Abstract
The chronic myelogenous leukemia (CML)–like myeloproliferative disorder observed in the BCR/ABL murine bone marrow transduction and transplantation model shares several features with the human disease, including a high response rate to the tyrosine kinase inhibitor imatinib mesylate (STI571). To study the impact of chronic imatinib mesylate treatment on the CML-like illness, mice were maintained on therapeutic doses of this drug and serially monitored. Unexpectedly, despite excellent systemic control of the CML-like illness, many of the mice developed progressive neurologic deficits after 2 to 4 months of imatinib mesylate therapy because of central nervous system (CNS) leukemia. Analysis of imatinib mesylate cerebral spinal fluid concentrations revealed levels 155- fold lower than in plasma. Thus, in the mouse, the limited ability of imatinib mesylate to cross the blood-brain barrier allowed the CNS to become a sanctuary for Bcr/Abl-induced leukemia. This model will be a useful tool for the future study of novel anti-CML drugs and in better defining the mechanisms for limited imatinib mesylate penetration into the CNS.
Introduction
The murine BCR/ABL retroviral transduction and transplantation model of chronic myelogenous leukemia (CML) and Philadelphia chromosome–positive (Ph+) hematologic malignancies has provided important insights into the molecular pathophysiology of Bcr/Abl-induced leukemia. Although this mouse model is best known for the CML-like myeloproliferative disorder, Bcr/Abl-induced leukemias of the lymphoid and macrophage lineages have also been reported.1-5 With recent improvements in the retroviral transduction of CML target cells,5 however, 100% of mice die from the fulminant CML-like illness before they have the opportunity to manifest the acute lymphoblastic leukemia (ALL) or macrophage tumor diseases. Previously, we demonstrated that the tyrosine kinase inhibitor imatinib mesylate (STI571, Gleevec; Novartis, Basel, Switzerland) prolongs the survival of mice with CML-like myeloproliferative disorder, resulting in a disease closely resembling CML in chronic phase.6
Because of the marked efficacy of imatinib mesylate in humans with Ph+ hematologic malignancies,7-12 it is likely that a significant cohort of CML patients will be managed chronically on imatinib mesylate therapy.13 Imatinib mesylate–resistant Ph+ leukemia has already been reported by several groups,14-18 but thus far it appears to be a relatively rare event. Whether imatinib mesylate will permanently control or even cure CML is unknown. To address these questions, mice reconstituted with BCR/ABL-transduced bone marrow (BM) cells were maintained chronically on imatinib mesylate and monitored by serial physical exam and peripheral blood counts. Unexpectedly, most mice developed focal neurologic signs indicative of central nervous system (CNS) leukemia.
Study design
Murine BM retroviral transduction and transplantation
Lethally irradiated Balb/c mice (Charles River laboratories, Wilmington, MA) were reconstituted with syngeneic 5-fluorouracil–primed BM cells transduced with a replication-defective P210BCR/ABL retrovirus as previously described.6 Mice received 50 mg/kg imatinib mesylate every morning and 100 mg/kg every evening orally beginning on day 10 after BM reconstitution.6 Complete brain pathologic and necropsy analysis was performed on 8 mice.
Immunohistochemistry (IHC) and polymerase chain reaction (PCR)
Pathologic sections from brain, spinal cord, and liver were examined by routine hematoxylin and eosin (H&E) staining, or by IHC using anti-CD45R/B220 or anti-Mac3 antibodies (BD PharMingen, San Diego, CA) with appropriate isotype controls. To confirm the presence of BCR/ABL, pathologic sections from focal brain lesions or an adjacent region of unaffected brain were dissected using laser-capture microdissection19 and analyzed by PCR using the P210BCR/ABL primers (all 5′ to 3′), AGGAAGATGATGAGTCTCCGGGG (forward) and TCACTGGGTCCAGCGAGAAGGT (reverse); and the ribosomal-associated protein L7 primers, GAAGCTCATCTATGAGAAGGC (forward) and L7 AAGACGAAGGAGCTGCAGAAC (reverse).
Analysis of mouse imatinib mesylate cerebral spinal fluid (CSF) and plasma concentrations
CSF samples were obtained from nonleukemic imatinib mesylate–treated or sham-treated mice as described.20 After CSF was obtained, plasma samples were obtained from the same animals by retro-orbital venipuncture. Concentrations of imatinib mesylate and its metabolite, CGP 74588, were determined by a validated high-pressure liquid chromatography (HPLC)/mass spectrometry (MS) assay.21
Results and discussion
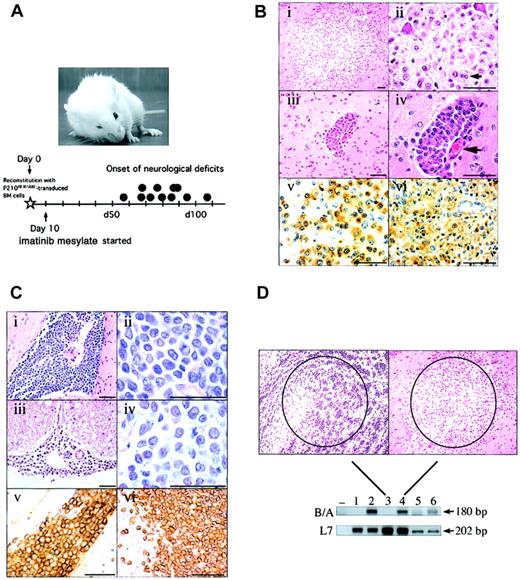
Many of the imatinib mesylate–treated CML mice developed focal and progressive neurologic signs, including altered posture, problems with coordination, and cranial nerve palsy (Figure 1A, upper), from day 56 to day 108 (46 and 98 days of imatinib mesylate treatment, respectively) after BM reconstitution (Figure 1A, lower). Besides modest peripheral blood granulocytosis and splenomegaly, the animals appeared clinically well.
Clinical, pathological, and molecular characterization of Bcr/Abl-induced CNS leukemia. (A) A mouse with CML-like myeloproliferative disorder with right forelimb paresis and right ptosis that developed on imatinib mesylate therapy, and a timeline illustrating the onset of neurologic signs (•; N=11). (B) H&E–stained section of a parenchymal lesion in the cerebral cortex (i-ii) and a meningeal infiltrate in the Virchow Robin space (iii-iv; the associated blood vessel is shown at the large arrow) composed of cells with abundant cytoplasm and punctate nucleoli. Note occasional binucleated forms (ii, small arrow). Anti–Mac-3 IHC of the brain lesion (v) or a control section from the liver of a mouse with Bcr/Abl-induced macrophage leukemia (vi)1,2 confirms the cells are of the macrophage lineage. Scale bars are 50 μM. (C) A massive infiltration of the pyriform sinus (i-ii) and spinal cord meninges (iii-iv) by a homogeneous population of medium-sized cells with scant cytoplasm and indistinct nucleoli. Anti–B-lymphoid cell antibody (CD45R/B220) staining of the pyriform mass (v) and control normal mouse spleen (vi) confirms the B-lymphoid cell lineage. Scale bars are 50 μM. (D) A laser-dissected brain lesion (right photomicrograph; lane 4) and an adjacent area of normal brain (acumbens) composed of small neurons (left photomicrograph; lane 3) were analyzed by PCR using primers specific for the P210BCR/ABL cDNA junction (B/A, top blot). For comparison, a section from the liver of a mouse with macrophage leukemia (lane 6) or normal liver (lane 5) was processed in the same fashion. 32D cells expressing P210 Bcr/Abl (lane 2) or neomycin alone (lane 1), or water only (–), were amplified using the same primers. To control for equivalent specimen quality, samples were also analyzed using primers detecting expression of the ribosomal-associated gene L7. Sizes of the PCR products are shown at right. The results depicted were confirmed in 3 additional PCR experiments from 2 independent sets of tissue dissections. Original magnification, × 10.
Clinical, pathological, and molecular characterization of Bcr/Abl-induced CNS leukemia. (A) A mouse with CML-like myeloproliferative disorder with right forelimb paresis and right ptosis that developed on imatinib mesylate therapy, and a timeline illustrating the onset of neurologic signs (•; N=11). (B) H&E–stained section of a parenchymal lesion in the cerebral cortex (i-ii) and a meningeal infiltrate in the Virchow Robin space (iii-iv; the associated blood vessel is shown at the large arrow) composed of cells with abundant cytoplasm and punctate nucleoli. Note occasional binucleated forms (ii, small arrow). Anti–Mac-3 IHC of the brain lesion (v) or a control section from the liver of a mouse with Bcr/Abl-induced macrophage leukemia (vi)1,2 confirms the cells are of the macrophage lineage. Scale bars are 50 μM. (C) A massive infiltration of the pyriform sinus (i-ii) and spinal cord meninges (iii-iv) by a homogeneous population of medium-sized cells with scant cytoplasm and indistinct nucleoli. Anti–B-lymphoid cell antibody (CD45R/B220) staining of the pyriform mass (v) and control normal mouse spleen (vi) confirms the B-lymphoid cell lineage. Scale bars are 50 μM. (D) A laser-dissected brain lesion (right photomicrograph; lane 4) and an adjacent area of normal brain (acumbens) composed of small neurons (left photomicrograph; lane 3) were analyzed by PCR using primers specific for the P210BCR/ABL cDNA junction (B/A, top blot). For comparison, a section from the liver of a mouse with macrophage leukemia (lane 6) or normal liver (lane 5) was processed in the same fashion. 32D cells expressing P210 Bcr/Abl (lane 2) or neomycin alone (lane 1), or water only (–), were amplified using the same primers. To control for equivalent specimen quality, samples were also analyzed using primers detecting expression of the ribosomal-associated gene L7. Sizes of the PCR products are shown at right. The results depicted were confirmed in 3 additional PCR experiments from 2 independent sets of tissue dissections. Original magnification, × 10.
The neurologic findings were not a direct neurotoxic effect of chronic imatinib mesylate treatment since an animal treated with the same regimen for 118 days was normal. Moreover, a synchronous cohort of mice receiving total body irradiation, followed by reconstitution with syngeneic vector-control BM cells and 7½ months of imatinib mesylate treatment, also did not develop any signs of neurologic impairment (n = 3). Overall, of the 16 imatinib mesylate–treated CML mice studied, 13 developed definite focal neurologic physical findings, and 3 animals had suspicious but nondiagnostic neurologic signs, compared with 0 of 3 imatinib mesylate–treated mice reconstituted with vector control BM cells (P = .02, Fisher exact test). These findings suggested that the neurologic syndromes of mice with the CML-like myeloproliferative disorder on chronic imatinib mesylate therapy were unlikely to be explained by side effects of CNS radiation, imatinib mesylate, or the combination.
To determine the cause of the neurologic deficits, affected mice were examined by complete necropsy. Surprisingly, many of the animals had multifocal brain and meningeal mass lesions. These masses were composed of cells with abundant cytoplasm and an eccentrically placed nucleus (Figure 1Bi-ii), morphologic features of macrophages. Some cells appeared dysplastic, with prominent nucleoli and occasional binucleate forms (Figure 1Bii, small arrow). Some mice had primarily parenchymal brain lesions, while others had isolated cellular infiltrates along the meningeal surface, often forming focal aggregates within the Virchow Robin spaces (Figure 1Biii-iv). Macrophage meningeal infiltrates were also observed along the spinal cord in some animals (data not shown). Analysis of these brain lesions by anti-MAC3 IHC confirmed the cells to be of the macrophage lineage (Figure 1Bv). Interestingly, the cells had the same morphology and IHC features as the previously described Bcr/Abl-induced macrophage tumor (Figure 1Bvi).1,2 However, in contrast to mice with the macrophage tumor disease, animals receiving chronic imatinib mesylate therapy for murine CML had no evidence of macrophage infiltration outside of the CNS (data not shown).
In some cases, brain masses and spinal cord infiltrates were composed of smaller mononuclear cells with oval nuclei, prominent nucleoli, and scant cytoplasm, morphologic features of lymphoblasts (Figure 1C). These cells formed tumorlike lesions in brain parenchyma (Figure 1Ci-ii, pyriform sinus) and also infiltrated the meninges (Figure 1Ciii-iv). Immunostaining with anti-B220 (CD45) antibody confirmed the B-cell nature of these cellular infiltrates (Figure 1Cv). None of the imatinib mesylate–treated mice with CNS lymphoid infiltrates had evidence of peripheral blood lymphocytosis or adenopathy, as might be expected in murine Bcr/Abl-induced B-cell ALL (data not shown).
CNS leukemia and chloroma-like brain masses have not been previously reported in this murine CML model. To determine whether these cells were a manifestation of a Bcr/Abl-induced leukemia, a focal brain lesion (Figure 1D, right) was dissected from paraffin-embedded sections by laser-capture microdissection19 and examined by PCR for the presence of P210BCR/ABL. As a negative control, an adjacent, similarly sized area of normal brain (the acumbens) was analyzed (Figure 1D, left). BCR/ABL was readily detected in the focal brain lesions by PCR (Figure 1D, lane 4) but not detected in unaffected areas (Figure 1D, lane 3), confirming that the infiltrates represented a Bcr/Abl-induced CNS leukemia or chloroma.
The observation that mice developed CNS leukemia while otherwise experiencing excellent systemic response to imatinib mesylate suggested that imatinib mesylate did not penetrate across the blood-brain barrier. To test this hypothesis, a cohort of 9 mice were started on oral imatinib mesylate treatment for several days to ensure steady-state plasma drug concentrations, and parallel blood and CSF samples were obtained and analyzed for the concentration of imatinib mesylate and its metabolite CGP 74588.22 Because an average of only 15 to 20 μL CSF can be obtained from mice without risk of blood contamination,20 CSF samples were pooled and compared with the average plasma values of the entire cohort (Table 1).
Comparison of CSF and plasma imatinib mesylate concentrations
. | Imatinib mesylate . | . | . | . | |||
|---|---|---|---|---|---|---|---|
| . | Drug . | . | Metabolite . | . | |||
| Concentration . | ng/mL . | μM . | ng/mL . | μM . | |||
| Plasma, average | 6958 (± 2082) | 11.8 (± 3.5) | 611 (± 179) | 1.0 (± 0.3) | |||
| CSF, pooled | 45 | 0.08 | 0 | 0 | |||
| Plasma/CSF ratio | 154.6 | NA | |||||
. | Imatinib mesylate . | . | . | . | |||
|---|---|---|---|---|---|---|---|
| . | Drug . | . | Metabolite . | . | |||
| Concentration . | ng/mL . | μM . | ng/mL . | μM . | |||
| Plasma, average | 6958 (± 2082) | 11.8 (± 3.5) | 611 (± 179) | 1.0 (± 0.3) | |||
| CSF, pooled | 45 | 0.08 | 0 | 0 | |||
| Plasma/CSF ratio | 154.6 | NA | |||||
Results depict the average plasma concentrations of imatinib mesylate and CGP 74588 (metabolite) and the pooled CSF values of a cohort of imatinib mesylate—treated mice (N = 9). The standard error for the imatinib mesylate plasma values is shown in parentheses. NA indicates not applicable.
Despite an average plasma imatinib mesylate concentration of 6958 ng/mL (11.8 μM, n = 9; some animals received an additional imatinib mesylate dose 4 to 5 hours after their previous dose to ensure adequate plasma levels at the time of CSF harvest), the concentration of imatinib mesylate in the CSF was only 45 ng/mL (0.08 μM), 155 times lower than in the plasma and 3-fold lower than the level required to achieve 50% inhibition of cellular Bcr/Abl-related tyrosine phosphorylation (0.25 μM).23 The poor penetration of imatinib mesylate into the CSF was not anticipated on the basis of the published data regarding this small-molecule kinase inhibitor. In fact, only one case report has been published concerning concentrations of imatinib mesylate in the CSF. In this report, a patient with CML in lymphoid blast crisis was treated with imatinib mesylate, and although an excellent systemic response was achieved, the patient experienced a lymphoid CNS relapse. Further investigation revealed a CSF imatinib mesylate concentration 2-log lower than in plasma,24 remarkably similar to what was found in our mouse study. These results suggest that the mechanism for limited imatinib mesylate CSF penetration may be shared across species.
The development of CNS lymphoid leukemia in some of our animals on chronic imatinib mesylate therapy shares some features in common with human Ph+ ALL, in which patients treated with imatinib mesylate alone would be considered at high risk for an isolated CNS relapse without appropriate intrathecal chemotherapy.25 Although the Bcr/Abl-induced macrophage tumor illness in mice does not have a known correlate in humans, the striking propensity for these cells to survive and proliferate in the brain led us to search for any evidence of other myeloid lineage cell involvement in the CNS. We found no evidence of granulocytic or myeloblast infiltration of the brain or spinal cord in any of our imatinib mesylate–treated mice, however; although a small aggregation of basophils was found in one animal. This suggests that although the CNS was a potential sanctuary site for malignant lymphoid and macrophage cells in our model, this site did not offer a suitable environment for the survival and expansion of myeloid progenitors responsible for the CML-like illness. Perhaps in humans, the CNS will also prove to be an unsuitable location for CML progenitors to survive and avoid therapeutic doses of imatinib mesylate.
In summary, we have demonstrated that the murine BM retroviral transduction and transplantation model of Bcr/Abl-induced leukemia shares yet another similarity with the human disease: the therapeutic agent imatinib mesylate has a quite limited ability to cross the blood-brain barrier. The feasibility of CSF drug measurements in this CML mouse may prove useful in defining the mechanisms for limited imatinib mesylate penetration across the blood-brain barrier in CNS leukemia and in the study of future anti-CML drugs.
Prepublished online as Blood First Edition Paper, February 20, 2003; DOI 10.1182/blood-2002-10-3059.
Supported by National Institutes of Health grant RO1 CA61764 and the Leukemia Association of North Central Texas.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Elisabeth Buchdunger, Sandra Silberman, and Jose Leis for helpful advice; Robert Parise for assistance with the HPLC/MS CSF and plasma analysis; Shashank Heda and Adi Gazdar for assistance with the laser-capture microdissection; John Shelton for preparing scaled photomicrographs; and David Holtzman for sharing details about mouse CSF harvest prior to publication. We also thank Richard Gaynor and John Minna for critical review of the manuscript.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal