karyotype was confirmed by a second analysis on another aliquot. During the first 2 years of the LALA-94 trial, quality control for RT-PCR analysis was performed on diagnosis samples. Results were retrospectively and blindly verified by a different laboratory on a second aliquot in 1 of 3 patients (n = 110 samples). All results but 2 degraded samples were concordant. The RT-PCR reaction was performed on bone marrow samples only during follow-up. Similar quality controls were performed on these remission samples with a similar concordance. Immunophenotyping of ALL cells was systematically performed on bone marrow and/or on peripheral blood samples using monoclonal antibodies against the CD2, cytoplasmic CD3, CD5, CD7; CD19, CD10, cytoplasmic CD22 or CD79a, cytoplasmic immunoglobulin μ, κ, and λ chains; CD34, CD33, CD13, CD65, HLA-DR antigens, and surface immunoglobulins. In addition, anti-CD20, anti-CD22, anti-CD3, anti-CD1a, anti-CD4, anti-CD8, anti–T-cell receptor-αβ (anti-TCRαβ), and anti-TCRγδ monoclonal antibodies were used to confirm the B-lineage or T-lineage origin of the leukemic cell population. During the study, all cytogenetic, molecular, and immunophenotyping data were prospectively and centrally reviewed by 3 distinct working committees.
Treatment
Treatment schedule and dosages are indicated in Table1. The induction course was administered over a 4-week period and consisted of prednisone, vincristine, cyclophosphamide, and daunorubicin or idarubicin according to initial randomization. On day 35, all patients with a diagnosis of Ph+ and/or BCR-ABL+ ALL were eligible for a second course of consolidation (or salvage), whatever the response to the induction course was. This consolidation/salvage course consisted of mitoxantrone and intermediate-dose cytarabine (HAM). All patients in complete remission after this HAM course were eligible for HSCT irrespective of their molecular status. Patients who did not reach a complete remission at that time were not considered for further treatment in the protocol. Based on an intention-to-treat principle, all patients eligible for HSCT were distributed in 1 of the 3 following HSCT groups: (1) matched related allogeneic bone marrow transplantation if they had a matched related donor (allo-ID group); (2) matched unrelated allogeneic bone marrow transplantation if they were considered by local investigators as eligible for such a procedure and had an identified matched unrelated donor (allo-MUD group); and (3) autologous peripheral blood stem cell transplantation if they did not meet criteria for the first 2 groups (autologous group). Identification of a matched related donor was considered at baseline in all patients. Identification of a matched unrelated donor was considered at Ph+ ALL diagnosis in all eligible patients without a related donor. A matched unrelated donor was defined as a donor-recipient pair matched for 9 or 10 of 10 HLA antigens using high-resolution molecular typing for the HLA-A, -B, -C, -DRB1, and -DQB1 genes. Transplantation was planned to be performed at 3 months (day 90 following initial randomization). Patients eligible for HSCT received 1 or 2 additional cycles of chemotherapy consisting of methotrexate and l-asparaginase. Granulocyte colony-stimulating factor–mobilized autologous peripheral blood stem cells were harvested during the myeloid recovery following the HAM consolidation course for patients in complete remission after induction and after 1 methotrexate/l-asparaginase cycle for those reaching a complete remission after HAM salvage. During the early study period, few patients (n = 6 patients) have undergone transplantation with purged autologous bone marrow stem cells. In these patients, bone marrow purging was performed by complement-dependent lysis using anti-CD19 and anti-CD20 monoclonal antibodies. For allo-ID transplantation, the prophylaxis of graft-versus-host disease was determined by local investigators but did not include T-cell depletion. For allo-MUD transplantation, the most appropriate conditioning regimen and graft-versus-host disease prophylaxis were determined by local investigators. Central nervous system prophylaxis consisted of 5 intrathecal injections. In patients with clinical or cytologic evidence of central nervous system disease at diagnosis, therapy consisted of 18 triple intrathecal injections associated a with pretransplantation 15-Gy cranial irradiation.
Treatment schedule for adult Ph+and/or BCR-ABL+ ALL: the LALA-94 trial
| Drug . | Dose . | Days . |
|---|---|---|
| Induction course | ||
| Prednisone, PO or IV | 60 mg/m2 | 1-7, 15-21 |
| Vincristine, IV | 2 mg | 1, 8, 15, 22 |
| Cyclophosphamide, IV | 750 mg/m2 | 1, 8 |
| Daunorubicin, IV* | 30 mg/m2 | 1-3, 15-16 |
| Idarubicin, IV* | 9 mg/m2 | 1-3, 8 |
| Methotrexate, IT | 15 mg | 1†, 8, 15, 22 |
| Cytosine arabinoside, IT | 40 mg | 1†, 8, 15, 22 |
| Methylprednisolone, IT | 40 mg | 1†, 8, 15, 22 |
| Consolidation/salvage course | ||
| Cytosine arabinoside, 3-h IV | 1 000 mg/m2/12 h | 1-4 (8 infusions) |
| Mitoxantrone, IV | 10 mg/m2 | 3-5 |
| Methotrexate, IT | 15 mg | 1 |
| Cytosine arabinoside, IT | 40 mg | 1 |
| Methylprednisolone, IT | 40 mg | 1 |
| Pretransplantation course(s)‡ | ||
| Methotrexate, IV | 1 500 mg/m2 | 1, +/− 15 |
| l-asparaginase, IV | 10 000 IU/m2 | 2, +/− 16 |
| HSCT1-153 | ||
| Etoposide, 6-h IV | 50 mg/kg | − 4 |
| Cyclophosphamide, 2-h IV | 60 mg/kg | − 3, − 2 |
| Total body irradiation | 12 Gy (6 fractions) or 10 Gy (1 fraction) |
| Drug . | Dose . | Days . |
|---|---|---|
| Induction course | ||
| Prednisone, PO or IV | 60 mg/m2 | 1-7, 15-21 |
| Vincristine, IV | 2 mg | 1, 8, 15, 22 |
| Cyclophosphamide, IV | 750 mg/m2 | 1, 8 |
| Daunorubicin, IV* | 30 mg/m2 | 1-3, 15-16 |
| Idarubicin, IV* | 9 mg/m2 | 1-3, 8 |
| Methotrexate, IT | 15 mg | 1†, 8, 15, 22 |
| Cytosine arabinoside, IT | 40 mg | 1†, 8, 15, 22 |
| Methylprednisolone, IT | 40 mg | 1†, 8, 15, 22 |
| Consolidation/salvage course | ||
| Cytosine arabinoside, 3-h IV | 1 000 mg/m2/12 h | 1-4 (8 infusions) |
| Mitoxantrone, IV | 10 mg/m2 | 3-5 |
| Methotrexate, IT | 15 mg | 1 |
| Cytosine arabinoside, IT | 40 mg | 1 |
| Methylprednisolone, IT | 40 mg | 1 |
| Pretransplantation course(s)‡ | ||
| Methotrexate, IV | 1 500 mg/m2 | 1, +/− 15 |
| l-asparaginase, IV | 10 000 IU/m2 | 2, +/− 16 |
| HSCT1-153 | ||
| Etoposide, 6-h IV | 50 mg/kg | − 4 |
| Cyclophosphamide, 2-h IV | 60 mg/kg | − 3, − 2 |
| Total body irradiation | 12 Gy (6 fractions) or 10 Gy (1 fraction) |
PO indicates per os; IV, intravenously; IT, intrathecally; HSCT, hematopoietic stem cell transplantation.
Patients received either daunorubicin or idarubicin, according to initial randomization.
The first IT infusion may be performed at day 1, 2, or 3.
Patients may receive 1 or 2 identical pretransplantation courses.
For allo-ID and autologous HSCT.
Criteria for response and relapse
Morphologic response was evaluated on a bone marrow aspiration and peripheral blood examination (1) after the induction course (on day 35 or after myeloid recovery); (2) after myeloid recovery following the HAM consolidation/salvage course; and (3) at 3 months (day 90 following initial randomization). Responses were classified as complete remission (CR) or failure, including resistant disease and early death. Patients were considered to be in CR when the neutrophil count was more than 1.5 × 109/L, the platelet count was more than 150 × 109/L, the result of the bone marrow examination was normal, and all extramedullary disease had resolved. In patients reaching a complete remission, molecular response was tested by RT-PCR on the bone marrow specimen of the first and second response evaluations. There was no molecular evaluation of reinfused autologous cells. All samples tested were classified asBCR-ABL+ or BCR-ABL−, whatever the sensitivity of the reaction. In addition, patients had to be classified as early responders or nonresponders on a bone marrow aspiration and peripheral blood examination performed at day 8 of the induction course. Early response was defined as the absence of peripheral blood blasts associated with fewer than 5% marrow blasts or a very hypoplastic bone marrow.
Statistical analysis
Response data were compared with the Fisher exact test for binary variables. Survival and complete remission duration data were censored at the median follow-up and estimated by the Kaplan-Meier method.21 The log-rank test22 was used to determine univariate prognostic significance of binary variables. Simultaneous effects of multiple covariates were estimated with the maximum-likelihood logistic regression model for response23 and with the Cox model for complete remission duration and survival24 and tested by the likelihood-ratio test, also used in univariate analyses for continuous variables. Survival was calculated from the date of initial randomization. To assess the prognostic significance of response to therapy and HSCT group allocation, landmark survival analyses were performed with a landmark period of 90 days.25 Estimated hazard ratios are reported as relative risks with 95% confidence intervals [CI].P values were derived from 2-sided tests.P = .05 or less was considered to indicate statistical significance. STATA statistical software (Release 7.0, Stata, College Station, TX) was used for statistical analyses.
Results
Patient characteristics
Between June 15, 1994, and February 15, 2000, 701 patients were registered in the LALA-94 trial. A total of 157 patients (22%) were diagnosed with a Ph+ and/or BCR-ABL+ALL within the 35 days following initial randomization in 154 of them. The results reported here, which are for all these 154 patients from 33 French and Belgian participating centers, are based on follow-up data as of January 1, 2002 (median follow-up, 4.5 years). Patient characteristics are indicated in Table2. There were 89 men and 65 women. The median age was 42 years (range, 17 to 56 years). Median leukocyte count was 17 × 109/L (range, 0.5 × 109/L to 626 × 109/L). Sixty-six of the 154 patients (43%) had 25 × 109/L peripheral blood white blood cells or more at diagnosis. Seven patients (5%) had evidence of central nervous system disease at baseline (diagnosed by morphologic examination of the cerebrospinal fluid and/or by the presence of clinical symptoms). All patients had B-lineage ALL. Leukemic cells expressed the CD34 antigen in 134 (89%) of the 151 cases tested. The expression of a myeloid marker was less common (15% and 20% for the CD33 and CD13 antigen, respectively). Overall, the incidence of CD34+CD33+CD13+ ALLs was 10 (7%) of 150 informative cases. No differences in baseline characteristics were observed between the 2 randomization groups (idarubicin or daunorubicin).
Patient characteristics
| Characteristic . | Result . |
|---|---|
| Baseline characteristics, N = 154 patients | |
| Age, y | |
| Median (range) | 42 (17-56) |
| 45 y or more | 66 |
| Sex ratio, M/F | 89/65 |
| CNS involvement | 7 |
| Anthracycline, IDA/DNR* | 83/71 |
| Leukocyte count | |
| Median (range) | 17 × 109/L (0.5 × 109/L to 626 × 109/L) |
| 25 × 109/L or more | 66 |
| Immunophenotyping | |
| B-lineage ALL | 154 |
| Myeloid markers† | 10 (of 150) |
| Cytogenetics, N = 141 patients | |
| Hyperploidy | 12 |
| Monosomy 7 | 13 |
| NN-AN/AA | 102/39 |
| NN/AN-AA | 15/126 |
| Ph+ | 122 |
| Additional abnormalities | 91 |
| Ph duplication | 20 |
| RT-PCR, N = 142 patients | |
| m-bcr | 97 |
| M-bcr | 40 |
| m/M-bcr | 5 |
| Characteristic . | Result . |
|---|---|
| Baseline characteristics, N = 154 patients | |
| Age, y | |
| Median (range) | 42 (17-56) |
| 45 y or more | 66 |
| Sex ratio, M/F | 89/65 |
| CNS involvement | 7 |
| Anthracycline, IDA/DNR* | 83/71 |
| Leukocyte count | |
| Median (range) | 17 × 109/L (0.5 × 109/L to 626 × 109/L) |
| 25 × 109/L or more | 66 |
| Immunophenotyping | |
| B-lineage ALL | 154 |
| Myeloid markers† | 10 (of 150) |
| Cytogenetics, N = 141 patients | |
| Hyperploidy | 12 |
| Monosomy 7 | 13 |
| NN-AN/AA | 102/39 |
| NN/AN-AA | 15/126 |
| Ph+ | 122 |
| Additional abnormalities | 91 |
| Ph duplication | 20 |
| RT-PCR, N = 142 patients | |
| m-bcr | 97 |
| M-bcr | 40 |
| m/M-bcr | 5 |
CNS indicates central nervous system; IDA, idarubicin; DNR, daunorubicin; Ph, Philadelphia chromosome; m-bcr, minor breakpoint cluster region; and M-bcr, major breakpoint cluster region.
Anthracycline randomly allocated for the induction course.
Defined as the coexpression of CD34, CD33, and CD13 antigens (available in 150 patients).
A total of 110 (71%) of 154 patients were diagnosed with Ph+ and BCR-ABL+ ALL based on both standard cytogenetic and molecular analyses. Thirty-two patients (21%) were diagnosed with BCR-ABL+ ALL on molecular analysis only, because standard karyotype failed in 13 patients or showed no Philadelphia chromosome in 19 patients. Twelve patients (8%) were diagnosed with Ph+ ALL on standard karyotype only, because RT-PCR was not done in 5 patients, was negative in 1 patient, and failed in the remaining 6 patients.
Overall, 141 patients had an informative karyotype (Table 2). Thirty-nine patients were classified as AA (no normal metaphases), 87 patients as AN (persistence of normal metaphases), and 15 patients as NN (normal karyotype). The Philadelphia chromosome was present in 122 patients. It was the sole chromosomal abnormality in 31 patients and was associated with additional abnormalities in 91 patients. A duplication of the Philadelphia chromosome was observed in 20 patients. An associated monosomy 7 was observed in 13 patients. Hyperdiploidy was observed in 12 patients. Overall, 142 patients had an informative RT-PCR (Table 2). Ninety-seven patients (68%) had m-bcr ALL, 40 patients (28%) had M-bcr ALL, and 5 patients (4%) had m/M-bcr ALL. These 5 m/M-bcr cases were classified with the M-bcr cases for further comparisons.
Some baseline characteristics differed between younger and older patients. First, there was a negative correlation between age and leukocyte count. Median leukocyte count was 21 × 109/L (range, 0.6 × 109/L to 626 × 109/L) in the 88 patients aged less than 45 years as compared with 10 × 109/L (range, 0.5 × 109/L to 500 × 109/L) in the 66 patients aged 45 years or more (P = .045 by the Mann-Whitney test). Secondly, the proportion of patients with M-bcr ALL increased with age (25% of patients aged less than 45 years vs 41% of patients aged 45 years or more; P = .045 by the Fisher exact test). Finally, a duplication of the Philadelphia chromosome was more frequently observed in older patients (9% in patients aged less than 45 years vs 27% in patients aged 45 years or more; P = .01 by the Fisher exact test).
Overall survival
At 3 years, the estimated survival of the whole patient population was 19% (95% confidence interval, 13% to 25%). In univariate analysis, advanced age and high leukocyte count were the only baseline patient or disease characteristics identified as associated with a shorter survival. Prognostic significance was observed either when these factors were tested as continuous variables (P = .04 and P < .001 for age and leukocyte count, respectively, using the univariate Cox model) or when using an age cutoff at 45 years (relative risk in the older group, 1.47; 95% CI, 1.03 to 2.09;P = .03 by the log-rank test) and a leukocyte cutoff at 25 × 109/L (relative risk in the high-count group, 1.66; 95% CI, 1.16 to 2.35; P = .004 by the log-rank test). These 2 factors remained significantly associated with shorter survival after adjustment using the Cox model (P = .01 andP < .001 for age and leukocyte count, respectively, when both were considered as continuous variables; P = .01 andP = .002 for age and leukocyte count, respectively, when using these 45 years and 25 × 109/L cutoffs). Early response was evaluated on day 8 of the induction course in 81 patients (53%). In these patients, the lack of early response and advanced age remained the 2 bad-prognosis factors for survival in a 3-variable model, including day 8 response, age with the 45 years cutoff, and leukocyte count with the 25 × 109/L cutoff (relative risk in nonresponders at day 8, 2.59; 95% CI, 1.26 to 5.32;P = .01) (relative risk in older patients, 1.77; 95% CI, 1.05 to 3.0; P = .03).
Response to therapy
After induction, 81 (53%) of the 154 patients were alive in CR (Table 3). After HAM consolidation/salvage, 103 (67%) of the 154 patients were alive in CR. Among the 69 patients alive with resistant disease after induction, 29 reached a CR after HAM, representing a HAM-associated salvage rate of 42%. Meanwhile, 6 patients in CR after induction were in very early relapse after HAM consolidation, and 1 patient died during HAM consolidation. At 3 months (which was the time-planned for HSCT), 95 patients (62%) were still alive in CR. Among the 81 patients in CR after induction, 49 patients were tested by RT-PCR for minimal residual disease, and 13 (27%) were negative. Among the 103 patients in CR after HAM consolidation/salvage, 63 patients were tested by RT-PCR for minimal residual disease, and 24 (38%) were negative. No differences in baseline characteristics were found between CR patients tested and those not tested for minimal residual disease, either after 1 or 2 cycles of chemotherapy.
Response to therapy (N = 154 patients)
| . | No. of patients after induction (%) . | No. of patients after consolidation/salvage (%) . | No. of patients at 3 months (%) . |
|---|---|---|---|
| Complete remission | 81/154 (53) | 103/154 (67) | 95/154 (62) |
| Failure | |||
| Resistant disease | 69 (45) | 43 (28) | 50 (32) |
| Death | 4 (2) | 8 (5) | 9 (6) |
| . | No. of patients after induction (%) . | No. of patients after consolidation/salvage (%) . | No. of patients at 3 months (%) . |
|---|---|---|---|
| Complete remission | 81/154 (53) | 103/154 (67) | 95/154 (62) |
| Failure | |||
| Resistant disease | 69 (45) | 43 (28) | 50 (32) |
| Death | 4 (2) | 8 (5) | 9 (6) |
Because HSCT was planned to be performed at 3 months, prognostic factors were evaluated for hematologic response at 3 months (Table4). Ninety-five patients alive in complete remission were compared with 59 patients in failure. In a 3-variable analysis (leukocyte count, bcr subtype, NN feature) performed in 129 patients with all variables available, a leukocyte count below 25 × 109/L (P = .02) and the M-bcr subtype (P = .04) were independent favorable factors for being in CR at 3 months. Remission rate was 70% in the 88 patients with fewer than 25 × 109/L leukocytes as compared with 50% in the 66 patients with 25 × 109/L leukocytes or more. Remission rate was 73% in the 45 patients with M-bcr ALL as compared with 58% in the 97 patients with m-bcr ALL. In a 4-variable analysis (leukocyte count, bcr subtype, NN feature, and early response) performed in 69 patients also tested on day 8 for early response, the early response was the only factor that predicted CR persistence at 3 months (P = .05).
Prognostic factors for CR at three months (N = 154 patients)
| Variable . | No. of patients considered . | P (RR, 95% CI) Univariate analyses4-150 . | P (RR, 95% CI) Multivariate analyses4-151 . | |
|---|---|---|---|---|
| N = 129 pts . | N = 69 pts . | |||
| Age | ||||
| As a continuous variable | 154 | .76 | — | — |
| Less than 45 y | 154 | .50 | — | — |
| Sex | 154 | .74 | — | — |
| CNS involvement | 154 | .99 | — | — |
| Myeloid markers‡ | 150 | .18 | — | — |
| Anthracycline4-153 | 154 | .10 | — | — |
| Leukocyte count | ||||
| As a continuous variable | 154 | .01 | — | — |
| Less than 25 × 109/L | 154 | .01 | .02 | .09 |
| 1.41 (1.07-1.86) | 2.53 (1.15-5.57) | 2.59 (0.86-7.75) | ||
| Cytogenetics | ||||
| Hyperploidy | 141 | .13 | — | — |
| Monosomy 7 | 141 | .37 | — | — |
| Additional abnormalities4-155 | 122 | .67 | — | — |
| Ph duplication4-155 | 122 | .33 | — | — |
| NN vs AN-AA | 141 | .05 | .14 | .15 |
| 1.48 (1.15-1.89) | 3.29 (0.68-15.9) | 5.06 (0.56-45.8) | ||
| bcr subtype: M-bcr vs m-bcr | 142 | .09 | .04 | .47 |
| 1.27 (0.99-1.62) | 2.52 (1.06-6.01) | 1.55 (0.47-5.10) | ||
| Early response to therapy4-154 | 81 | .03 | — | .05 |
| 1.59 (1.16-2.18) | 5.21 (1.03-26.4) | |||
| Variable . | No. of patients considered . | P (RR, 95% CI) Univariate analyses4-150 . | P (RR, 95% CI) Multivariate analyses4-151 . | |
|---|---|---|---|---|
| N = 129 pts . | N = 69 pts . | |||
| Age | ||||
| As a continuous variable | 154 | .76 | — | — |
| Less than 45 y | 154 | .50 | — | — |
| Sex | 154 | .74 | — | — |
| CNS involvement | 154 | .99 | — | — |
| Myeloid markers‡ | 150 | .18 | — | — |
| Anthracycline4-153 | 154 | .10 | — | — |
| Leukocyte count | ||||
| As a continuous variable | 154 | .01 | — | — |
| Less than 25 × 109/L | 154 | .01 | .02 | .09 |
| 1.41 (1.07-1.86) | 2.53 (1.15-5.57) | 2.59 (0.86-7.75) | ||
| Cytogenetics | ||||
| Hyperploidy | 141 | .13 | — | — |
| Monosomy 7 | 141 | .37 | — | — |
| Additional abnormalities4-155 | 122 | .67 | — | — |
| Ph duplication4-155 | 122 | .33 | — | — |
| NN vs AN-AA | 141 | .05 | .14 | .15 |
| 1.48 (1.15-1.89) | 3.29 (0.68-15.9) | 5.06 (0.56-45.8) | ||
| bcr subtype: M-bcr vs m-bcr | 142 | .09 | .04 | .47 |
| 1.27 (0.99-1.62) | 2.52 (1.06-6.01) | 1.55 (0.47-5.10) | ||
| Early response to therapy4-154 | 81 | .03 | — | .05 |
| 1.59 (1.16-2.18) | 5.21 (1.03-26.4) | |||
RR indicates relative risk for being in complete remission at 3 months; CI, confidence interval; CNS, central nervous system; Ph, Philadelphia chromosome; M-bcr, major breakpoint cluster region; and m-bcr, minor breakpoint cluster region.
Using the Fisher exact test for binary variables and a univariate logistic model for continuous variables.
Using a multivariate logistic model.
Defined as the coexpression of CD34, CD33, and CD13 antigens.
Anthracycline randomly allocated for the induction course.
Comparisons were performed in the 122 patients with Philadelphia chromosome.
Evaluated on the day 8 bone marrow aspiration.
Prognostic factors were also analyzed for hematologic response to the first course of induction and to salvage HAM chemotherapy, respectively. In multivariate analysis (not shown), a low leukocyte count was the only baseline characteristic identified for being in CR after induction. Again, the early response was the only factor that predicted CR after induction in patients tested for response at day 8, even after adjustment on leukocyte count (P = .03). In patients alive with resistant disease after induction, the only factor predictive of a CR at 3 months was the M-bcr subtype (salvage rate, 55% in patients with M-bcr ALL vs 30% in those with m-bcr ALL;P = .05 by the Fisher exact test).
In the 63 CR patients tested by RT-PCR for minimal residual disease after HAM consolidation/salvage, the only prognostic factor identified for being BCR-ABL negative was hyperploidy (P = .02 by the Fisher exact test).
Outcome of patients eligible for HSCT
Outcome was analyzed on an intention-to-treat basis for the 103 patients alive in complete remission after the HAM course of chemotherapy and thus eligible for HSCT. As expected, the survival of the 43 patients alive with resistant disease at that time was significantly shorter than the survival of these 103 responders (relative risk, 2.14; 95% confidence interval, 1.45 to 3.15;P < .001 by the log-rank test). These 103 patients were distributed within the 3 HSCT groups as follows: 46 patients in the allo-ID group, 14 patients in the allo-MUD group, and 43 patients in the autologous group. In addition, an unsuccessful search for a MUD was performed in 14 of the 43 patients from the autologous group. Because survival in the allo-MUD group did not differ from survival in the allo-ID group (relative risk in the allo-MUD, 0.53; 95% CI, 0.25 to 1.16; P = .11 by the log-rank test), comparisons were performed between a donor group (n = 60 patients) and a no-donor group (n = 43 patients). No differences in baseline characteristics and marrow response at day 8 were found between these 2 patient groups.
Transplantation was actually performed in 44 of the 46 patients of the allo-ID group (including 1 patient in relapse at HSCT time), in 12 of the 14 patients of the allo-MUD (including 4 patients in relapse at HSCT time), and in 24 of the 43 patients of the autologous group (including 1 patient in relapse at HSCT time). In addition, 1 patient from the autologous group did not receive the planned autologous HSCT but received an allo-MUD transplantation in second CR because of late identification of a MUD. Overall, 51 patients from the donor group received an allogeneic HSCT in first CR (43 allo-ID and 8 allo-MUD), and 23 patients from the no-donor group received an autologous HSCT in first CR. Individual posttransplantation outcome for the 74 patients actually undergoing transplantation in first CR is indicated in Table5 according to the HSCT group and to the pretransplantation (ie, post-HAM) BCR-ABLstatus. In these patients, the median time from initial randomization to HSCT was 113 days (range, 90 to 192 days) in the allo-ID group, 138 days (range, 92 to 227 days) in the allo-MUD group, and 128 days (range, 79 to 350 days) in the autologous group. Nine eligible patients from the donor group and 20 eligible patients from the no-donor group did not receive HSCT in first CR. The reason was relapse within the 3 months following CR achievement in 5 patients from the donor group (8%) as compared with 10 patients from the no-donor group (23%) (P = .05 by the Fisher exact test). The 4 remaining patients from the donor group and the 10 remaining patients from the no-donor group did not undergo transplantation in first CR for other various reasons and received various further chemotherapy regimens.
Individual post-HSCT outcome of patients actually undergoing transplantation in first CR (N = 74 patients)
| Patient no. . | Pretransplantation BCR-ABLstatus5-150 . | Relapse . | Time to hematologic relapse, mo . | Mortality . | Post-HSCT survival, mo . |
|---|---|---|---|---|---|
| Autologous group | |||||
| 02011 | − | − | − | 76+ | |
| 02009 | − | − | − | 78+ | |
| 21006 | − | + | 3 | + | 9 |
| 02005 | − | + | 6 | + | 11 |
| 16002 | − | + | 33 | + | 35 |
| 11013 | − | + | 23 | + | 57 |
| 02025 | + | − | + | 1 | |
| 02014 | + | − | + | 4 | |
| 14012 | + | + | 2 | + | 3 |
| 38001 | + | + | 6 | + | 8 |
| 07007 | + | + | 7 | + | 9 |
| 15004 | + | + | 2 | + | 9 |
| 01024 | + | + | 6 | + | 11 |
| 06005 | + | + | 5 | + | 12 |
| 01036 | + | + | 8 | + | 23 |
| 10011 | + | + | 4 | + | 27 |
| 02023 | + | + | 27 | + | 29 |
| 03022 | nd | − | + | 1 | |
| 14015 | nd | − | + | 1 | |
| 01010 | nd | + | 3 | + | 10 |
| 13011 | nd | + | 8 | + | 10 |
| 07048 | nd | + | 4 | + | 14 |
| 23011 | nd | + | 7 | + | 16 |
| Allo-ID group | |||||
| 26020 | − | − | − | 51+ | |
| 37009 | − | − | − | 61+ | |
| 15002 | − | − | − | 74+ | |
| 37033 | − | − | + | 1 | |
| 37023 | − | − | + | 26 | |
| 35001 | − | − | + | 77 | |
| 07049 | − | + | 14 | + | 15 |
| 23002 | − | + | 17 | + | 17 |
| 14008 | − | + | 17 | + | 18 |
| 09006 | − | + | 43 | + | 44 |
| 01057 | + | − | − | 22+ | |
| 14020 | + | − | − | 26+ | |
| 02047 | + | − | − | 32+ | |
| 17013 | + | − | − | 43+ | |
| 07017 | + | − | − | 72+ | |
| 27018 | + | + | 17 | + | 20 |
| 28008 | + | − | − | 44+ | |
| 37007 | + | − | − | 64+ | |
| 05029 | + | − | + | 0.5 | |
| 04054 | + | − | + | 3 | |
| 26006 | + | − | + | 3 | |
| 14021 | + | + | 4 | + | 8 |
| 14016 | + | + | 4 | + | 8 |
| 16003 | + | + | 7 | + | 10 |
| 28001 | + | + | 14 | + | 14 |
| 01014 | + | + | 15 | + | 16 |
| 09013 | + | + | 16 | + | 27 |
| 17018 | nd | − | − | 23+ | |
| 03050 | nd | − | − | 29+ | |
| 21011 | nd | − | − | 30+ | |
| 31004 | nd | − | − | 76+ | |
| 28014 | nd | − | + | 0.5 | |
| 10007 | nd | − | + | 0.5 | |
| 28034 | nd | − | + | 4 | |
| 23009 | nd | − | + | 6 | |
| 07033 | nd | − | + | 7 | |
| 13004 | nd | − | + | 22 | |
| 03045 | nd | + | 2 | + | 4 |
| 04037 | nd | + | 4 | + | 5 |
| 23004 | nd | + | 5 | + | 7 |
| 03043 | nd | + | 2 | + | 8 |
| 04021 | nd | + | 10 | + | 14 |
| 27004 | nd | + | 17 | + | 27 |
| Allo-MUD group | |||||
| 15011 | − | − | − | 16+ | |
| 13012 | − | − | − | 51+ | |
| 01018 | − | − | − | 66+ | |
| 03035 | − | − | + | 0.5 | |
| 28022 | + | − | − | 36+ | |
| 05006 | nd | − | − | 73+ | |
| 35036 | nd | + | 3 | + | 4 |
| 35009 | nd | + | 14 | + | 39 |
| Patient no. . | Pretransplantation BCR-ABLstatus5-150 . | Relapse . | Time to hematologic relapse, mo . | Mortality . | Post-HSCT survival, mo . |
|---|---|---|---|---|---|
| Autologous group | |||||
| 02011 | − | − | − | 76+ | |
| 02009 | − | − | − | 78+ | |
| 21006 | − | + | 3 | + | 9 |
| 02005 | − | + | 6 | + | 11 |
| 16002 | − | + | 33 | + | 35 |
| 11013 | − | + | 23 | + | 57 |
| 02025 | + | − | + | 1 | |
| 02014 | + | − | + | 4 | |
| 14012 | + | + | 2 | + | 3 |
| 38001 | + | + | 6 | + | 8 |
| 07007 | + | + | 7 | + | 9 |
| 15004 | + | + | 2 | + | 9 |
| 01024 | + | + | 6 | + | 11 |
| 06005 | + | + | 5 | + | 12 |
| 01036 | + | + | 8 | + | 23 |
| 10011 | + | + | 4 | + | 27 |
| 02023 | + | + | 27 | + | 29 |
| 03022 | nd | − | + | 1 | |
| 14015 | nd | − | + | 1 | |
| 01010 | nd | + | 3 | + | 10 |
| 13011 | nd | + | 8 | + | 10 |
| 07048 | nd | + | 4 | + | 14 |
| 23011 | nd | + | 7 | + | 16 |
| Allo-ID group | |||||
| 26020 | − | − | − | 51+ | |
| 37009 | − | − | − | 61+ | |
| 15002 | − | − | − | 74+ | |
| 37033 | − | − | + | 1 | |
| 37023 | − | − | + | 26 | |
| 35001 | − | − | + | 77 | |
| 07049 | − | + | 14 | + | 15 |
| 23002 | − | + | 17 | + | 17 |
| 14008 | − | + | 17 | + | 18 |
| 09006 | − | + | 43 | + | 44 |
| 01057 | + | − | − | 22+ | |
| 14020 | + | − | − | 26+ | |
| 02047 | + | − | − | 32+ | |
| 17013 | + | − | − | 43+ | |
| 07017 | + | − | − | 72+ | |
| 27018 | + | + | 17 | + | 20 |
| 28008 | + | − | − | 44+ | |
| 37007 | + | − | − | 64+ | |
| 05029 | + | − | + | 0.5 | |
| 04054 | + | − | + | 3 | |
| 26006 | + | − | + | 3 | |
| 14021 | + | + | 4 | + | 8 |
| 14016 | + | + | 4 | + | 8 |
| 16003 | + | + | 7 | + | 10 |
| 28001 | + | + | 14 | + | 14 |
| 01014 | + | + | 15 | + | 16 |
| 09013 | + | + | 16 | + | 27 |
| 17018 | nd | − | − | 23+ | |
| 03050 | nd | − | − | 29+ | |
| 21011 | nd | − | − | 30+ | |
| 31004 | nd | − | − | 76+ | |
| 28014 | nd | − | + | 0.5 | |
| 10007 | nd | − | + | 0.5 | |
| 28034 | nd | − | + | 4 | |
| 23009 | nd | − | + | 6 | |
| 07033 | nd | − | + | 7 | |
| 13004 | nd | − | + | 22 | |
| 03045 | nd | + | 2 | + | 4 |
| 04037 | nd | + | 4 | + | 5 |
| 23004 | nd | + | 5 | + | 7 |
| 03043 | nd | + | 2 | + | 8 |
| 04021 | nd | + | 10 | + | 14 |
| 27004 | nd | + | 17 | + | 27 |
| Allo-MUD group | |||||
| 15011 | − | − | − | 16+ | |
| 13012 | − | − | − | 51+ | |
| 01018 | − | − | − | 66+ | |
| 03035 | − | − | + | 0.5 | |
| 28022 | + | − | − | 36+ | |
| 05006 | nd | − | − | 73+ | |
| 35036 | nd | + | 3 | + | 4 |
| 35009 | nd | + | 14 | + | 39 |
nd indicates not done.
Post-HAM BCR-ABL status.
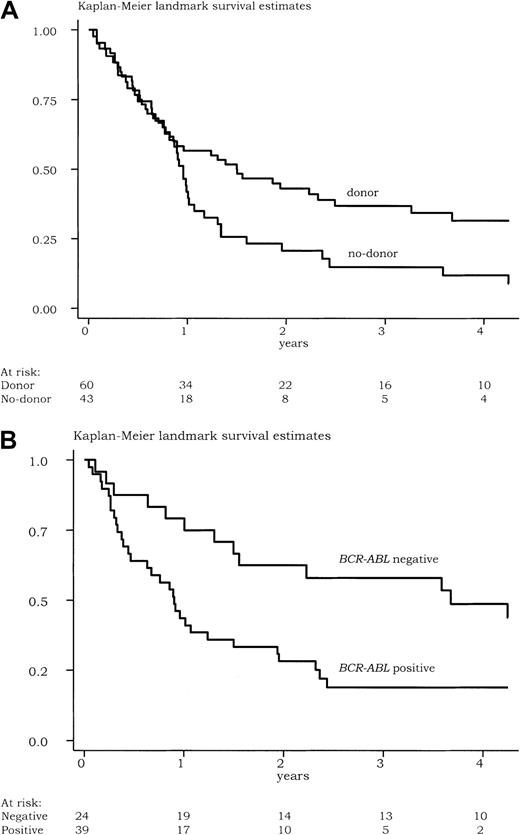
Of the 103 patients eligible for HSCT, 77 died (39 in the donor group and 38 in the no-donor group). Estimated 3-year survival was 28% (95% CI, 19% to 37%). Prognostic factors for survival are summarized in Table 6. Survival of patients without a donor was significantly shorter than survival of those with a donor (Figure 1A). At 3 years, the estimated survival was 37% in the donor group (95% CI, 24% to 49%) and 12% in the no-donor group (95% CI, 4% to 24%) (relative risk in the no-donor group, 1.71; 95% CI, 1.09 to 2.68; P = .02 by the log-rank test). Sixty-three patients (61%) were tested forBCR-ABL status after the HAM course. Survival was significantly longer in the 24 BCR-ABL−patients when compared with the 39 BCR-ABL+patients (Figure 1B). At 3 years, the estimated survival was 54% in the BCR-ABL− group (95% CI, 32% to 71%) and 19% in the BCR-ABL+ group (95% CI, 8% to 33%) (relative risk in the BCR-ABL+ group, 2.46; 95% CI, 1.26 to 4.78; P = .006 by the log-rank test). Interestingly, in 45 patients tested, the BCR-ABLstatus after only 1 course of induction chemotherapy had no significant influence on outcome. After adjustment on age (with the 45 years cutoff), leukocyte count (as a continuous variable), and number of courses of chemotherapy required to reach the CR, the existence of an allogeneic donor and the BCR-ABL− status after HAM remained the 2 independent favorable prognostic factors for survival (P = .02 and P = .01, respectively). Of note, there was no difference in BCR-ABL status after HAM between the 23 patients from the no-donor group who actually received an autologous HSCT in first CR and the 20 patients who did not receive it, even if there was a trend for more patients tested in the former subgroup (17 of 23 patients vs 9 of 20 patients; P = .07 by the Fisher exact test).
Prognostic factors for survival in patients eligible for stem cell transplantation (N = 103 patients)
| Variable . | No. of patients considered . | P (RR, 95% CI) Univariate analyses6-150 . | P (RR, 95% CI) Multivariate analysis6-151 N = 63 patients . |
|---|---|---|---|
| Age | |||
| As a continuous variable | 103 | .06 | — |
| Less than 45 y | 103 | .05 | .13 |
| 1.57 (1.01-2.47) | |||
| Sex | 103 | .29 | — |
| CNS involvement | 103 | .63 | — |
| Myeloid markers6-152 | 100 | .50 | — |
| Anthracycline | 103 | .65 | — |
| Leukocyte count | |||
| As a continuous variable | 103 | .06 | .23 |
| Less than 25 × 109/L | 103 | .35 | — |
| Cytogenetics | |||
| Hyperploidy | 94 | .27 | — |
| Monosomy 7 | 94 | .86 | — |
| Additional abnormalities6-153 | 80 | .28 | — |
| Ph duplication6-153 | 80 | .15 | — |
| NN-AN vs AA | 94 | .36 | — |
| NN vs AN-AA | 94 | .62 | — |
| bcr subtype | 97 | .64 | — |
| No allogeneic donor | 103 | .02 | .02 |
| 1.71 (1.09-2.68) | 2.12 (1.11-40.5) | ||
| Poor response to therapy | |||
| No CR in 1 course | 103 | .08 | .11 |
| BCR-ABL status after 1 course | 45 | .50 | — |
| BCR-ABL status after 2 courses | 63 | .006 | .01 |
| 2.46 (1.26-4.78) | 2.39 (1.21-4.74) |
| Variable . | No. of patients considered . | P (RR, 95% CI) Univariate analyses6-150 . | P (RR, 95% CI) Multivariate analysis6-151 N = 63 patients . |
|---|---|---|---|
| Age | |||
| As a continuous variable | 103 | .06 | — |
| Less than 45 y | 103 | .05 | .13 |
| 1.57 (1.01-2.47) | |||
| Sex | 103 | .29 | — |
| CNS involvement | 103 | .63 | — |
| Myeloid markers6-152 | 100 | .50 | — |
| Anthracycline | 103 | .65 | — |
| Leukocyte count | |||
| As a continuous variable | 103 | .06 | .23 |
| Less than 25 × 109/L | 103 | .35 | — |
| Cytogenetics | |||
| Hyperploidy | 94 | .27 | — |
| Monosomy 7 | 94 | .86 | — |
| Additional abnormalities6-153 | 80 | .28 | — |
| Ph duplication6-153 | 80 | .15 | — |
| NN-AN vs AA | 94 | .36 | — |
| NN vs AN-AA | 94 | .62 | — |
| bcr subtype | 97 | .64 | — |
| No allogeneic donor | 103 | .02 | .02 |
| 1.71 (1.09-2.68) | 2.12 (1.11-40.5) | ||
| Poor response to therapy | |||
| No CR in 1 course | 103 | .08 | .11 |
| BCR-ABL status after 1 course | 45 | .50 | — |
| BCR-ABL status after 2 courses | 63 | .006 | .01 |
| 2.46 (1.26-4.78) | 2.39 (1.21-4.74) |
All survival analyses were performed using the landmark method with a landmark period of 90 days.
RR indicates relative risk of failure; CI, confidence interval; CNS, central nervous system; and Ph, Philadelphia chromosome.
Using the log-rank test for binary variables and univariate Cox models for continuous variables.
Using the Cox model.
Defined as the coexpression of CD34, CD33, and CD13 antigens.
Comparisons were performed in the 80 patients with Philadelphia chromosome.
Survival of patients eligible for stem cell transplantation.
Kaplan-Meier landmark survival estimates (with a 90-day landmark period) for patients eligible for HSCT (A) according to the existence of an allogeneic donor (N = 103 patients) (relative risk in the no-donor group, 1.71; 95% confidence interval, 1.09 to 2.68;P = .02 by the log-rank test) and (B) according to theBCR-ABL status after 2 courses of chemotherapy (N = 63 patients) (relative risk in the BCR-ABL+ group, 2.46; 95% confidence interval, 1.26 to 4.78; P = .006 by the log-rank test).
Survival of patients eligible for stem cell transplantation.
Kaplan-Meier landmark survival estimates (with a 90-day landmark period) for patients eligible for HSCT (A) according to the existence of an allogeneic donor (N = 103 patients) (relative risk in the no-donor group, 1.71; 95% confidence interval, 1.09 to 2.68;P = .02 by the log-rank test) and (B) according to theBCR-ABL status after 2 courses of chemotherapy (N = 63 patients) (relative risk in the BCR-ABL+ group, 2.46; 95% confidence interval, 1.26 to 4.78; P = .006 by the log-rank test).
Relapse was the main cause of treatment failure. Of the 103 patients, 64 relapsed (28 in the donor group and 36 in the no-donor group). Estimated 3-year relapse rate was 65% (95% CI, 55% to 76%). The existence of an allogeneic donor and theBCR-ABL− status after HAM were also favorable prognostic factors for the duration of complete remission. At 3 years, the estimated incidence of relapse was 50% in the donor group (95% CI, 37% to 65%) as compared with 90% in the no-donor group (95% CI, 76% to 97%) (relative risk in the no-donor group, 3.17; 95% CI, 1.86 to 5.41; P < .001 by the log-rank test) and 41% in theBCR-ABL− group (95% CI, 23% to 66%) as compared with 75% in the BCR-ABL+ group (95% CI, 59% to 89%) (relative risk in the BCR-ABL+group, 2.55; 95% CI, 1.20 to 5.40; P = .01 by the log-rank test). In a multivariate analysis, the prognostic value of these 2 factors remained significant (P < .001 andP = .01, respectively) when also taking into consideration the 2 other following independent factors: a high leukocyte count (P = .01) and the need for 2 courses of chemotherapy to reach the CR (P = .05). Here again, the BCR-ABLstatus after only 1 course of induction chemotherapy had no significant influence on CR duration (P = .64 by the log-rank test).
Of the 103 patients, 17 died in complete remission (12 in the donor group and 5 in the no-donor group). At 2 years, the estimated incidence of deaths in complete remission was 24% in the donor group (95% CI, 13% to 39%) and 24% in the no-donor group (95% CI, 11% to 54%) (relative risk in the donor group, 1.18; 95% CI, 0.41 to 3.38; P = .79 by the log-rank test). Subgroup analysis was hindered by the low number of patients tested for BCR-ABLstatus in each HSCT group. The prognostic value of BCR-ABLstatus appeared, however, to differ between the donor and the no-donor group. In the donor group, 22 patients wereBCR-ABL+ and 15BCR-ABL−. Estimated 3-year survival was 35% (95% CI, 16% to 55%) in BCR-ABL+patients versus 59% (95% CI, 31% to 79%) inBCR-ABL− patients (P = .18 by the log-rank test). In the no-donor group, 17 patients wereBCR-ABL+ and 9 onlyBCR-ABL−. Estimated 3-year survival was 0% inBCR-ABL+ patients versus 44% (95% CI, 14% to 72%) in BCR-ABL− patients (P = .005 by the log-rank test).
Finally, because an unrelated donor was searched in only 28 of the 57 patients without an identified matched related donor, a selection bias may have been introduced in assigning patients to the allo-MUD or autologous groups. Age appeared to be the main selection criteria for MUD search, because the median age of patients with a MUD search was 36 years (range, 16 to 54 years) compared with 49 years (range, 33 to 55 years) in those without MUD search (P < .0001 by the Mann-Whitney test). A similar intent-to-treat analysis was thus performed comparing the 46 patients from the allo-ID group with the 57 patients from the autologous or allo-MUD groups. Results were roughly the same because, after adjustment on leukocyte count, age, and number of courses of chemotherapy required to reach the CR, the existence of a matched related donor and the BCR-ABL− status after HAM remained 2 independent prognostic factors for longer survival (P = .03 and P = .01, respectively) and longer duration of complete remission (P < .001 andP = .006, respectively).
Discussion
As demonstrated in this first large-scale prospective study in adult patients with Ph+ and/orBCR-ABL+ ALL, the combination of both cytogenetic and molecular analyses at baseline allows one to early diagnose Ph+ and/or BCR-ABL+ ALL in about 20% of all patients with ALL between the ages of 15 and 55 years. Given the poor prognosis associated with this variant of ALL in adults, such an early diagnosis is a prerequisite to stratify the treatment, based on intensive chemotherapy and HSCT in first CR.
In fact, most specialists would recommend allogeneic HSCT for an adult with Ph+ and/or BCR-ABL+ ALL in first CR.10-12 This recommendation was essentially based on clinical experience, because there was no large prospective study demonstrating a clear advantage associated with this strategy. In 1992, a retrospective report from the International Bone Marrow Transplantation Registry showed an improved rate of leukemia-free survival in adult patients with Ph+ ALL who received an HSCT from a matched related donor.11 In our previous prospective LALA-87 study,26-28 allogeneic matched related HSCT resulted in improved leukemia-free and overall survival in patients with high-risk ALL, including those with Ph+ ALL. Because many patients eligible for allogeneic HSCT lack a matched related or unrelated donor, autologous HSCT has been suggested as an alternative in these patients.10,28 29
The poor prognosis associated with Ph+ and/orBCR-ABL+ ALL is due to a slow and partial reduction of the leukemic clone by initial chemotherapy as compared with less aggressive variants of ALL.14 Many patients never achieve a CR, and relapse is the most common cause of treatment failure in those achieving a CR. The molecular monitoring of minimalBCR-ABL residual disease during the early phases of therapy may thus provide an efficient prognostic tool and may help in the evaluation of novel therapeutic options.
We found in the present study that the existence of an allogeneic donor and the obtention of a molecular remission after 2 courses of chemotherapy represent 2 independent prognostic factors for longer remission duration and survival. Earlier assessment of minimal residual disease after only 1 course of induction therapy is an inaccurate indicator of clinical outcome. These observations are consistent with 2 other adult ALL reports using different molecular targets for minimal residual disease detection.30 31
In conclusion, several clinical recommendations may be drawn from the results of our study. First, the study prospectively confirms that allogeneic HSCT from either a matched related or a molecularly matched unrelated donor in first CR is the first-choice treatment option in adults with this disease. Secondly, the molecular BCR-ABLstatus has to be taken into account in patients eligible for HSCT, especially in those without an allogeneic donor, because it is suggested that cumulating both poor prognostic factors results in a very poor outcome. It is thus likely that autologous HSCT cannot be recommended in patients with persistent minimal BCR-ABLdisease, even if larger studies or overviews are needed to strongly validate this nonrecommendation. Larger studies or registry analyses are also needed to further evaluate the prognostic value of the pretransplantation BCR-ABL molecular status in patients with an allogeneic donor. In addition, the introduction of the innovative real-time quantitative PCR technology should improve the molecular monitoring of these patients in the near future.32 Third, the administration of additional cytarabine-based salvage chemotherapy like HAM is reasonable after induction failure because it is associated with a quite good response rate, especially in patients with M-bcr ALL. More generally, all efforts should be done to increase the rate of patients rapidly achieving a BCR-ABL−remission. The administration of new agents, such as STI571, has thus to be investigated within this early period of treatment. Because relapse is by far the most common cause of treatment failure, even in patients eligible for allogeneic HSCT, the dosage intensification of chemotherapy prior to transplantation remains another option. Early bone marrow evaluation, performed as early as after a week of chemotherapy, may be used to select slowly responding patients eligible for such intensified approaches.
We are indebted to Dr Marion Malphettes, Dr AndréBaruchel, Dr Laurent Degos, and Dr Gérard Socié for critical comments and assistance.
The members of the LALA Group are: P. Brice, J. M. Cayuela, A. Do, M. T. Daniel, L. Degos, H. Dombret, H. Espérou, E. Gluckman, G. Lebbé, O. Maarek, J. M. Micléa, P. Rousselot, F. Sigaux, G. Socié (Hôpital Saint Louis, Paris, France); J. N. Bastie, S. Castaigne, I. Garcia, C. Terré(Hôpital André Mignot, Versailles, France); C. Charrin, D. Fière, S. Hayette, V. Lheritier, J. P. Magaux, M. Michallet, A. Thiébaut, X. Thomas, D. Treille-Ritouet, E. Wattel (Hôpital Ed. Herriot, Lyon, France); C. Bilhou-Nabera, J. M. Boiron, H. Eghbali, P. Lacombe, G. Marit, V. Perreau, A. Pigneux, V. Praloran, J. Reiffers (Hôpital du Haut Lévêque, Bordeaux, France); M. Attal, N. Dastugue, C. Escriva, M. Georges, E. Kuhlein, G. Laurent, J. Pris, F. Rigal-Huguet, D. Schlaifer (Hôpital Purpan, Toulouse, France); X. Vallantin (Centre Hospitalier, Perpignan, France); J. Beaune, D. Bories, C. Cordonnier, H. Jouault, M. Kuentz (Hôpital Henri Mondor, Créteil, France); N. Ali Ammar, F. Davi, N. Dhedin, V. Leblond, H. Merle Beral, L. Sutton, J. P. Vernant (Hôpital PitiéSalpétrière, Paris, France); F. Léonard (Hôpital Bicètre, Le Kremlin Bicètre, France); D. Blaise, R. Bouabdallah, C. Faucher, J. Gabert, J. A. Gastaut, M. Lafage, D. Maraninchi, M. J. Mozziconacci, D. Redortier, D. Sainty, A.M. Stoppa, N. Vey (Institut Paoli Calmettes, Marseille, France); F. Dreyfus, A. Merlat, F. Picard, F. Viguié(Hôpital Cochin, Paris, France); A. Falkenrodt, M. P. Gaub, M. Lessard, B. Lioure, F. Maloisel, F. Uettwiller (Hôpital de Hautepierre, Strasbourg, France); E. Racadot (EFS, Besançon, France); N. Gratecos, P. Philip, S. Reynaud (Hôpital de l'Archet, Nice, France); V. Asnafi, A. Buzyn, E. Delabesse, O. Hermine, E. MacIntyre, F. Lefrère, I. Radford, F. Valensi, B. Varet (Hôpital Necker, Paris, France); A. Brizard, F. Guilhot, E. Randriamalala, A. Sadoun (Hôpital Jean Bernard, Poitiers, France); M. Favre, F. Garban, R. Gressin, M. C. Jacob, D. Leroux, V. Roland, J. J. Sotto, P. Swiercz (Hôpital Michallon, Grenoble, France); C. Bayle, A. Bernheim, J. H. Bourhis, G. Danglot, B. Debuire, N. Frenoy, K. Rideller, J. M. Vantelon (Institut Gustave Roussy, Villejuif, France); C. Boucheix, A. Talia (INSERM U268, Villejuif, France); D. Caillot, R. O. Casasnovas, M. Grandjean, H. Guy, M. Maynadie, F. Mugneret (Hôpital du Bocage, Dijon, France); N. Bellal, F. Isnard, A. Najman, C. Perot, J. Van den Akker, V. Vergé (Hôpital Saint Antoine, Paris, France); M. Leporrier, O. Reman, V. Salaun, X. Troussard (Centre Hospitalier, Caen, France); B. Drenou, J. Y. Leprisé (Hôpital Pontchaillou, Rennes, France); F. Bauduer, L. Ducout, M. Renoux (Centre Hospitalier de la Côte Basque, Bayonne, France); D. Espinouse, E. Callet-Bauchu, B. Coiffier, P. Felman, J. Reglioni, G. Salles (Centre Hospitalier, Lyon Sud, France); P. Bourin, T. De Revel, G. Nedellec, T. Samson, B. Souleau (HIA Percy, Clamart, France); F. Desangles (HIA Val de Grâce, Paris, France); M. Blanc, B. Dingeon, M. Hacini, S. Fert (Centre Hospitalier, Chambéry, France); D. Bordessoule, C. Brigaudeau, I. Lorget, F. Trimoreau, P. Turlure (Hôpital Dupuytren, Limoges, France); O. Boulat, G. Lepeu, S. Rossanino, A. Soria (Centre Hospitalier, Avignon, France); B. Audhuy, I. Mazurier (Hôpital Louis Pasteur, Colmar, France); E. Jandidier (Centre Hospitalier, Mulhouse, France); B. Lenormand, C. Bastard, A. Stamatoullas-Bastard, H. Tilly (Centre H. Becquerel, Rouen, France); J. Chassagne, B. Choufi, M. Giollant, A. Lénat, O. Tournilhac, P. Travade (Centre Hospitalier, Clermont-Ferrand, France); B. Bève, P. Fenaux, J. P. Jouet, J. L. Lai, C. Preudhomme (Centre Hospitalier, Lille, France); M. Amar, C. Gardin, J. J. Kiladjian (Hôpital Beaujon, Clichy, France); B. Corront, C. Martin (Centre Hospitalier, Annecy, France); M. de Bruyère, V. Deneys, A. Ferrant, P. Martiat, J. L. Michaux, L. Michaux, N. Streatmans, J. L. Vaerman, M. P. Vandermeer, C. Verellen (Cliniques St Luc, Bruxelles, Belgium); M. André, G. Bulliard (Centre Hospitalier Notre Dame et Reine Fabiola, Charleroi, Belgium); A. Bosly, B. Chatelain, G. Mineur, A. Sonet (Cliniques de Mont Godinne, Yvoir, Belgium); C. Sibille (ASBL, Loverval, Belgium); C. Fourneaux, V. Hennaux, P. Mineur (Hôpital St Joseph, Gilly, Belgium); A. Delannoy, C. Ravoet (Hôpital de Jolimont, Haine St Paul, Belgium); P. Fally, J. L. d'Hautecourt (Hôpital Saint Joseph, Mons, Belgium); M. Boland, B. De Prijck (Hôpital de la Citadelle, Liège, Belgium); A. Hagemeijer (Laboratoire de Cytogénétique, Leuven, Belgium); L. Bories, M. Dupont, C. Exbrayat, N. Fegueux, J. Taı̈eb (Centre Hospitalier Lapeyronie, Montpellier, France); C. Maurice, S. Caillères (Centre Hospitalier, Aix en Provence, France); W. Godefroy (Hôpital Jean Monod, Le Havre, France); C. Soussain, T. Ben Othman (Centre Hospitalier, Meaux, France); J. Y. Perrot (Hôpital Hôtel Dieu, Paris, France); H. Mossafa (Laboratoire CERBA, Val d'oise, France); M. Barnoux, B. Boval, A. C. Braux (Hôpital Lariboisière, Paris, France); H. Fezoui, C. Fourcade, M. Pulik (Hôpital Victor Dupouy, Argenteuil, France); B. Dupriez, P. Morel (Centre Hospitalier Dr Schaffner, Lens, France); F. Le Baron (Centre Hospitalier, Valenciennes, France); N. Cambier, C. Rose (Centre Hospitalier Saint Vincent, Lille, France); I. Plantier (Centre Hospitalier, Roubaix, France); and M. Taillefer (ETS, Lille, France).
Supported in part by Le Programme Hospitalier de Recherche Clinique (PHRC no. 94-95-97.02), Ministère de l'Emploi et de la Solidarité, France; and by L'Association Contre le Cancer (National Grants ARC no. 6237, 9623, and 5484).
Submitted March 6, 2002; accepted April 22, 2002. Prepublished online as Blood First Edition Paper, May 13, 2002; DOI 10.1182/blood-2002-03-0704.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Hervé Dombret, Hôpital Saint-Louis, Département d'Hématologie, 1 avenue Claude Vellefaux, 75010 Paris, France; email: herve.dombret@sls.ap-hop-paris.fr.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal