Abstract
In this study we demonstrate that thrombopoietin (TPO)–stimulated Src family kinases (SFKs) inhibit cellular proliferation and megakaryocyte differentiation. Using the Src kinase inhibitors pyrolopyrimidine 1 and 2 (PP1, PP2), we show that TPO-dependent proliferation of BaF3/Mpl cells was enhanced at concentrations that are specific for SFKs. Similarly, proliferation is increased after introducing a dominant-negative form of Lyn into BaF3/Mpl cells. Murine marrow cells from Lyn-deficient mice or wild-type mice cultured in the presence of the Src inhibitor, PP1, yielded a greater number of mature megakaryocytes and increased nuclear ploidy. Truncation and targeted mutation of the Mpl cytoplasmic domain indicate that Y112 is critical for Lyn activation. Examining the molecular mechanism for this antiproliferative effect, we determined that SFK inhibitors did not affect tyrosine phosphorylation of Janus kinase 2 (JAK2), Shc, signal transducer and activator of transcription (STAT)5, or STAT3. In contrast, pretreatment of cells with PP2 increased Erk1/2 (mitogen-activated protein kinase [MAPK]) phosphorylation and in vitro kinase activity, particularly after prolonged TPO stimulation. Taken together, our results show that Mpl stimulation results in the activation of Lyn kinase, which appears to limit the proliferative response through a signaling cascade that regulates MAPK activity. These data suggest that SFKs modify the rate of TPO-induced proliferation and are likely to affect cell cycle regulation during megakaryocytopoiesis.
Introduction
Thrombopoietin (TPO) is an essential hematopoietic cytokine that regulates megakaryocytopoiesis via the activation of its cognate receptor, Mpl, expressed on hematopoietic stem cells and megakaryocytic cells (for reviews, see Drachman,1 Geddis et al,2 and Kaushansky3 ). Like other cytokine receptors, Mpl does not contain a kinase domain but, rather, activates members of the receptor-associated Janus kinase (JAK) family on TPO stimulation. Accumulated evidence shows that JAK2 is critical for megakaryocyte (MK) development and that once activated, JAK2 phosphorylates the fourth tyrosine of the Mpl cytoplasmic domain (Y112) and associated signaling molecules, resulting in the activation of the Jak/signal transducer and activator of transcription (STAT) and Ras/Raf/MAPK pathways.4-8 The JAK/STAT pathway is essential for TPO-stimulated proliferation, whereas the activation of mitogen-activated protein kinase (MAPK) promotes differentiation, especially during sustained activation.9,10 TPO activation of both the JAK/STAT and MAPK pathways has been well established in both cell lines and primary cells.11-13 However, the complexity of signaling networks suggests that other cellular kinases, such as the Src family of tyrosine kinases, may be activated following TPO/Mpl interaction.
The Src kinase family consists of 8 members (Src, Yes, Fgr, Fyn, Lck, Lyn, Blk, and Hck) that regulate a variety of cellular functions including proliferation, differentiation, and migration, depending on cellular milieu.14,15 Comparison of all 8 Src family kinases (SFKs) reveals a modular organization that determines subcellular organization, protein-protein interactions, and function.16 The amino-terminal myristoylation and palmitoylation sequences direct SFKs to the inner surface of the cell membrane. The Src homology 3 (SH3) domain directs association with polyproline motifs on interacting signaling molecules, whereas the SH2 domain binds phosphotyrosine residues in a sequence-specific manner. The kinase domain contains an adenosine triphosphate (ATP)–binding site and a catalytic tyrosine residue that undergoes autophosphorylation during activation. Lastly, a highly conserved tyrosine at the carboxyl terminus blocks catalytic activity via intramolecular interaction with the SH2 domain when phosphorylated.
Although Src is nearly ubiquitous in human tissues, many family members are selectively expressed in hematopoietic cells.17 Src kinases have been linked to signaling by a number of cytokine receptors, including interleukin 5 (IL-5), IL-3, granulocyte colony-stimulating factor (G-CSF), and erythropoietin receptor (EPO-R), as well as tyrosine kinase receptors, such as platelet-derived growth factor receptor (PDGF-R) and c-Kit.18-23 In addition, several different SFK members have been shown to coprecipitate with hematopoietic growth factor receptors.20,24-26 It has been difficult to assess the critical role of SFKs in knock-out experiments because multiple family members are present in most cells and can frequently function interchangeably. Some substrates of phosphorylation by Src kinases include receptors and STAT proteins.21,27 It has been demonstrated that in certain model systems, Src kinases rather than Janus kinases are responsible for activation of STAT3 and STAT5.28,29 Furthermore, it has been reported that Src kinases can bind directly to Janus kinases, suggesting that these 2 tyrosine kinases may regulate one another.21,30 Defects in the Src family tyrosine kinases have been observed in patients with hematologic disease.14 Additionally, inhibitors of Src kinases have been shown to block blood cell function and leukemic cell growth.31
Despite the fact that many SFKs are known to be present in MKs and platelets, no role for these kinases have previously been defined in MK development.32,33 In this report, we provide convincing evidence that TPO stimulation results in activation of SFKs and that this signaling event reduces the proliferative response in cell lines and primary cells. We examine the inhibition of SFKs in a TPO-responsive cell line (BaF3/Mpl), and in primary murine MKs and marrow progenitors. These results demonstrate that of Src kinases negatively regulate TPO-induced proliferation and megakaryocytopoiesis, perhaps through limiting the duration and intensity of MAPK activation.
Materials and methods
Cell culture and culture conditions
BaF3 cells expressing full-length and truncated forms of the Mpl receptor were previously described.34 Cells were maintained in RPMI 1640 (BioWhittaker, Walkersville, MD) containing 10% heat-inactivated fetal calf serum (FCS), 2 mM l-glutamine, 100 U/L penicillin, 100 mg/mL streptomycin (BioWhittaker) supplemented with murine IL-3 (mIL-3; 0.2% vol/vol conditioned medium containing recombinant mIL-3, 1.5 ng/mL), and 1000 μg/mL of the selectable antibiotic G418 (Invitrogen, Carlsbad, CA). Wild-type (Lynwt) and mutated Lyn (lysine 275 to leucine; Lyn (K275L)) cDNAs were kindly provided by Diana Linnekin (National Cancer Institute, Frederick, MD).35 Both cDNAs were subcloned into the BamHI site of the expression vector pCDNA3.1 (Invitrogen) and proper cDNA orientation was confirmed by fluorescent sequencing (BigDye Terminator; PE Biosystems, Foster City, CA). BaF3/Mpl transfections with lynwt and lyn (K275L) cDNA were done by electroporation (300 V and 800 microfarads [μF]) in the presence of 20 to 30 μg linearized plasmid. After 24 hours, cells were selected with 500 μg/mL phleomycin 01 (Zeocin; Invitrogen). Individual clones were isolated through limiting dilution in 96-well plates. In BaF3/Mpl, BaF3/Mpl/Lynwt, and BaF3/Mpl/Lyn (K275L) cell lines, flow cytometry analysis confirmed equal Mpl surface expression between full-length and truncated receptor mutants.
Cellular extracts
Cells were washed twice to remove traces of serum and IL-3 and were then starved for 12 hours in RPMI containing 0.5% bovine serum albumin (BSA) and 2 mM l-glutamine. Equal numbers of cells were either unstimulated or stimulated for 10 minutes with the addition of 15 ng/mL murine TPO (mTPO; 2% vol/vol; conditioned medium). After washing once in cold phosphate-buffered saline (PBS), the cell pellet was resuspended in lysis buffer (50 mM HEPES [N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid], pH 7.4, 150 mM NaCl, 10% glycerol, 1% Triton X-100, 1.5 mM MgCl2, 1 mM EGTA [ethylene glycol tetraacetic acid], 100 mM NaF, 1 mM phenylmethylsulfonyl fluoride, 1 mM NaVO4, 1 μg/mL leupeptin, and 1 μg/mL aprotinin) for 15 minutes on ice. Whole-cell lysates were obtained by centrifugation at 14 000g for 15 minutes at 4° C, and the soluble protein was stored at –80°C until use.
Proliferation assay
BaF3 cells were washed twice in RPMI and resuspended in RPMI 1640 containing10% heat-inactivated FCS and 2 mM l-glutamine. Then, 50 000 cells in 50 μL were aliquoted into individual wells of a 96-well plate. Pryolopyrimidine 1 (PP1) or PP2 (Calbiochem, La Jolla, CA) was dissolved in dimethyl sulfoxide (DMSO) and added to cells at various concentrations 40 minutes prior to cytokine stimulation; all inhibitor dilutions were 1:1000. An equal concentration of DMSO (0.1%) was used in the control wells. Then, TPO (2% vol/vol; conditioned medium, 15 ng/mL) or mIL-3–conditioned media (maximal proliferative dose) was added, and cells were grown in a humidified incubator at 37°C and 5% CO2. After 48 hours, proliferation was measured with a 3-(4,5-dimethylthiazol-2-yl)-5-(carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) proliferation kit according to the instructions supplied by the manufacturer (Promega, Madison, WI). Absorbance values (490 nm) were recorded on triplicate samples using a Bio-Rad plate reader (Hercules, CA).
Western blotting
Protein concentrations were determined using a modified Lowry assay (Protein D/C; Bio-Rad). For each sample, 25 μg total protein was denatured by boiling for 10 minutes in loading buffer containing sodium dodecyl sulfate (SDS) and β-mercaptoethanol and was separated on a 10% polyacrylamide gel with prestained molecular weight markers (Bio-Rad). Transfer to nitrocellulose, blocking, probing with antibodies, and chemiluminescence were performed as previously described.13 Membranes were probed with the following primary antibodies: monoclonal antiphosphotyrosine (4G10), polyclonal anti-JAK2, polyclonal antiphospho-JAK2 (Y1007/Y1008), polyclonal anti-Shc (Upstate Biotechnology, Lake Placid, NY), polyclonal antiphospho-STAT3, polyclonal anti-STAT3, polyclonal antiphospho-STAT5, polyclonal anti-STAT5, polyclonal antiphospho-Akt, polyclonal anti-Akt, polyclonal antiphospho–extracellular signal–related kinase 1 and 2 (Erk1/2), polyclonal anti-Erk1/Erk2 (Cell Signaling Technology, Beverly, MA), and polyclonal Mpl antiserum (kindly provided by Amgen, Thousand Oaks, CA). Secondary antibodies, horseradish peroxidase-coupled goat antimouse IgG, or goat antirabbit were purchased from Bio-Rad.
Lyn/Fyn kinase assays
Equal amounts of protein from whole-cell lysates (1.2 mg) were precleared with 25 μL protein A/G Plus-agarose beads (Santa Cruz Biotechnology, Santa Cruz, CA) for 4 hours at 4°C with continuous mixing. Precleared lysates were recovered by centrifugation and incubated with either a polyclonal Lyn antibody (Lyn 44; Santa Cruz Biotechnology), or polyclonal Fyn antibody (FYN4; Santa Cruz Biotechnology) overnight at 4°C. Protein A/G Plus-agarose beads were then added and incubated for 2 additional hours at 4°C. The pellets were washed once with lysis buffer and 3 times with kinase buffer (100 mM Tris [tris(hydroxymethyl)aminomethane]–HCl, pH 7.2, 125 mM MgCl2, 25 mM MnCl2, 2 mM EGTA, 250 μM NaVO4, 2 mM dithiothreitol [DTT]). After the final wash, 10 μL of 1 mg/mL [Lys19] cdc2(6-20) peptide substrate (Upstate Biotechnology), 10 μL ATP mix (500 μM ATP containing 10 μCi γ-32P] ATP [0.37 MBq]), and 30 μL kinase buffer were added sequentially. Reaction mixtures were incubated at 30°C with agitation for 10 minutes before stopping the reaction by adding 20 μL 40% trichloroactetic acid (TCA). Following a 5-minute room temperature incubation, 25 μL of each sample was spotted onto the center of a P81 ion-exchange paper square. Filter squares were washed, dried, and counted to quantify adsorbed radioactivity (LS6500 scintillation counter; Beckman Coulter, Fullerton, CA).
MAPK assay
MAPK activity in BaF3/Mpl cell lysates was measured as the ability to phosphorylate Elk-1. Whole-cell lysate (200 μL; 250 μg total protein) was incubated overnight at 4°C with 15 μL agarose-immobilized phospho-44/42 MAPK monoclonal antibody (Cell Signaling Technology). Immunocomplexes were isolated by centrifugation at 4°C, and pellets were washed 5 times with kinase buffer (25 mM Tris-HCl, pH 7.2, 5 mM β-glycerolphosphate, 2 mM DTT, 0.1 mM Na3VO4, 10 mM MgCl2). Following the last wash, pellets were resuspended in 50 μL kinase buffer supplemented with 200 μM ATP and 2 μg Elk-1 fusion protein (Cell Signaling Technology). After 30 minutes at 30°C, the reaction was stopped by the addition of SDS sample buffer. Samples were then separated on a 10% polyacrylamide gel, transferred to nitrocellulose, and probed with a phospho-specific anti–Elk-1 antibody (Cell Signaling Technology). Membranes were then incubated with goat antirabbit IgG coupled to horseradish peroxidase and visualized using enhanced chemiluminescence (Perkin Elmer Life Sciences, Boston, MA). Quantitation of immunoreactive bands was performed by using the Kodak Image Station 440cf (Rochester, NY).
Murine bone marrow cell culture
C57BL/J (Jackson Laboratories, Bar Harbor, ME) and Lyn-deficient (generously provided by Janet Oliver, University of New Mexico, Albuquerque) mice were killed, and femurs and tibias were removed. The bone marrow was flushed from the bones with Iscove modified Dulbecco medium (IMDM) containing 2% heat-inactivated FCS. The dilute bone marrow was filtered through 70-μm mesh to remove bone particles; red blood cells (RBCs) were lysed in hypotonic buffer; and nucleated cells were counted and resuspended (1 × 106 cells/mL) in IMDM supplemented with 1% Nutridoma-SP (Roche Molecular Biochemicals, Indianapolis, IN), and 2mM l-glutamine. Recombinant mTPO was added (in the form of enriched culture medium) to achieve a final concentration of 37.5 ng/mL. In the C57BL/J bone marrow cultures the Src kinase inhibitor PP1 was added (concentration range, 0-25 μM) resulting in a final concentration of 0.1% DMSO in all samples. After 72 hours of growth (37°C, 5% CO2, humidified incubator), cells were stained with anti-CD41–fluorescein isothiocyanate (FITC; BD PharMingen, San Diego, CA) and propidium iodide to analyze ploidy by flow cytometry as previously described.8
Results
Stimulation of BaF3/Mpl cells with TPO activates Src kinases
Five SFK members were detected by Western blot analysis of BaF3 cell lysates (Blk, Fyn, Lck, Lyn, and Yes; data not shown). To determine whether TPO activates Src family members, we used BaF3 cells engineered to express Mpl (BaF3/Mpl). Whole-cell lysates were generated from cells that were either unstimulated or exposed to exogenous TPO. In the kinase assay, 2 peptide substrates were used: [Lys19] cdc2(6-20)-NH2, an efficient substrate for all Src tyrosine kinases, and [Lys19Ser14Val12] cdc2(6-20)-NH2, a negative control, which has 2 critical substitutions resulting in a decrease in substrate efficiency (Upstate Biotechnology). As shown in Figure 1A, TPO stimulation had no effect on kinase activity of the parental BaF3 cells. However, cells expressing Mpl demonstrated a 2-fold increase in 32P incorporation of the substrate peptide, but not in the control peptide. These results indicate that TPO/Mpl binding activates Src kinases in BaF3/Mpl cells.
TPO-induced activation of Src kinases. Parental BaF3 cells and those engineered to express c-Mpl were starved overnight. Half of the cells were stimulated with TPO (15 ng/mL) for 10 minutes before making whole-cell lysates. (A) Kinase assays were performed using γ-32P] ATP and peptides that function as efficient ([Lys19] cdc2(6-20)-NH2) and inefficient ([Lys19Ser14Val12] cdc2(6-20)-NH2) substrates for Src kinases. (B) Immune complex kinase assay. Lysates normalized for total protein concentration were immunoprecipitated with either anti-Lyn or anti-Fyn antibodies. Kinase activity was measured by phosphorylation of substrate ([Lys19] cdc2(6-20)-NH2) in immune complexes. (C) Cells were pretreated for 45 minutes with DMSO (vehicle = 0) or the stated concentration of PP2 and stimulated with TPO. Lysates were immunoprecipitated with anti-Lyn, after which immune complex kinase assays were performed. The data have been normalized as a percentage of maximal kinase activity induced by TPO in the presence of DMSO alone. Immunoblots of immunoprecipitated lysates (A-B) were performed to ensure equal amounts of Lyn and Fyn were assayed (data not shown). All assay data are representative of 3 independent experiments. Error bars indicate SD.
TPO-induced activation of Src kinases. Parental BaF3 cells and those engineered to express c-Mpl were starved overnight. Half of the cells were stimulated with TPO (15 ng/mL) for 10 minutes before making whole-cell lysates. (A) Kinase assays were performed using γ-32P] ATP and peptides that function as efficient ([Lys19] cdc2(6-20)-NH2) and inefficient ([Lys19Ser14Val12] cdc2(6-20)-NH2) substrates for Src kinases. (B) Immune complex kinase assay. Lysates normalized for total protein concentration were immunoprecipitated with either anti-Lyn or anti-Fyn antibodies. Kinase activity was measured by phosphorylation of substrate ([Lys19] cdc2(6-20)-NH2) in immune complexes. (C) Cells were pretreated for 45 minutes with DMSO (vehicle = 0) or the stated concentration of PP2 and stimulated with TPO. Lysates were immunoprecipitated with anti-Lyn, after which immune complex kinase assays were performed. The data have been normalized as a percentage of maximal kinase activity induced by TPO in the presence of DMSO alone. Immunoblots of immunoprecipitated lysates (A-B) were performed to ensure equal amounts of Lyn and Fyn were assayed (data not shown). All assay data are representative of 3 independent experiments. Error bars indicate SD.
We have previously shown that TPO induces the activity of both Lyn and Fyn kinase in primary murine MKs.33 To determine if Lyn and Fyn are activated in BaF3/Mpl cells, both kinases were immunoprecipitated from the lysates of cells unstimulated or stimulated with TPO and kinase activity was measured using in vitro phosphorylation assays. Lyn kinase activity, measured as [Lys19]cdc2(6-20) phosphorylation, was induced by TPO stimulation. Compared to unstimulated BaF3/Mpl cells, brief TPO exposure induced a 2- to 3-fold increase in Lyn activity but no change in Fyn activity (Figure 1B). This represents a significant difference in the utilization of SFKs between BaF3 cells and primary MKs. These results suggest that lineage-specific signaling molecules other than the Mpl receptor must be involved in the recruitment and activation of SFKs.
To further extend these observations we examined the activation of Lyn by TPO in the presence of the Src kinase inhibitor PP2. Cells were incubated with different concentrations of PP2 for 45 minutes before TPO stimulation. Figure 1C shows that PP2 inhibited Lyn activation in a dose-dependent manner. The inhibitory concentration of 50% (IC50) appears to be approximately 0.25 μM in the BaF3 cell line. Reprobing the blots with an antibody against Lyn confirms that similar amounts of protein were immunoprecipitated from each sample (data not shown). Based on these observations, TPO appears to be a direct regulator of Lyn activity in BaF3/Mpl cells.
The membrane-distal portion of the Mpl cytoplasmic domain is required for the activation of Lyn kinase
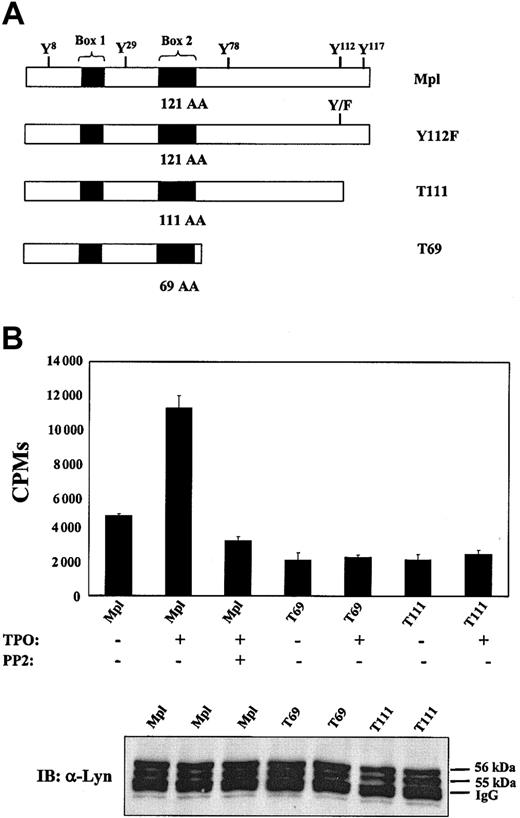
The cytoplasmic domain of murine Mpl contains 121 amino acids. To determine which regions of the Mpl receptor are necessary for activation of SFKs, we studied 2 truncated mutants of Mpl (T69, deletion of cytoplasmic residues 70-121; and T111, deletion of residues 112-121) and 1 point mutation (Y112F), replacing the primary site of receptor tyrosine phosphorylation with phenylalanine (Figure 2A). These receptors and their signaling and growth characteristics have been previously described.34 After starving each BaF3 cell line for 10 hours under serum-free, cytokine-free conditions, TPO was added for 10 minutes and cellular lysates were generated. Lysates were normalized for total protein concentration, immunoprecipitated with Lyn polyclonal antibody, and assayed for Lyn activity with the γ-32P]ATP transfer assay. As shown in Figure 2B, some basal Lyn activity was observed in unstimulated cells. However, exposure to TPO for 10 minutes significantly increased the amount of active Lyn kinase in BaF3/Mpl cells. In contrast, TPO stimulation of T69 and T111 was unable to induce Lyn activity (Figure 2B). These results not only support our previous finding that TPO/Mpl stimulation activates Lyn kinase, but suggest that Y112, the major site of receptor tyrosine phosphorylation, is critical for SFK activation.
Carboxyl region of Mpl is required for the activation of Lyn kinase. (A) Mpl receptor mutations. Truncations are named for the number of residues that remain. (B) BaF3/Mpl, T69, and T111 cells were starved for 12 hours, preincubated with DMSO vehicle or 2 μM PP2, and then stimulated with TPO (15 ng/mL) for 10 minutes. Lysates normalized for total protein concentration were immunoprecipitated with anti-Lyn antibody. A kinase assay was then performed using immune complexes. Data are representative of 3 independent experiments. Immunoprecipitated cell lysates were subject to Western blot analysis using anti-Lyn antibody to ensure amounts of kinase were assayed (bottom). Data are representative of 3 independent experiments. Error bars indicate SD.
Carboxyl region of Mpl is required for the activation of Lyn kinase. (A) Mpl receptor mutations. Truncations are named for the number of residues that remain. (B) BaF3/Mpl, T69, and T111 cells were starved for 12 hours, preincubated with DMSO vehicle or 2 μM PP2, and then stimulated with TPO (15 ng/mL) for 10 minutes. Lysates normalized for total protein concentration were immunoprecipitated with anti-Lyn antibody. A kinase assay was then performed using immune complexes. Data are representative of 3 independent experiments. Immunoprecipitated cell lysates were subject to Western blot analysis using anti-Lyn antibody to ensure amounts of kinase were assayed (bottom). Data are representative of 3 independent experiments. Error bars indicate SD.
Effect of Src kinase inhibitors on TPO-induced proliferation
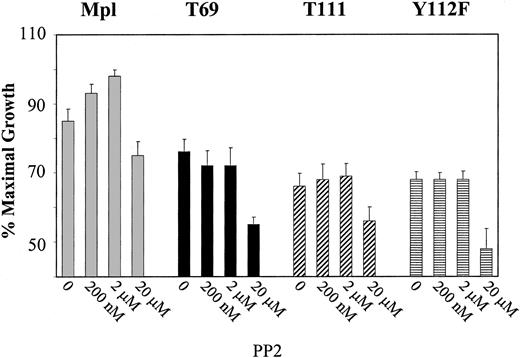
To determine the functional significance of Lyn kinase activation, PP2 was used to specifically block the activation of Lyn following TPO stimulation of BaF3/Mpl cells. Cells were pretreated in the presence or absence of PP2 and stimulated with TPO. After 48 hours, rates of cellular proliferation were measured by MTS assays. Proliferation data are expressed as the mean of triplicate values and are represented as a percentage of maximum IL-3–induced proliferation. As illustrated in Figure 3, pretreatment of BaF3/Mpl cells with 200 nM PP2 increased the proliferative rate (P < .05) and was further enhanced at 2 μM concentrations (P < .01). Because PP2 was dissolved in DMSO, an equal amount of DMSO was added to control cells (without PP2). Increasing the concentration of PP2 to cytotoxic levels (> 10 μM) resulted in a decrease in proliferation, likely due to nonspecific inhibition of other tyrosine kinases. Interestingly, under the same growth conditions the mutant Mpl receptors, T69, and T111, Y112F did not support a change in proliferation after addition of low doses of PP2 (Figure 3). To determine if PP2 changed the proliferation of BaF3/Mpl by affecting apoptosis, staining with propidium iodide and annexin V was performed on cells grown in TPO for 48 hours with various concentrations of inhibitor. Staining was quantitated by flow cytometry and showed no significant change in the apoptotic rate between TPO-stimulated BaF3/Mpl in the presence or absence of inhibitor (data not shown).
Effects of PP2 on proliferation. BaF3 cell lines expressing wild-type Mpl, T69, T111, and Y112F were grown in the presence of added TPO plus the indicated concentrations of Src kinase inhibitor (PP2). Proliferation was measured in triplicate wells by MTS assay in 96-well plates. Results of MTS proliferation assays are expressed as the mean values of 4 separate experiments. To account for differences in cell number between cell lines, the data have been normalized as a percentage of maximal proliferation induced by IL-3. Error bars indicate SD.
Effects of PP2 on proliferation. BaF3 cell lines expressing wild-type Mpl, T69, T111, and Y112F were grown in the presence of added TPO plus the indicated concentrations of Src kinase inhibitor (PP2). Proliferation was measured in triplicate wells by MTS assay in 96-well plates. Results of MTS proliferation assays are expressed as the mean values of 4 separate experiments. To account for differences in cell number between cell lines, the data have been normalized as a percentage of maximal proliferation induced by IL-3. Error bars indicate SD.
Dominant-negative Lyn increases proliferation of BaF3/Mpl cells
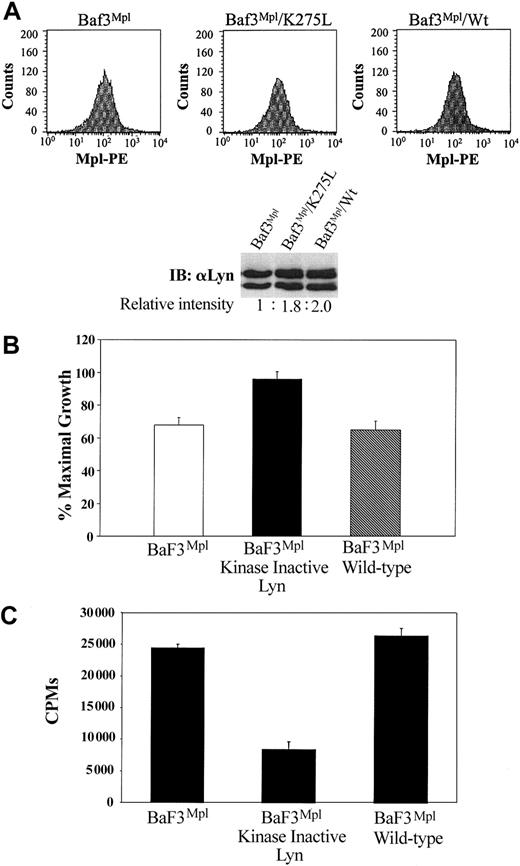
Because of the inherent concern of nonspecific inhibition using chemical inhibitors, we sought to confirm our results using a dominant-negative form of Lyn. BaF3/Mpl cells were engineered to express either the wild-type Lyn construct or dominant-negative (K275L) form of the kinase. Equivalent expression of Mpl on the surface of each cell line was confirmed by flow cytometry (Figure 4A, graphs). Immunoblotting and densitometry demonstrated a 1.8- and 2-fold increase in Lyn expression in the cells engineered to overexpress either K275L and wild-type Lyn, respectively (Figure 4A, blots). In support of our data with PP2, expression of Lyn K275L resulted in increased proliferation of BaF3/Mpl cells (∼32%), whereas expression of wild-type Lyn did not significantly change proliferation (Figure 4B). As predicted, cells expressing the kinase inactive LynK257L had decreased Lyn kinase activity, whereas expression of wild-type Lyn resulted in kinase activity similar to parental BaF3/Mpl cells (Figure 4C).
Expression of a kinase-inactive Lyn mutant increase TPO-induced proliferation. BaF3/Mpl cells were transfected with either dominant-negative Lyn or wild-type Lyn cDNA. (A) Equal cell surface expression of Mpl was determined by flow cytometry using a primary anti-Mpl antibody and a secondary phycoerythrin (PE)–conjugated antirabbit antibody. In the lower panel, a Western blot demonstrates the relative Lyn expression in each cell line. (B) Cell proliferation assays were used to compare BaF3/Mpl cell lines expressing the mutant (K275L) or wild-type forms of Lyn. Data represent the mean and SD of triplicate wells. (C) Lyn immune complex kinase assay was performed as described in Figure 1. Kinase data are representative of 3 independent experiments. Error bars indicate SD.
Expression of a kinase-inactive Lyn mutant increase TPO-induced proliferation. BaF3/Mpl cells were transfected with either dominant-negative Lyn or wild-type Lyn cDNA. (A) Equal cell surface expression of Mpl was determined by flow cytometry using a primary anti-Mpl antibody and a secondary phycoerythrin (PE)–conjugated antirabbit antibody. In the lower panel, a Western blot demonstrates the relative Lyn expression in each cell line. (B) Cell proliferation assays were used to compare BaF3/Mpl cell lines expressing the mutant (K275L) or wild-type forms of Lyn. Data represent the mean and SD of triplicate wells. (C) Lyn immune complex kinase assay was performed as described in Figure 1. Kinase data are representative of 3 independent experiments. Error bars indicate SD.
Inhibition of Lyn reduces the phosphorylation of Mpl in BaF3/Mpl cells
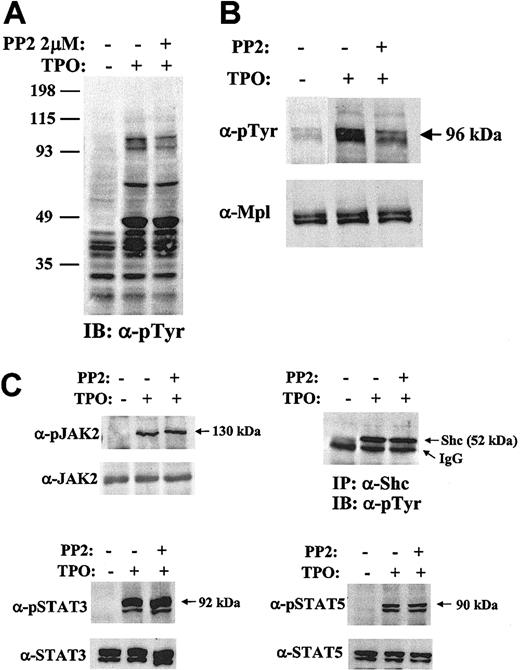
To study the role of Lyn kinase in TPO-induced signaling, intracellular phosphorylation patterns were examined in BaF3/Mpl cells in the presence or absence of the inhibitor, PP2. Whole-cell lysates were prepared and analyzed by Western blotting with an antiphosphotyrosine antibody (Figure 5A). Although the pattern of phosphotyrosine incorporation appears quite similar with or without added PP2 (2 μM), a doublet with diminished intensity migrates at about 95 kDa, the approximate molecular weight of Mpl, STAT3, and STAT5. To determine which signaling pathways were affected by SFK inhibition, we studied the TPO-induced tyrosine phosphorylation of Mpl, JAK2, Shc, STAT3, and STAT5 with and without added PP2 (Figure 5B-C). Western blots demonstrated that there was no evident change in JAK2, Shc, STAT3, and STAT5 tyrosine phosphorylation (Figure 5C) or in Akt serine phosphorylation (data not shown). In contrast, repeated experiments showed that the Mpl receptor was phosphorylated less efficiently in the presence of PP2 (Figure 5B). Stripping and reprobing these blots demonstrated equal protein loading in each lane.
Inhibition of Lyn alters TPO-induced signaling. BaF3/Mpl cells were starved, pretreated with 2 μm PP2 or DMSO, and were unstimulated (–) or stimulated (+) with TPO 15 ng/mL. Total cell lysates, normalized for protein concentration, were separated by SDS-10% polyacrylamide gel electrophoresis (PAGE) and immunoblotted with specific antibodies. (A) Whole-cell lysates were immunoblotted with a phosphotyrosine antibody (4G10). The numbers on the left indicate the sizes of the molecular mass makers in kilodaltons. (B) Mpl was immunoprecipitated with Mpl antiserum, analyzed by Western blot, and probed with antiphosphotyrosine antibody. (C) JAK2, Shc, STAT3, and STAT5 were immunoprecipitated, subjected to Western blotting, and probed with the indicated phospho-specific antibodies (“Materials and methods”). Arrows to the right of the gel refer to phosphoproteins. Blots were stripped and reprobed with appropriate antibodies to ensure equal amount of protein in each lane.
Inhibition of Lyn alters TPO-induced signaling. BaF3/Mpl cells were starved, pretreated with 2 μm PP2 or DMSO, and were unstimulated (–) or stimulated (+) with TPO 15 ng/mL. Total cell lysates, normalized for protein concentration, were separated by SDS-10% polyacrylamide gel electrophoresis (PAGE) and immunoblotted with specific antibodies. (A) Whole-cell lysates were immunoblotted with a phosphotyrosine antibody (4G10). The numbers on the left indicate the sizes of the molecular mass makers in kilodaltons. (B) Mpl was immunoprecipitated with Mpl antiserum, analyzed by Western blot, and probed with antiphosphotyrosine antibody. (C) JAK2, Shc, STAT3, and STAT5 were immunoprecipitated, subjected to Western blotting, and probed with the indicated phospho-specific antibodies (“Materials and methods”). Arrows to the right of the gel refer to phosphoproteins. Blots were stripped and reprobed with appropriate antibodies to ensure equal amount of protein in each lane.
Lyn kinase is involved in the regulation of MAPK
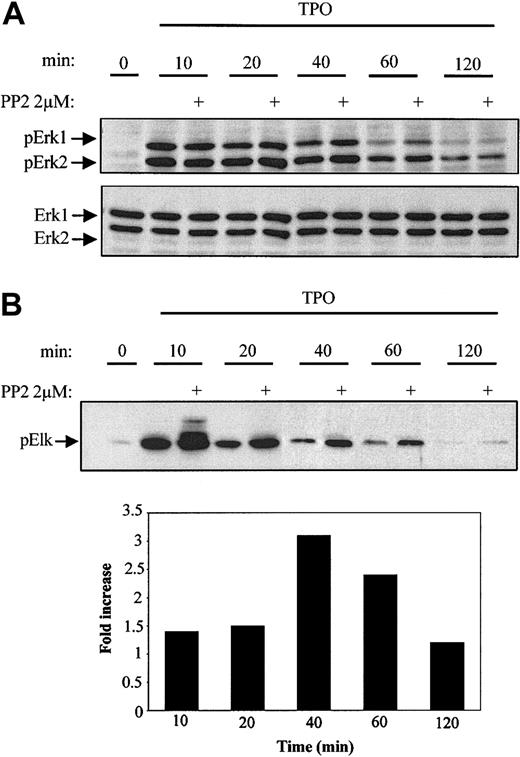
MAPK signaling plays a critical role in the regulation of both cellular proliferation and differentiation. Previous studies have shown that JAK/STAT and Erk1/2 (MAPK) pathways are both activated during TPO stimulation.5,8,9,36 Because the JAK/STAT pathway is unaffected by the inhibition of Lyn, we next investigated the potential effect of Lyn kinase on MAPK activity. BaF3/Mpl cells were stimulated in the presence and absence of PP2 for 10, 20, 40, 60, and 120 minutes, after which Western blot analysis was performed on lysates to detect the level of Erk1/2 phosphorylation. In the absence of PP2, Erk1/2 phosphorylation was greatest at 10 minutes and then progressively decreased over the 2-hour period (Figure 6A). However, cells grown in the presence of the SFK inhibitor showed greater sustained Erk1/2 phosphorylation levels over the 2-hour time course (Figure 6A). At 40 and 60 minutes Erk1/2 phosphorylation was 2-fold and 4-fold higher, respectively, in cells that were pretreated with PP2 prior to TPO stimulation. To confirm our results, we performed in vitro kinase assays by immunoprecipitating phospho-Erk1/2 and measuring its ability to phosphorylate Elk-1. Similar to phosphorylation, kinase activity peaked 10 minutes after stimulation (Figure 6B). In the presence of 2 μM PP2, Erk1/2 activation was increased at 10, 20, 40, and 60 minutes (1.4-, 1.4-, 3.1-, and 2.4-fold, respectively) until reaching basal levels at 120 minutes (Figure 6B). These results suggest that the Erk1/2 activity is diminished in both amplitude and duration by Lyn kinase.
Inhibition of Lyn activity increases Erk1/2 phosphorylation and activation in BaF3/Mpl cells. BaF3/Mpl cells were starved overnight, stimulated with 15 ng/mL TPO in the presence or absence of 2 μM PP2, and lysates prepared after the indicated times. (A) Samples normalized for protein concentration were separated by SDS-10% PAGE and analyzed by immunoblotting using an antibody specific for dually phosphorylated Erk1/2 (p42/p44). Blots were stripped and reprobed with Erk1/2-specific antibody to ensure equal loading. (B) Erk1/2 kinase activity was assayed by using Elk-1 as a substrate. Elk-1 phosphorylation at S383 was detected by Western blotting using a phospho-specific Elk-1 antibody. The increase in Elk-1 phosphorylation was quantitated by densitometry and expressed as fold change (in bar graph) and is representative of 2 separate experiments.
Inhibition of Lyn activity increases Erk1/2 phosphorylation and activation in BaF3/Mpl cells. BaF3/Mpl cells were starved overnight, stimulated with 15 ng/mL TPO in the presence or absence of 2 μM PP2, and lysates prepared after the indicated times. (A) Samples normalized for protein concentration were separated by SDS-10% PAGE and analyzed by immunoblotting using an antibody specific for dually phosphorylated Erk1/2 (p42/p44). Blots were stripped and reprobed with Erk1/2-specific antibody to ensure equal loading. (B) Erk1/2 kinase activity was assayed by using Elk-1 as a substrate. Elk-1 phosphorylation at S383 was detected by Western blotting using a phospho-specific Elk-1 antibody. The increase in Elk-1 phosphorylation was quantitated by densitometry and expressed as fold change (in bar graph) and is representative of 2 separate experiments.
Treatment of primary bone marrow cells with the Src inhibitor PP1 results in an increased number of polyploid cells
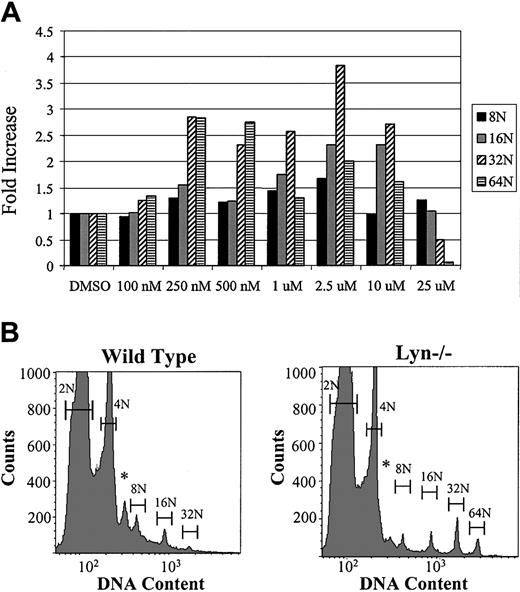
To evaluate the results of Src inhibition in primary cells, fresh bone marrow from C57BL/J mice was prepared in serum-free media containing 1% Nutridoma SP and 37.5 ng/mL recombinant mTPO in the form of conditioned medium. Cells were treated with either DMSO alone (0.1%) or PP1 dissolved in DMSO to achieve a final concentration of 0 to 25 μM (Figure 7A). After 72 hours in culture, cells were stained with anti-CD41–FITC antibody and propidium iodide and analyzed by flow cytometry to identify polyploid cells of the megakaryocytic lineage. The percentage of each ploidy class is expressed as a fold increase or fold decrease relative to no inhibitor (ie, no inhibitor = 1). In the presence of the Src inhibitor, PP1, MK number, and polyploidization were increased. We next explored the possibility that Lyn-deficient mice would have increased megakaryocytopoiesis in vivo. Whole bone marrow cells from wild-type and lyn–/– mice were cultured in serum-free media with TPO for 3 days. As seen in Figure 7B, MKs from lyn–/– mice show a marked increase in DNA content, with a greater number of cells reaching 32N and 64N. These results indicate that Src kinases inhibit endomitosis in MKs similar to the negative effect on mitosis in cell lines.
Effect of Src kinase inhibitor PP1 on primary murine MKs. Fresh bone marrow from C57BL/J mice was washed and resuspended in RPMI and 1% Nutridoma plus recombinant mTPO. DMSO alone (final concentration, 0.1%) or the Src inhibitor PP1, dissolved in DMSO, was added to the cells at the indicated final concentrations. After 72 hours of incubation, the entire cell culture was analyzed by flow cytometry for the presence of polyploid cells. The percentage of each ploidy class is expressed as a fold increase or decrease relative to no inhibitor (ie, no inhibitor = 1). Data are representative of 3 separate experiments. (B) Wild-type mice (C57BL/J) and lyn–/– mice were killed and femurs from 4 mice (of each type) were pooled, subjected to RBC lysis, and cultured under serum-free conditions with exogenous recombinant mTPO (37.5 ng/mL) for 72 hours. Nuclear ploidy was determined by flow cytometry and plotted on a semilog scale (500 000 events/sample). Bars mark the DNA contents of 2N, 4N, 8N, 16N, and 64N, respectively. *Anueploid cells.
Effect of Src kinase inhibitor PP1 on primary murine MKs. Fresh bone marrow from C57BL/J mice was washed and resuspended in RPMI and 1% Nutridoma plus recombinant mTPO. DMSO alone (final concentration, 0.1%) or the Src inhibitor PP1, dissolved in DMSO, was added to the cells at the indicated final concentrations. After 72 hours of incubation, the entire cell culture was analyzed by flow cytometry for the presence of polyploid cells. The percentage of each ploidy class is expressed as a fold increase or decrease relative to no inhibitor (ie, no inhibitor = 1). Data are representative of 3 separate experiments. (B) Wild-type mice (C57BL/J) and lyn–/– mice were killed and femurs from 4 mice (of each type) were pooled, subjected to RBC lysis, and cultured under serum-free conditions with exogenous recombinant mTPO (37.5 ng/mL) for 72 hours. Nuclear ploidy was determined by flow cytometry and plotted on a semilog scale (500 000 events/sample). Bars mark the DNA contents of 2N, 4N, 8N, 16N, and 64N, respectively. *Anueploid cells.
Discussion
Previous studies have demonstrated important functional roles played by Src family members in the signaling events of a number of hematopoietic cytokine receptors (IL-2, IL-3, IL-5, CSF-1, and EPO-R).19,21,22,30 In this report, we provide evidence for a role played by SFKs in TPO-mediated cell proliferation. Our studies demonstrate that TPO activates Lyn and that Y112 of the Mpl cytoplasmic domain is necessary for activation of the kinase. Inhibition of Lyn results in enhanced TPO-induced proliferation in both hematopoietic cell lines and normal bone marrow cells. This physiologic effect may be due to greater Erk1/2 phosphorylation and activity.
The up-regulation of tyrosine phosphorylation in response to TPO requires the association and activation of nonreceptor tyrosine kinases. It has been well established that TPO stimulation results in activation of JAK2, immediately followed by the tyrosine phosphorylation of numerous signaling molecules, including Mpl, STAT3, STAT5, Cbl, Shc, Vav, Raf-1, Ras, MAPK, SHIP, and phosphoinositol 3-kinase (PI3K).9,36-38 To investigate the possible role of SFKs in Mpl signaling, we used a cytokine-dependent murine cell line, BaF3, which was engineered to express Mpl on the cell surface. The results presented in this study demonstrate that the addition of TPO to BaF3/Mpl cells results in the activation of Lyn kinase. The observed TPO activation of Lyn can be abolished when BaF3/Mpl cells are treated with the SFK inhibitor PP2. Although these chemicals are quite specific for SFKs at concentrations less than 10 μM, caution must be used in interpreting these results because of possible effects on other kinases. However, we found that at the maximally effective inhibitor concentration (2 μM PP2) there was no change in the level of JAK2 phosphorylation (Figure 5). Moreover, the transfection of BaF3/Mpl cells with kinase-inactive Lyn yielded a similar increase in proliferation rate and a corresponding decrease in kinase activity, thus confirming our results with chemical inhibitors. Recently, an inhibitory role for Lyn has been described in macrophages, suggesting that this may be a more general observation.39
In cell lines and in mice expressing mutant receptor constructs, our laboratory and others have shown that the 121–amino acid signaling domain can be divided into functional domains. The membrane-proximal portion of the cytoplasmic domain of Mpl (up to 69 residues) is necessary and sufficient for proliferation.34 This region contains 2 conserved motifs, box1 and box2, that are required for the activation of the JAK2 kinase. The membrane-distal portion (carboxyl terminus) of Mpl contains the primary site of receptor tyrosine phosphorylation (Y112). When phosphorylated, Y112 serves as a docking site for the Shc, STAT molecules, and other signaling proteins including Grb2, Cbl, Vav, and SHIP (Src homology 2 domain–containing inositol polyphosphate phosphatase-1). Our results indicate that activation of Lyn is also dependent on this residue. This raises the possibility that phosphorylated Y112 or an associated phosphoprotein binds to the SH2 domain of Lyn, displacing the regulatory carboxyl tyrosine and permitting kinase activation. Interestingly, Mpl phosphorylation is diminished in BaF3/Mpl cells pretreated with PP2 prior to TPO stimulation. Mpl contains 5 tyrosine residues (Y8, Y29, Y78, Y112, and Y117), which may be potential sites of phosphorylation. It is tempting to hypothesize that Y78 is the potential site of perturbed phosphorylation because previous studies have suggested the region between residues 69 and 83 of the cytoplasmic domain contains a potential inhibitory domain. Porteu et al have shown that this region is responsible for inhibiting proliferation of UT7/Mpl cells and is necessary for TPO-dependent megakaryocytic differentiation.40 Further investigation, using BaF3 cells expressing an Mpl receptor mutant containing a point mutation of Y78 resulted in an increased proliferation rate.34 However, our studies demonstrate that Y78 acts as an inhibitor of proliferation even in the context of a truncated receptor that does not activate Lyn. Studies are ongoing to determine if Lyn associates directly with Mpl and if Mpl is a substrate for activated SFKs.
Erk1/2, also known as MAPK, are activated by a number of cytokines and play critical roles in the regulation of cellular proliferation and differentiation.41 Erk1/2 signaling cascade is a tightly controlled pathway, and the magnitude and duration of kinase activity determines the physiologic response. Sustained Erk signaling has been shown to play a role in MK maturation.8,10 Studies in the human megakaryoblastic leukemia cell line (CMK) have shown that the introduction of a constitutively active form of Erk induced expression of megakaryocytic-specific surface makers.42 In UT7/Mpl cells, TPO-induced megakaryocytic differentiation is dependent on the sustained activation of Erk1/2.10 In addition, murine whole bone marrow cells cultured in the presence of TPO and the MEK inhibitor PD 98059 blocked the Erk signaling pathway and resulted in a reduction in MK polyploidization.8 Pretreatment of BaF3/Mpl cells with PP2 resulted in higher and more persistent Erk1/2 activity, which correlated with an increase in proliferation. Although Erk1/2 activity is not sustained for more than 2 hours, it is noteworthy that the greatest fold increase in phosphorylation and kinase activity is achieved after longer exposures to TPO with inhibitor (40 and 60 minutes, respectively). Such results suggest that active Lyn kinase may partially block Erk1/2 activation or accelerate Erk1/2 inactivation. These possibilities are currently under investigation in our laboratory.
On the basis of our results in cell lines, we expanded our studies of Src tyrosine kinases signaling to primary murine bone marrow cultures, which provide a physiologically relevant model for studying hematopoiesis. As MK progenitors mature and differentiate they undergo endomitosis (DNA replication without cytokinesis), resulting in polyploid cells with 8N, 16N, 32N, and 64N complement of chromosomes. As shown in Figure 7, murine marrow cells cultured for 3 days in the presence of TPO and PP1 resulted in a greater percentage of polyploid cells, suggesting that Src kinases normally limit MK proliferation and maturation. In 3 separate experiments, the marrow cells cultured in the presence of PP1 (< 10 μM) consistently yielded a higher number of polyploid cells. Two possible explanations could account for this observation: (1) inhibiting SFKs could accelerate cell cycle progression during mitosis and endomitosis, or (2) blocking SFKs might delay entry of MKs into endomitosis, allowing extra cell divisions prior to terminal maturation. Experiments to distinguish between these 2 hypotheses are currently ongoing.
Studies have shown that Lyn can act as a positive or negative regulator of cellular processes, depending on both cell type and stimulus.43 Published reports have shown that Lyn kinase can augment proliferation (eg, stem cell factor and prolactin signaling) or differentiation (eg, EPO and G-CSF signaling).19,30,35,44 Similarly, Erk1/2 activation can regulate distinct aspects of cell cycle progression including G1/S and G2/M transitions.45-47 Therefore, it is likely that physiologic responses to active Lyn and Erk1/2 are dependent on the precise cellular context and the composition of the activated signaling complexes. It is not yet clear whether these signaling pathways function differently in MKs compared to other hematopoietic cells.
In conclusion, we found that Lyn kinase is activated by TPO in cell lines expressing the Mpl receptor. Inhibition of Src kinase activity increases the proliferative rate of BaF3/Mpl cells and normal murine MK progenitors, demonstrating that Lyn, and perhaps other SFKs, negatively regulate TPO-induced growth. Identifying the downstream targets of Lyn and understanding the molecular pathway that allows Lyn to regulate Erk1/2 activity are areas for future investigation.
Prepublished online as Blood First Edition Paper, January 15, 2004; DOI 10.1182/blood-2003-10-3566.
Supported by the National Institutes of Health grants R01HL65498 and K01DK065129-01.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We wish to thank those individuals who have contributed to this manuscript: Diana Linnekin (National Institutes of Health) for wild-type Lyn and kinase-null Lyn cDNA constructs; Janet Oliver (University of New Mexico) for lyn–/– mice; Paul Stein (Northwestern University) for advice and discussions; Amgen and Zymogenetics for originally providing Mpl cDNA and anti-Mpl antiserum; Noel Blake for assistance with flow cytometry; and Linda Hibbeln and Jennifer Minear for manuscript preparation.

![Figure 1. TPO-induced activation of Src kinases. Parental BaF3 cells and those engineered to express c-Mpl were starved overnight. Half of the cells were stimulated with TPO (15 ng/mL) for 10 minutes before making whole-cell lysates. (A) Kinase assays were performed using γ-32P] ATP and peptides that function as efficient ([Lys19] cdc2(6-20)-NH2) and inefficient ([Lys19Ser14Val12] cdc2(6-20)-NH2) substrates for Src kinases. (B) Immune complex kinase assay. Lysates normalized for total protein concentration were immunoprecipitated with either anti-Lyn or anti-Fyn antibodies. Kinase activity was measured by phosphorylation of substrate ([Lys19] cdc2(6-20)-NH2) in immune complexes. (C) Cells were pretreated for 45 minutes with DMSO (vehicle = 0) or the stated concentration of PP2 and stimulated with TPO. Lysates were immunoprecipitated with anti-Lyn, after which immune complex kinase assays were performed. The data have been normalized as a percentage of maximal kinase activity induced by TPO in the presence of DMSO alone. Immunoblots of immunoprecipitated lysates (A-B) were performed to ensure equal amounts of Lyn and Fyn were assayed (data not shown). All assay data are representative of 3 independent experiments. Error bars indicate SD.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/103/10/10.1182_blood-2003-10-3566/6/m_zh80100461480001.jpeg?Expires=1769105583&Signature=lrVvZM34vU9Ok4Wfdy9I0NEC2eMsLp1aOkIAHdiEFQHIReqZIHZDsHebmEO-KwOVjrl33LBGylwVrHHGh11y-azsM3xz4H~enQ8bvHr-kZAe7jKdjdSR1IFVwun0vfVtDs-ufzoNS2G9JuTY9sgOF9QecI7GTRg-QW0o9Y7rtt1b~Gkm3U1YMmiOcriLkV7DRDb2o8xZxHAp6mwyOl-B~6bgqVUkKjrcEl9NVEzyX9vrGbAdqlzx7bguQwbPKae3JnbX~Eayve1T5GrRP8QwaQwWD4rreS3WWLn45MNK-f9h4feu~NkK-fcHItaJrT0ypGRD7k65xzYkUBKIbhHdNw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal