Abstract
Polycythemia vera (PV) is a human clonal hematologic disorder. Previously we demonstrated that erythroid colony-forming cells (ECFCs) from PV patients contained a hyperactive membrane-associated tyrosine phosphatase. We now show that this phosphatase corresponded to protein tyrosine phosphatase (PTP)-MEG2, an intracellular enzyme with a putative lipid-binding domain. The increased activity of PTP-MEG2 in PV cells is due to its elevated distribution in the membrane fraction. With the development of ECFCs to mature red cells, the protein level of PTP-MEG2 decreased gradually, but membrane-associated PTP-MEG2 was sustained for a longer period of time in PV cells, which correlated with an enhanced colony-forming capability of the cells. Importantly, expression of dominant-negative mutant forms of PTP-MEG2 suppressed in vitro growth and expansion of both normal and PV ECFCs. The data indicate that PTP-MEG2 has an important role in the development of erythroid cells. (Blood. 2003;102:4354-4360)
Introduction
Polycythemia vera (PV) is a clonal myeloproliferative disorder characterized by trilineage marrow hyperplasia with increased production of red cells, granulocytes, and platelets.1,2 It usually occurs in older people aged between 40 and 60 years with an annual incidence of about 14 per million in the population. So far there is no effective cure for the disease and phlebotomy is the mainstay of treatment.3 The mortality is high if the disease is untreated or is associated with leukemia. Despite extensive studies in recent years, the molecular etiology of PV remains unknown.1,4
A major feature of PV is that hematopoietic progenitors in patients display hypersensitive responses to many growth factors and cytokines including erythropoietin (EPO), interleukin-3, stem cell factor (SCF), granulocyte-macrophage colony-stimulating factor, and insulin-like growth factor-I.5-16 Despite these abnormal responses, the numbers of receptors for the growth factors and cytokines on the surface of these cells are normal, suggesting a primary defect in a shared signaling pathway in these cells. Protein tyrosine phosphorylation controlled by coordinate actions of protein tyrosine kinases (PTKs) and phosphatases (PTPs) plays a crucial role in the development of cells.17-20 The hypersensitivity of PV hematopoietic progenitor cells to growth factors and cytokines could be due to abnormality in PTPs and/or PTKs. By comparing PTP activity in erythroid colony-forming cells (ECFCs) from PV and normal blood, we recently showed that the PV cells contained a hyperactive membrane or membrane-associated PTP with 2- to 5-fold higher activity above the normal level.21 In the present study, we have identified this PTP and investigated its role in the development of human erythroid cells in vitro.
Materials and methods
Purification of ECFCs and fractionation of cell extracts
Peripheral blood was obtained from healthy volunteers and PV patients. Approval was obtained from the Vanderbilt University institutional review board for these studies. Informed consent was provided according to the Declaration of Helsinki. All the patients met the established criteria for PV3 and had no other disease history. They were treated with phlebotomy only. The blood samples (400 mL) were collected in sodium heparin at a final concentration of 20 U/mL and were used within 24 hours. Purification of erythroid progenitors and subsequent culture from day 1 were performed as previously described.21 By day 8, most cells were erythroid colony-forming units (CFU-Es). Cell extraction, subcellular fractionation, and separation of cell extracts on fast protein liquid chromatography (FPLC) chromatographic columns were carried out as previously described.21
PTP activity assay
Generation of anti-PTP-MEG2 antibodies, Western blotting, and immunofluorescent cell staining
Anti-PTP-MEG2 antibody was generated in rabbits against a glutathione S-transferase (GST) fusion protein carrying PTP-MEG2 and was purified through antigen-affinity chromatography. For Western blotting analyses, cell extracts (usually 10 μg) were separated on 10% sodium dodecyl sulfate (SDS) gels and transferred to polyvinylidene fluoride (PVDF) membranes. Detection was made by using enhanced chemiluminescence (ECL), and quantification of the gel bands was performed by using a gel scanner. For immunofluorescent cell staining, cells were attached to polylysine-coated cover slips and fixed with formaldehyde before staining with affinity-purified anti-PTP-MEG2 and then with Cy3-conjugated antirabbit secondary antibody.
Retrovirus-mediated transfer of PTP-MEG2 and its dominant mutants into ECFCs
We employed retroviral vector pMSCV-IRES-GFP (plasmid murine stem cell virus-internal ribosomal entry site-green fluorescence protein; provided by Dr Arthur Nienhuis, St Jude Children's Research Hospital, Memphis, TN) to carry native PTP-MEG2, Cys515Ser mutant PTP-MEG2 (C→S), and Asp470Ala mutant PTP-MEG2 (D→A). Both mutants are catalytically inactive.23 The pMSCV-IRES-GFP vector has an internal ribosome entry site (IRES) that enables expression of target gene and green fluorescence protein (GFP) as separated proteins from a single transcript initiating in the 5′ long-terminal repeat. GFP is used as a marker to indicate the efficiency of gene transfer. Recombinant retroviruses were generated by transfection of the φNX amphotropic retroviral packaging line with the cDNA constructs by using the Fugene 6 cell transfection reagent (Boehringer Mannheim, Mannheim, Germany). The viruses were recovered in the culture medium after 48 hours of cell transfection and directly used to infect normal or PV day-5 ECFCs following a procedure that uses polybrene and fibronectin-coated plates.24 The infected cells were used for methylcellulose colony assays and liquid cultures as previously described.21
Statistical analysis
This was performed via paired or unpaired t tests by using the INSTAT program (GraphPad, San Diego, CA). The level of significance was P < .05 (2-tailed test). Throughout the manuscript error bars denote standard deviation.
Results
Identification of PTP-MEG2 as the hyperactive PTP in PV ECFCs
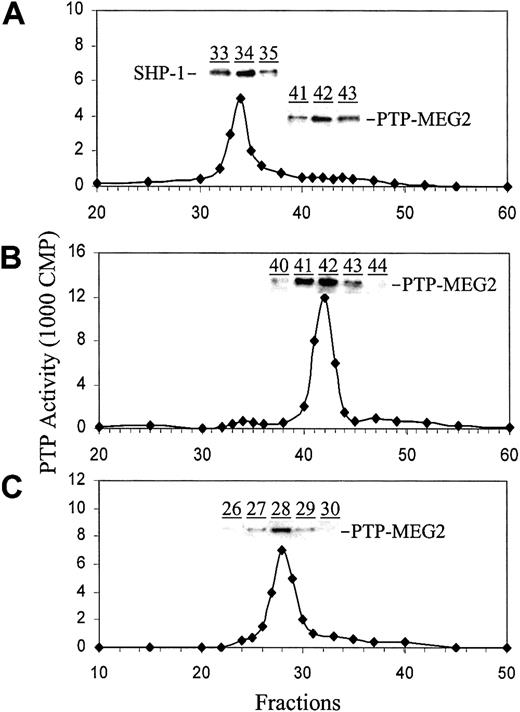
Our earlier studies demonstrated that the PTP activity in the membrane extracts of ECFCs from PV blood was 2 to 5 times higher than that from normal blood. Further fractionation of cell extracts on chromatographic columns revealed that this was likely caused by a single hyperactive PTP in the PV cells.21 To identify the PTP, we first performed reverse transcriptase-polymerase chain reaction (RT-PCR) amplification of cDNA from normal and PV ECFCs with degenerate PCR primers derived from 2 highly conserved regions of PTPs with the consensus sequences of H/DFWRMI/VW and WPDF/HGVP. After sequencing of more than 100 clones, we isolated 9 PTPs including SHP-1 (Src homology 2 domain containing tyrosine phosphatase 1), SHP-2, RPTPα (receptor protein tyrosine phosphatase α), TC-PTP (T-cell-PTP), PTP-MEG1, PTP-MEG2, PTP-PEST (PTP-proline-glutamic acid-serine-threonine), density-enhanced phosphatase (DEP), and CD45. These enzymes do not necessarily represent all the PTPs expressed in erythroid progenitor cells but provided us with major candidates. We then used antibodies against these PTPs to analyze the column fractions of cell extracts from PV and normal ECFCs. When the cytosolic extracts were separated on an anion-exchange Mono-Q column, one major activity peak was observed. Western blot analyses with a specific anti-SHP-1 antibody revealed that this active peak corresponded to SHP-121 (Figure 1A). Separation of the membrane extracts on the Mono-Q column also gave rise to a major activity but at a different position. Importantly, Western blot analyses revealed that anti-PTP-MEG2 immunoreactivity exactly matched the activity profile (Figure 1B). This was also true for the fractions separated on an S12 gel filtration column (Figure 1C). We have also employed antibodies against other PTPs to analyze the Mono-Q and S12 column fractions but none of them showed immunoreactivity that matched the major activity peaks (not shown). These data thus suggest that the abnormal PTP in the membrane fraction of PV ECFCs corresponded to PTP-MEG2. This was further confirmed by immunodepletion experiments that showed more than 90% removal of PTP activity from the Mono-Q column fractions of the membrane extracts by anti-PTP-MEG2 antibodies bound to protein A-agarose (Figure 2). Together, the data also suggest that PTP-MEG2 represents a major PTP activity in the membrane fraction of cell extracts from PV ECFCs. Note that PTP-MEG2 was also present in the cytosolic cell extracts but showed no significant activity (Figure 1A).
Identification of PTP-MEG2 by chromatographic fractionation and immunoblotting. Cytosolic (A) and membrane (B) extracts of day 6 PV ECFCs were separated on an FPLC anionic-exchange Mono-Q column. Samples corresponding to the activity peak in panel B (fractions 41-43) were combined and further separated on an FPLC Superose-12 gel filtration column (C). PTP activity was analyzed as described in “Materials and methods.” Protein levels of PTP-MEG2 and SHP-1 in the indicated fractions were determined by Western blotting analyses with anti-PTP-MEG2 and anti-SHP-1 antibodies, respectively. Representative data from at least 4 independent experiments are shown. Similar results were seen with normal ECFCs except for a lower total PTP activity in the membrane extracts.
Identification of PTP-MEG2 by chromatographic fractionation and immunoblotting. Cytosolic (A) and membrane (B) extracts of day 6 PV ECFCs were separated on an FPLC anionic-exchange Mono-Q column. Samples corresponding to the activity peak in panel B (fractions 41-43) were combined and further separated on an FPLC Superose-12 gel filtration column (C). PTP activity was analyzed as described in “Materials and methods.” Protein levels of PTP-MEG2 and SHP-1 in the indicated fractions were determined by Western blotting analyses with anti-PTP-MEG2 and anti-SHP-1 antibodies, respectively. Representative data from at least 4 independent experiments are shown. Similar results were seen with normal ECFCs except for a lower total PTP activity in the membrane extracts.
Identification of PTP-MEG2 by immunodepletion. Combined Mono-Q column fractions (nos. 40-43; Figure 1B) obtained with membrane extracts of day-6 PV ECFCs were incubated with purified anti-PTP-MEG2 or irrelevant rabbit immunoglobulin G (IgG) bound to protein A-agarose for 4 hours at 4°C. PTP activity in the untreated control and treated supernatant samples were then determined. Data represent relative activity obtained with 3 different samples.
Identification of PTP-MEG2 by immunodepletion. Combined Mono-Q column fractions (nos. 40-43; Figure 1B) obtained with membrane extracts of day-6 PV ECFCs were incubated with purified anti-PTP-MEG2 or irrelevant rabbit immunoglobulin G (IgG) bound to protein A-agarose for 4 hours at 4°C. PTP activity in the untreated control and treated supernatant samples were then determined. Data represent relative activity obtained with 3 different samples.
Expression and intracellular partition of PTP-MEG2 in PV and normal ECFCs
To find any possible structural abnormality of PTP-MEG2, we cloned the entire coding sequence of PTP-MEG2 from PV and normal ECFCs by RT-PCR. Complete sequencing analyses of PCR products and cDNA clones from 4 PV and 2 normal samples confirmed the reported sequence of PTP-MEG2 and revealed no mutation of PTP-MEG2 in the PV cells. We further analyzed PTP-MEG2 at the protein level (Figure 3). Using day-6 ECFCs, we found no significant difference in the protein level of PTP-MEG2. Furthermore, PTP-MEG2 from both normal and PV ECFCs ran at 68 kDa on SDS gels as expected from its primary structure. Figure 3A (top) shows representative protein samples from 4 different PV patients and 4 different healthy volunteers. Note that the specificity of our anti-PTP-MEG2 antibody is verified by a more intensive PTP-MEG2 band seen with normal ECFCs infected with recombinant retrovirus carrying PTP-MEG2 (the last lane in Figure 3A; see also later in Figure 4). We conclude that the elevated PTP-MEG2 activity in PV cells is not caused by a mutation or altered protein expression of the enzyme. PTP-MEG2 is an intracellular enzyme but it contains a putative lipid-binding domain at its N-terminus.25 This structural feature suggests that it may be a membrane-associated enzyme, and thus translocation between the membrane and cytosol may be an important mechanism by which the enzyme is regulated. We then analyzed the partition of the enzyme in the cytosolic and membrane fractions. In at least 6 PV and 5 normal samples analyzed, there was a significantly (P < .001) higher level of PTP-MEG2 in the membrane fraction of PV cells than that in the normal cells. In PV cells, 50% ± 10% of PTP-MEG2 was distributed in the membrane fractions, whereas in the normal cells only 24% ± 8% of PTP-MEG2 was membrane associated. Figure 3B shows representative data from 2 normal and 2 PV blood samples. When the Mono-Q column fractions of the cytosolic extracts were analyzed with anti-PTP-MEG2 antibody, cytosolic PTP-MEG2 was resolved in the column fractions corresponding to 0.3 M NaCl but no correspondent activity was found (Figure 1A). In contrast, the membrane-associated PTP-MEG2 was eluted at the same salt concentration and corresponded to a major PTP activity (Figure 1B). Therefore, the cytosolic PTP-MEG2 is apparently inactive, while the membrane-associated PTP-MEG2 is active, and the enhanced PTP-MEG2 activity in the membrane fraction of the PV cells is due to the elevated distribution of the enzyme in the membrane fraction.
Protein level, distribution, and activity of PTP-MEG2 in normal and PV ECFCs and the correlation with in vitro ECFC development. Normal and PV ECFCs were cultured in conditioned liquid medium and analyzed at the indicated times. (A-C) Whole-cell extracts (A) or cytosolic and membrane fractions (B-C) of day 6 (A-B) or day-8 to -21 (C) ECFCs from different PV patients and healthy volunteers were subjected to Western blot analyses with anti-PTP-MEG2 antibody. The far right lane in panel A represents extracts from normal day-6 ECFCs infected with retrovirus carrying PTP-MEG2 (Figure 4A). (D) Quantitative representation of the protein level of PTP-MEG2 (based on gel scanning) in cytosolic (open symbols) and membrane (closed symbols) fractions of day-8 to day-20 PV (▪) and normal (♦) ECFCs. Data represent the average of at least 4 experiments with different PV and normal samples. (E) PTP activity in the membrane extracts of day-8 to day-20 PV (▪) and normal (♦) ECFCs. (F) For erythroid colony-forming assays, 1000 day-8 to day-20 PV (▪) and normal (♦) ECFCs were seeded in semisolid methylcellulose medium under optimal culture conditions. Erythroid colonies were counted after 10 days of culture.
Protein level, distribution, and activity of PTP-MEG2 in normal and PV ECFCs and the correlation with in vitro ECFC development. Normal and PV ECFCs were cultured in conditioned liquid medium and analyzed at the indicated times. (A-C) Whole-cell extracts (A) or cytosolic and membrane fractions (B-C) of day 6 (A-B) or day-8 to -21 (C) ECFCs from different PV patients and healthy volunteers were subjected to Western blot analyses with anti-PTP-MEG2 antibody. The far right lane in panel A represents extracts from normal day-6 ECFCs infected with retrovirus carrying PTP-MEG2 (Figure 4A). (D) Quantitative representation of the protein level of PTP-MEG2 (based on gel scanning) in cytosolic (open symbols) and membrane (closed symbols) fractions of day-8 to day-20 PV (▪) and normal (♦) ECFCs. Data represent the average of at least 4 experiments with different PV and normal samples. (E) PTP activity in the membrane extracts of day-8 to day-20 PV (▪) and normal (♦) ECFCs. (F) For erythroid colony-forming assays, 1000 day-8 to day-20 PV (▪) and normal (♦) ECFCs were seeded in semisolid methylcellulose medium under optimal culture conditions. Erythroid colonies were counted after 10 days of culture.
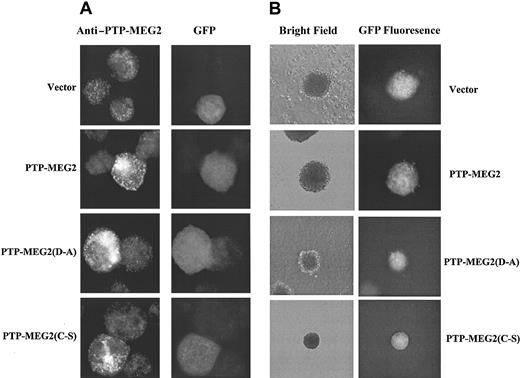
Alteration of PTP-MEG2 expression and the effects on erythroid colony formation. Day-5 PV ECFCs were infected with recombinant retroviruses carrying vector control, PTP-MEG2, PTP-MEG2(D→A), and PTP-MEG2(C→S). Cells were collected 3 days later for immunofluorescent cell staining with anti-PTP-MEG2 (A) and for erythroid colony-forming assays (B). Data show fluorescent anti-PTP-MEG2 staining, GFP fluorescence, and bright field photo images. Note the exposure time for antibody staining of the vector control was longer. Original magnifications, × 1000 (A) and × 40 (B).
Alteration of PTP-MEG2 expression and the effects on erythroid colony formation. Day-5 PV ECFCs were infected with recombinant retroviruses carrying vector control, PTP-MEG2, PTP-MEG2(D→A), and PTP-MEG2(C→S). Cells were collected 3 days later for immunofluorescent cell staining with anti-PTP-MEG2 (A) and for erythroid colony-forming assays (B). Data show fluorescent anti-PTP-MEG2 staining, GFP fluorescence, and bright field photo images. Note the exposure time for antibody staining of the vector control was longer. Original magnifications, × 1000 (A) and × 40 (B).
Correlation of PTP-MEG2 activity with colony-forming ability of ECFCs
The protein level of PTP-MEG2 in ECFCs remained unchanged between day 6 and day 8 of cell culture when most of the cells possessed the capacity to form erythroid colonies. Along with the differentiation of these cells, as evidenced by an increased number of more mature erythroblasts and decreased colony-forming ability from day 8 to day 21 of culture, the protein level of PTP-MEG2 in the membrane fractions of normal cells rapidly declined and little was detected after day 14. Interestingly, PTP-MEG2 in PV cells also declined but at a slower rate and a significant amount of PTP-MEG2 was still maintained by day 17. In contrast, the level of PTP-MEG2 in cytosolic fractions of the PV cells was reduced at a faster rate than that of the normal cells (Figure 3C-D). The slower decrease in the membrane PTP-MEG2 protein in PV cells correlated with a sustained PTP activity and with an increased number of cells that were still capable of forming erythroid colonies at day 11 and thereafter (Figure 3E-F). It should be pointed out that from day 6 to day 10, PV and normal cells gave rise to similar numbers of red cell colonies but the average colony size was larger with the PV cells (data not shown). Furthermore, as the cells progress to maturation in the liquid culture, fewer cells have the ability to form colonies in colony assays, and the colony size gets smaller. Together, these data suggest that PV ECFCs, through possessing a higher level of PTP-MEG2 activity, may have a higher capability to proliferate and/or survive, thus producing more erythrocytes upon maturation and producing the erythroid hyperplasia in PV patients.
Expression of dominant-negative mutant forms of PTP-MEG2 suppresses development of both PV and normal ECFCs in vitro
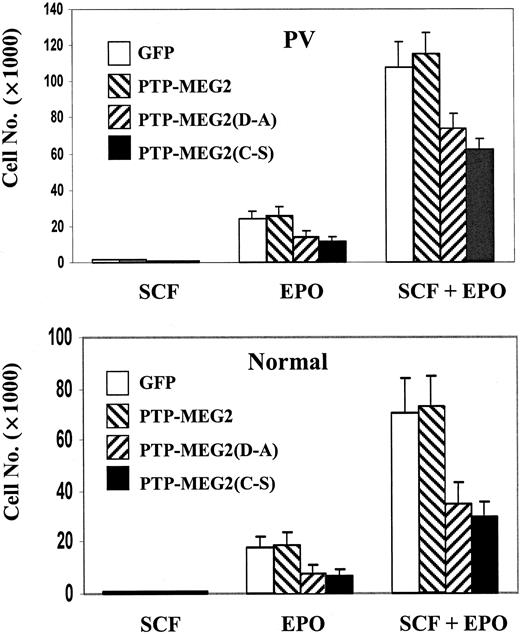
To confirm the role of PTP-MEG2 in the development of human erythroid cells, we altered the expression of PTP-MEG2 in PV and normal ECFCs by using recombinant retroviruses carrying PTP-MEG2 and 2 of its catalytically inactive mutants, namely, PTP-MEG2(C→S) and PTP-MEG2(D→A). Since these mutants have the intact lipid-binding domain that presumably competes with endogenous PTP-MEG2 for interacting with ligands, they may display dominant interfering effects. Upon infection of day-5 PV ECFCs with the recombinant retroviruses assisted by fibronectin and polybrene, we obtained 40% to 50% transfection efficiency as judged by GFP. Figure 4A demonstrates the overexpression of exogenous PTP-MEG2 and its mutant forms by immunofluorescent cell staining. Endogenous PTP-MEG2 displayed vesicular and cytosolic staining, and over-expression of PTP-MEG2 and its mutants caused increased staining in both cytosol and vesicular structure. These data indicate that the exogenously introduced PTP-MEG2 was indeed overexpressed and the mutant forms did not show obvious change in localization. We obtained similar staining with normal ECFCs except that the cytosolic background appeared more intensive (not shown). We then employed semisolid cultures to determine the growth of ECFCs with altered PTP-MEG2 expression. The data obtained with PV cells are summarized in Table 1. Compared with GFP controls, overexpression of native PTP-MEG2 had no significant effects on colony-forming capability of the cells. However, expression of either of the mutant forms caused significant suppression of colony formation, and the suppression was reflected in both colony size and number. The average sizes of colonies obtained with ECFCs expressing the mutant forms of PTP-MEG2 were significantly smaller than those observed with the cells expressing the GFP control or native PTP-MEG2. While essentially no colonies of 500 or more cells were seen with cells expressing mutant forms of PTP-MEG2, approximately 14% of colonies of this size were observed with the control and PTP-MEG2 cells. Figure 4B shows phase contrast and fluorescent photo images of typical colonies obtained with PV ECFCs expressing the different forms of PTP-MEG2. The PTP-MEG2 mutants also significantly reduced the total number of colonies. When cells were cultured with EPO only, an approximate 50% reduction was observed. We also initiated suspension cultures of the infected PV ECFCs. As shown in Figure 5, the mutant forms of PTP-MEG2 decreased the total cell numbers by approximately 50% when cells were cultured with EPO alone and by approximately 35% when both SCF and EPO were present. Again, overexpression of the native form of PTP-MEG2 showed no significant effects. We further performed the same experiments with normal ECFCs that were also efficiently infected with recombinant retroviruses. As expected, expression of mutant forms of PTP-MEG2 resulted in similar suppression of erythroid development (data not shown). Together, these data demonstrate an important role of PTP-MEG2 in the development of erythroid progenitor cells.
Colony formation of PV ECFCs expressing different forms of PTP-MEG2
. | . | No. of GFP-positive colonies . | . | . | . | |||
|---|---|---|---|---|---|---|---|---|
| ECFCs . | Cytokine(s) . | Small . | Medium . | Large . | Total . | |||
| GFP | SCF | 4 ± 2 | 0 | 0 | 4 ± 2 | |||
| GFP | EPO | 32 ± 5 | 5 ± 2 | 0 | 37 ± 6 | |||
| GFP | SCF+EPO | 39 ± 6 | 45 ± 8 | 14 ± 6 | 98 ± 12 | |||
| MEG2 | SCF | 7 ± 3 | 0 | 0 | 7 ± 3 | |||
| MEG2 | EPO | 34 ± 7 | 3 ± 2 | 0 | 37 ± 8 | |||
| MEG2 | SCF+EPO | 41 ± 9 | 51 ± 10 | 14 ± 4 | 106 ± 15 | |||
| MEG2(D→A) | SCF | 5 ± 2 | 0 | 0 | 5 ± 2 | |||
| MEG2(D→A) | EPO | 20 ± 3 | 0* | 0 | 20 ± 3* | |||
| MEG2(D→A) | SCF+EPO | 46 ± 8 | 32 ± 5* | 2 ± 2* | 80 ± 9* | |||
| MEG2(C→S) | SCF | 2 ± 1 | 0 | 0 | 2 ± 1 | |||
| MEG2(C→S) | EPO | 16 ± 4 | 1 ± 1* | 0 | 17 ± 4* | |||
| MEG2(C→S) | SCF+EPO | 36 ± 7 | 26 ± 4* | 1 ± 1* | 63 ± 9* | |||
. | . | No. of GFP-positive colonies . | . | . | . | |||
|---|---|---|---|---|---|---|---|---|
| ECFCs . | Cytokine(s) . | Small . | Medium . | Large . | Total . | |||
| GFP | SCF | 4 ± 2 | 0 | 0 | 4 ± 2 | |||
| GFP | EPO | 32 ± 5 | 5 ± 2 | 0 | 37 ± 6 | |||
| GFP | SCF+EPO | 39 ± 6 | 45 ± 8 | 14 ± 6 | 98 ± 12 | |||
| MEG2 | SCF | 7 ± 3 | 0 | 0 | 7 ± 3 | |||
| MEG2 | EPO | 34 ± 7 | 3 ± 2 | 0 | 37 ± 8 | |||
| MEG2 | SCF+EPO | 41 ± 9 | 51 ± 10 | 14 ± 4 | 106 ± 15 | |||
| MEG2(D→A) | SCF | 5 ± 2 | 0 | 0 | 5 ± 2 | |||
| MEG2(D→A) | EPO | 20 ± 3 | 0* | 0 | 20 ± 3* | |||
| MEG2(D→A) | SCF+EPO | 46 ± 8 | 32 ± 5* | 2 ± 2* | 80 ± 9* | |||
| MEG2(C→S) | SCF | 2 ± 1 | 0 | 0 | 2 ± 1 | |||
| MEG2(C→S) | EPO | 16 ± 4 | 1 ± 1* | 0 | 17 ± 4* | |||
| MEG2(C→S) | SCF+EPO | 36 ± 7 | 26 ± 4* | 1 ± 1* | 63 ± 9* | |||
Five hundred day-5 PV ECFCs infected with recombinant retroviruses carrying GFP control, PTP-MEG2, PTP-MEG2(D→A), and PTP-MEG2(C→S) were cultured in methylcellulose colony culture medium in the presence of SCF (100 ng/mL), EPO (4 units/mL), or SCF plus EPO. Erythroid cell colonies were counted 9 days later in situ using an inverted fluorescence microscope. Colonies were divided into small (50-100 cells), medium (100-500 cells), and large (>500 cells) categories. Data represent mean ± SD of at least 4 independent experiments. *Significantly different from the correspondent control (GFP and MEG2) data (P < .05).
PTP-MEG2 mutants affect growth of ECFCs in suspension culture. Two thousand day-5 PV (top) or normal (bottom) ECFCs infected with recombinant retroviruses carrying GFP control, PTP-MEG2, PTP-MEG2(D→A), and PTP-MEG2(C→S) were cultured in the presence of SCF, EPO, or EPO plus SCF. Cells were attached to glass sides by cytospin, and GFP-positive cells were counted under a fluorescence microscope. Data represent mean ± SD of at least 3 independent experiments.
PTP-MEG2 mutants affect growth of ECFCs in suspension culture. Two thousand day-5 PV (top) or normal (bottom) ECFCs infected with recombinant retroviruses carrying GFP control, PTP-MEG2, PTP-MEG2(D→A), and PTP-MEG2(C→S) were cultured in the presence of SCF, EPO, or EPO plus SCF. Cells were attached to glass sides by cytospin, and GFP-positive cells were counted under a fluorescence microscope. Data represent mean ± SD of at least 3 independent experiments.
Discussion
In the present study, we demonstrated that hyperactive PTP activity in the membrane fraction of PV ECFCs corresponds to PTP-MEG2, an intracellular PTP with a putative lipid-binding domain. We further demonstrated that the increased PTP activity is due to elevated and sustained distribution of the enzyme on the membrane where the enzyme is activated. The increased activity of PTP-MEG2 in PV ECFC membranes correlated with increased cell colony formation of ECFCs. Furthermore, expression of dominant-negative mutant forms of PTP-MEG2 significantly suppressed growth and expansion of both normal and PV ECFCs in vitro. The data indicate an important role of PTP-MEG2 in the development of human erythroid cells. Our study thus provides a potential target for development of therapeutic drugs to reduce red cell production.
The molecular basis of PV has been extensively studied in recent years. Silva et al26 demonstrated an elevated expression of the antiapoptotic protein Bcl-xL in PV erythroid progenitors. Moliterno et al27,28 reported that the expression of the thrombopoietin (TPO) receptor was virtually nondetectable in PV platelets due to altered glycosylation. Temerinac et al29 isolated a novel cell surface receptor named polycythemia rubra vera 1 (PRV-1), which is highly expressed in granulocytes from patients with PV. In a separate study, we recently found that tumor suppressors p16 (INK4a) and p14 (ARF) mRNA levels are enhanced in ECFCs from PV patients.30 Although none of the studies revealed a mutation in the sequences of the affected proteins, all suggest a possible abnormality in the control of gene expression or modification of key signaling mediators in PV cells. Our current study suggests abnormal regulation of a signaling enzyme at the protein level and thus provides a regulated enzyme as a potential target to treat PV. Defining specific targets is a crucial step in developing therapeutic drugs to treat diseases. For example, use of the BCR/Abl tyrosine kinase as a specific target has led to the development of imatinib mesylate, an inhibitor of the kinase, which has been proven to be a remarkably effective drug for the treatment of chronic myelogenous leukemia.31 Of course, whether PTP-MEG2 can be used as a target for drug screening also depends on its function in other cellular processes. Our study demonstrated that PTP-MEG2 is a major PTP activity in erythroid progenitor cells; it may have a primary role in erythropoiesis.
The enhancement of erythropoiesis in PV may lie in the upstream activator of PTP-MEG2. PTP-MEG2 is an intracellular PTP of 593 amino acids.25 Its C-terminal region has a catalytic domain, while the N-terminal region contains a putative lipid-binding domain. Recent studies showed that removal of the N-terminal domain significantly activated the enzyme, suggesting its regulatory role.23,32 Furthermore, PTP-MEG2 has been shown to be partially localized in intracellular membranes at the secretory vesicles.32,33 In this study, we have demonstrated that PTP-MEG2 is present in both cytosol and membrane fractions of ECFCs. In PV cells, a higher level of PTP-MEG2 is located in membranes where it is highly activated. We postulate that PTP-MEG2 stays inactive in the cytosol and is activated by translocation to membranes, which is mediated by a lipid or lipidlike substance. This partly explains why overexpression of the native form of PTP-MEG2 had no significant effects on the development of ECFCs. When activators of the enzyme are limited, increases in the protein level of the enzyme should not cause a significant increase in its enzymatic activity and thereby biologic function. This is reminiscent of the effects of SHP-2 and its catalytically inactive mutant dominant on growth factor-induced signaling transduction.34 For this reason, we attempted to express constitutively active PTP-MEG2 by targeting the enzyme to cell membranes with a myristoylation sequence derived from the 15 N-terminal amino acid residues of human c-Src attached to the N-termini of full-length form and catalytic domain of PTP-MEG2. However, retrovirus-mediated expression of the myristoylated forms of PTP-MEG2 did not enhance the colony-forming ability of erythroid progenitor cells (data not shown). We attributed this lack of effectiveness to the fact that PTP-MEG2 was not targeted to the right membrane compartments. Myristoylation primarily targets to the plasma membrane, but data from previously published studies32,33 and this study (Figure 4) showed that PTP-MEG2 is localized to intracellular membranes. We believe that identification of an activator of PTP-MEG2 should solve the problem. The N-terminal putative lipid-binding domain of PTP-MEG2 shares 24% to 29% sequence identity with cellular retinaldehyde binding protein (CRALBP), α-tocopherol transfer protein, and yeast SEC14P,25,35 which bind retinal, α-tocopherol, and phosphatidylinositol, respectively. Interestingly, a study by Kruger et al32 demonstrated that a GST fusion protein of PTP-MEG2 is significantly activated by phosphatidylinositol 4,5-diphosphate and other phospho-derivatives of phosphatidylinositol. However, with a nonfusion protein purified from an adenovirus expression system, we failed to show such an activation.23 Instead, our recent studies demonstrated a specific binding of PTP-MEG2 with phosphatidylserine through the putative lipid-binding domain but the lipid does not activate the enzyme in vitro.36 What activates PTP-MEG2 PV ECFCs remains to be identified. Isolation of a specific activator of PTP-MEG2 is not only important for studying its function but could also have an important therapeutic implication in boosting the number of red cells produced.
Tyrosine phosphatases play positive as well negative roles in cell signaling.17-20 Our study demonstrated 2 major PTP activities in the cell extracts of ECFCs: one is PTP-MEG2 and the other is SHP-1. SHP-1 is a key negative regulator in hematopoietic signaling. Moth-eaten and viable moth-eaten mice, which lack functional SHP-1 activity due to natural mutation of the SHP-1 gene, are characterized by a high proliferation of all hematopoietic lineages including erythroid progenitors.37,38 In addition, like PV cells, erythroid progenitor cells from moth-eaten and viable moth-eaten mice are hypersensitive to EPO.39 We and others21,40,41 have demonstrated that activity and expression of SHP-1 is normal in PV cells. Here we show that the activity of PTP-MEG2 is elevated in PV erythroid progenitors and is at least partly required for the growth of erythroid cells. Therefore, PTP-MEG2 and SHP-1 display opposite functions in erythropoiesis, and the balance between their activities is crucial for the normal development of erythroid cells. Our earlier study showed that PV and normal ECFCs displayed different responses to treatment by vanadate, a nonselective inhibitor of PTPs.42 This may be due to differential response of SHP-1 and PTP-MEG2 to the inhibitor. The signal transduction mechanism by which PTP-MEG2 affects growth and expansion of erythroid progenitor cells remains to be examined. Recent reports demonstrated a role of PTP-MEG2 in the regulation of secretory vesicles in hematopoietic cells and possibly in phagocytosis.32,33 How this is related to the role of PTP-MEG2 in erythroid progenitor cell proliferation still needs to be clarified. Identification of the direct downstream target of PTP-MEG2 may help to clarify this.
Prepublished online as Blood First Edition Paper, August 14, 2003; DOI 10.1182/blood-2003-04-1308.
Supported by grants CA75218 (Z.J.Z.), DK-15555 (S.B.K.), and CA-68485 (Vanderbilt-Ingram Cancer Center) from the National Institutes of Health and a Veterans Health Administration Merit Review grant (S.B.K.). M.-j.X. is supported by a fellowship grant from the Lymphoma Research Foundation.
M.-j.X. and X.S. contributed equally to this manuscript.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.



This feature is available to Subscribers Only
Sign In or Create an Account Close Modal