Abstract
Interactions between the Chk1 inhibitor UCN-01 and the farnesyltransferase inhibitor L744832 were examined in human leukemia cells. Combined exposure of U937 cells to subtoxic concentrations of UCN-01 and L744832 resulted in a dramatic increase in mitochondrial dysfunction, apoptosis, and loss of clonogenicity. Similar interactions were noted in other leukemia cells (HL-60, Raji, Jurkat) and primary acute myeloid leukemia (AML) blasts. Coadministration of L744832 blocked UCN-01-mediated phosphorylation of mitogen-activated protein kinase kinase/extracellular signal-regulated kinase (MEK/ERK), leading to down-regulation of phospho-cyclic adenosine monophosphate responsive element-binding protein (phospho-CREB) and -p90RSK and activation of p34cdc2 and stress-activated protein kinase/ERK kinase/c-Jun N-terminal kinase (SEK/JNK). Combined treatment also resulted in pronounced reductions in levels of phospho-Akt, -glycogen synthase kinase-3 (-GSK-3), -p70S6K, -mammalian target of rapamycin (-mTOR), -forkhead transcription factor (-FKHR), -caspase-9, and -Bad. Ectopic expression of Bcl-2 or Bcl-xL but not dominant-negative caspase-8 blocked UCN-01/L744832-mediated mitochondrial dysfunction and apoptosis but did not prevent activation of p34cdc2 and JNK or inactivation of MEK/ERK and Akt. Enforced expression of myristoylated Akt but not constitutively active MEK significantly attenuated UCN-01/L744832-induced apoptosis. However, dual transfection with Akt and MEK resulted in further protection from UCN-01/L744832-mediated lethality. Finally, down-regulation of JNK1 by siRNA significantly reduced the lethality of the UCN-01/L744832 regimen. Together, these findings suggest that farnesyltransferase inhibitors interrupt the cytoprotective Akt and MAPK pathways while reciprocally activating SAPK/JNK in leukemia cells exposed to UCN-01 and, in so doing, dramatically increase mitochondria-dependent apoptosis. (Blood. 2005;105:1706-1716)
Introduction
Ras proteins represent members of a guanosine triphosphate (GTP)-binding protein superfamily involved in proliferation and survival among other functions. Activating point mutations involving 3 major members of this family, H-Ras, K-Ras, or N-Ras, have been found in 30% of human tumors.1 Such mutations result in constitutive Ras activation, thereby conferring cells with a growth factor-independent survival advantage. Regulation of Ras function, in addition to the extent of GTP binding, involves posttranslational modification, most notably farnesylation or geranylgeranylation, which promote translocation of Ras from the cytosol to plasma membranes and activation of downstream targets,1 particularly the serine/threonine kinase Raf family, including Raf-1, B-Raf, and A-Raf.2 Upon translocation to the plasma membrane following Ras activation, Raf signals downstream to MEK1/2 (mitogen-activated protein [MAP] kinase kinase), which in turn activates the MAP kinase ERK1/2 (extracellular signal-regulated kinase 1/2).3 The Raf/MEK/ERK pathway activates a number of transcription factors, including members of the Ets family (ie, Elk1), cyclic adenosine monophosphate responsive element-binding protein (CREB), and c-Jun/AP-1.4-6 Activation of the Raf/MEK/ERK cascade generally promotes cell survival, particularly in the case of malignant hematopoietic cells.7 The phosphoinositide 3-kinase (PI3K)/Akt signaling pathway represents another important downstream Ras cascade. Ras interacts directly with the catalytic subunit of PI3K in a GTP-dependent manner through the Ras effector site, leading to synergistic activation of this lipid kinase with tyrosine kinases.8 PI3K/Akt also exerts diverse cytoprotective functions.7
The finding that Ras mutations associated with constitutive activation occur frequently in human cancers1 has prompted the development of farnesyltransferase inhibitors (FTIs), which interfere with Ras farnesylation and membrane translocation necessary for Ras function.9 FTIs have shown impressive activity in preclinical studies10 and are undergoing clinical evaluation in humans.11 However, FTIs have not yet fulfilled their clinical potential, possibly a consequence of alternative mechanisms of posttranslational Ras modifications, including geranylgeranylation, especially in the case of K-Ras and N-Ras.12 Nevertheless, initial evidence suggests that FTIs may play a useful therapeutic role in the treatment of certain hematopoietic malignancies (eg, leukemia and multiple myeloma).11,13,14
UCN-01 (7-hydroxystaurosporine) was originally developed as a protein kinase C (PKC) inhibitor but was subsequently shown to inhibit several other cell survival/cell cycle regulatory proteins, including Chk1 15 and 3-phosphoinositide-dependent protein kinase-1 (PDK1)/Akt.16 By inhibiting Chk1, UCN-01 blocks the proteasomal degradation of the cdc25C phosphatase, resulting in dephosphorylation (activation) of p34cdc2.15,17 In this way, UCN-01 functions as a checkpoint abrogator that potentiates the lethal effects of several DNA-damaging agents, including ara-C18 and camptothecin.19 UCN-01 is also a potent inducer of cell death in human leukemia cells, triggering apoptosis at submicromolar concentrations.20 Phase 1 trials of UCN-01 have been completed,21 and preliminary evidence of activity when combined with established cytotoxic agents in patients with hematologic malignancies has been reported.22
Previously, we reported that in malignant human hematopoietic cells, activation of p34cdc2 by UCN-01 is associated with a marked induction of the MEK/ERK pathway and that interference with this process (ie, by pharmacologic MEK1/2 inhibitors) resulted in a striking increase in mitochondrial dysfunction and apoptosis.23,24 Because MEK and ERK represent important downstream targets of Ras, the possibility therefore existed that FTIs might potentiate UCN-01 lethality through a similar mechanism. To test this hypothesis, we have examined interactions between UCN-01 and the FTI L744832 25 in a variety of human leukemia cell lines in relation to induction of cell death and effects on various signaling pathways. Here we report that coexposure of human leukemia cells to the FTI blocks UCN-01-induced activation of ERK, cooperates with UCN-01 to inactivate the Akt and activate the stress-activated protein kinase/c-Jun N-terminal kinase (SAPK/JNK) pathways and, in so doing, results in a highly synergistic increase in mitochondrial dysfunction, caspase activation, and apoptosis. Such findings suggest that combined treatment with UCN-01 and clinically relevant FTIs may warrant further examination as a novel antileukemic strategy.
Materials and methods
Cells and reagents
U937, HL-60, Jurkat, and Raji cells were provided by American Type Culture Collection (ATCC; Manassas, VA) and maintained in RPMI 1640 medium containing 10% fetal bovine serum (FBS).23 U937 stably transfected with dominant-negative (DN) caspase-8/pcDNA3.1, Bcl-xL/pcDNA3.1, and Bcl-2/pCEP4 were obtained as previously reported.18,26 Jurkat cells with Tet-on inducible myristoylated Akt (active) or constitutively active MEK1 (S218D/S222D) were obtained as described previously.27 All experiments utilized logarithmically growing cells (3 × 105 to 5 × 105/mL).
Peripheral blood samples were obtained with informed consent from 3 patients with acute myeloid leukemia (AML) undergoing routine diagnostic aspirations with approval from the institutional review board of Virginia Commonwealth University. Informed consent was provided according to the Declaration of Helsinki. AML blasts were isolated by centrifugation at 400g for 38 minutes over Histopaque-1077 (Sigma Diagnostics, St Louis, MO). Viability of the cells was regularly more than 95% by trypan blue exclusion. Blasts were incubated in RPMI 1640 medium containing 10% FBS in 96-well plates under the same conditions described for cell lines. Normal mononuclear cells were also obtained with informed consent from the bone marrow of patients with nonmalignant hematopoietic disorders in which the white blood cell series was uninvolved (eg, iron deficiency anemia, immune thrombocytopenia) as well as from peripheral blood donated by healthy volunteers. Mononuclear cell preparations were obtained as described for the isolation of AML blasts. Primary rodent hepatocytes were isolated from adult male Sprague-Dawley rats by a 2-step collagenase perfusion technique as previously described in detail.28
The Chk1 inhibitor UCN-01 (7-hydroxystauronsporine) was provided by Dr Edward Sausville (Developmental Therapeutics Program, National Cancer Institute [NCI]). L744832, a potent and selective thiol-containing peptidomimetic FTI,25 and FTI-277 29 were purchased from Calbiochem (San Diego, CA). They were dissolved in dimethyl sulfoxide (DMSO) as a stock solution, stored at -80°C, and diluted with serum-free RPMI medium prior to use. The pancaspase inhibitor BOC-D-fmk was provided by Enzyme Systems Products (Livermore, CA). In all experiments, the final concentration of DMSO did not exceed 0.1%.
Analysis of apoptosis and mitochondrial membrane potential (Δψm)
The extent of apoptosis was evaluated by assessing Wright-Giemsa-stained cytospin slides under light microscopy and by flow cytometric analysis with annexin V-fluorescein isothiocyanate (FITC) staining as described previously.30 Apoptosis was also evaluated by transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) staining and visualized using an Olympus AX70 fluorescence microscope (Olympus America, Melville, NY) with a CE camera and on RSImage software (Alpha Innotech, San Leandro, CA).23 To analyze loss of Δψm, cells were stained with 3,3′-dihexyloxacarbocyanine (DiOC6; Molecular Probes, Eugene, OR) and analyzed by flow cytometry to determine the percentage of cells exhibiting low levels of DiOC6 uptake.23,24 For analysis of apoptosis in primary rodent hepatocytes, cytospin slides were prepared and stained with Hoechst 33342.28 The percentage of apoptotic cells was determined by counting 500 cells from multiple randomly selected fields under fluorescence microscopy.28
Assays of cell viability and clonogenicity
In experiments involving transient transfection, the ViaCount assay was performed to evaluate cell viability. Briefly, 1 × 105 cells were stained with Guava ViaCount Reagent (Guava Technologies, Hayward, CA) for 5 minutes and the percentage of viable cells determined using a Guava Personal Cytometer (Gauva Technologies) as per the manufacturer's instructions. The ViaCount assay distinguishes between viable and nonviable cells based on the differential permeability of DNA-binding dyes and correlated closely with annexin V/propidium iodide (PI) staining. To evaluate colony-forming ability following drug treatment, a soft agar cloning assay was performed as described preciously.23 Colonies consisting of more than 50 cells were scored using an Olympus CK inverted microscope (Olympus America) and colony formation for each condition calculated in relation to values obtained for untreated control cells.
Western blot analysis
Samples from whole-cell pellets were prepared, and 30 μg protein for each condition was subjected to Western blot analysis following procedures previously described in detail.23 Alternatively, S-100 cytosolic samples for Western blot analysis were prepared using digitonin lysis buffer (75 mM NaCl, 8 mM Na2HPO4, 1 mM NaH2PO4, 1 mM EDTA [ethylenediaminetetraacetic acid], and 350 μg/mL digitonin).23 For blotting phosphoproteins, 1 mM each of sodium vanadate and sodium pyrophosphate was added to 1 × sample buffer, no SDS was included in the transfer buffer, and Tris-buffered saline (TBS) was used instead of phosphate-buffered saline (PBS) throughout. Where indicated, blots were reprobed with antibodies against actin (Transduction Laboratories, Lexington, KY) or tubulin (Calbiochem) to ensure equal loading and transfer of proteins. The following primary antibodies were used: caspase-2 and caspase-8 from Alexis (San Diego, CA); X-linked inhibitor of apoptosis protein (XIAP) and caspase-3 from Transduction Laboratories; phospho-JNK (Thr183/Tyr185), JNK1, JNK2, phospho-Akt (Ser473), Akt, phospho-Bad (Ser136), phospho-caspase-9 (Ser196), cytochrome c, apoptosis-inducing factor (AIF), Bcl-xS/L, Bax, Bak, and Mcl-1 from Santa Cruz Biotechnology (Santa Cruz, CA); phospho-MEK1/2 (Ser217/221), MEK1/2, phospho-p44/42 MAP kinase (ERK, Thr202/Tyr204), p44/42 MAP kinase, phospho-SAPK/ERK kinase-1/MKK4 (phospho-SEK1/MKK4) (Thr261), SAPK/JNK, phospho-p90RSK (Ser380), phospho-glycogen synthase kinase-3α/β (phospho-GSK-3α/β) (Ser21/9), phospho-forkhead transcription factor (phospho-FKHR) (Thr24)/FKHRL1 (Thr32), phospho-p70 S6 kinase (Thr389), phospho-mammalian target of rapamycin (phospho-mTOR) (Ser2448), phospho-4E-BP1 (Ser65), phospho-cdc2 (Tyr15), cdc2, cleaved caspase-3 (17 kDa), and cleaved poly(adenosine diphosphate-ribose) polymerase (PARP) (89 kDa) from Cell Signaling (Beverly, MA); second mitochondrial activator of caspase/direct inhibitor of apoptosis protein-binding protein with low isoelectric point (Smac/DIABLO) and phospho-CREB (Ser133) from Upstate Biotechnology (Lake Placid, NY); Bid and survivin from R&D Systems (Minneapolis, MN); anti-caspase-9 (PharMingen); anti-PARP (Biomol, Plymouth Meeting, PA); and anti-human Bcl-2 oncoprotein (Dako, Carpinteria, CA).
Transfection with cDNAs and siRNA
For dual transfection, U937 cells were stably transfected with myristoylated Akt/pUSEamp (Upstate Biotechnology), after which clones were selected with G418 and tested for expression of Myc-Tag and Akt. Akt/U937 cells were then transiently transfected with constitutively active MEK1 (S218D/S222D, activating mutations of serine 218 and 222 to aspartic acid) in pEGFP-C2 vector (Clontech, Palo Alto, CA) or its empty vector using the Amaxa Nucleofector Device (program V-01) with Cell Line Specific Nucleofector Kit V (Amaxa, Cologne, Germany) as per manufacturer's instructions. For siRNA transfection, U937 cells were transfected with 1 μg JNK1 (MAPK8) annealed dsRNAi oligonucleotide 5′-CGUGGAUUUAUGGUCUGUGTT-3′/3′-TTGCACCUAAAUACCAGACAC-5′ (Orbigen, San Diego, CA) by using Amaxa Nucleofector as described for transfection with cDNA. The transfected cells were recovered for 24 hours at 37°C, after which cells were treated with drugs for the indicated intervals (generally 8 hours) and subjected to analysis of cell viability (ViaCount, Guava) and annexin V-PE staining/flow cytometry.
Statistical analysis
For analysis of apoptosis (morphology and annexin V), Δψm, cell viability, and colony-forming ability, values represent the means ± SD for at least 3 separate experiments performed in triplicate. The significance of differences between experimental variables was determined using the Student t test. Analysis of synergism was performed according to median dose effect analysis30 using a commercially available software program, Calcusyn (Biosoft, Ferguson, MO).
Results
Combined exposure to subtoxic concentrations of UCN-01 and L744832 leads to a dramatic increase in apoptosis in multiple human leukemia cell types and primary AML blasts
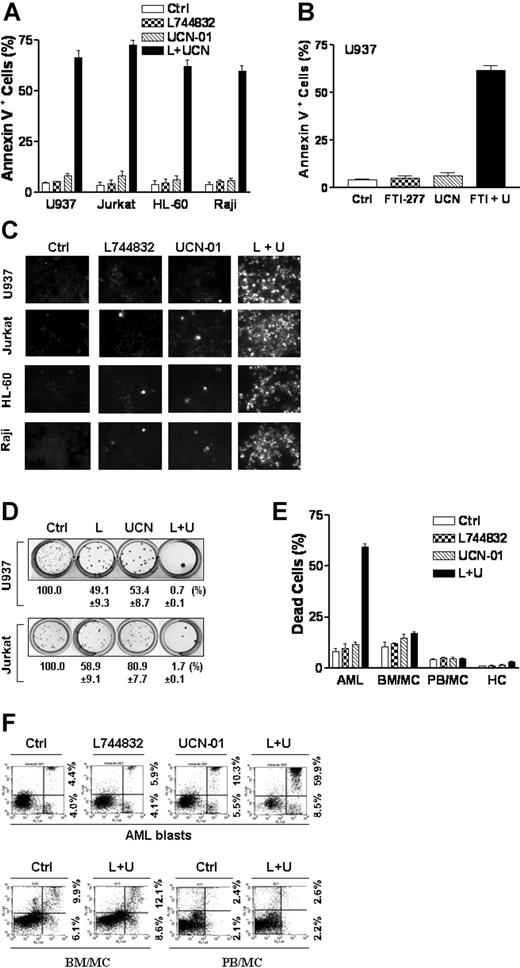
Various leukemia cell types, including U937 myelomonocytic, HL-60 promyelocytic, Jurkat T-lymphoblastic, and Raji B-lymphoblastic leukemic cells, were exposed to subtoxic concentrations of UCN-01 and L744832 alone and in combination for 18 hours, after which apoptosis was assessed by annexin V/PI analysis. In each cell line, treatment with the drugs alone minimally induced apoptosis, whereas combined exposure resulted in a dramatic increase in cell death (Figure 1A). Similar results were obtained when another FTI (FTI-277) was used (Figure 1B). TUNEL assays (Figure 1C) confirmed the striking potentiation of apoptosis following combined treatment. Furthermore, combined treatment resulted in the virtual abrogation of colony formation in each of U937 and Jurkat cell lines. (Figure 1D). Comparable reductions (ie, to less than 3% of control values) were also observed in HL-60 and Raji cells (data not shown).
Coadministration of UCN-01 and farnesyltransferase inhibitors dramatically induces apoptosis and loss of clonogenicity in human leukemia cells. (A) U937, Jurkat, HL-60, and Raji cells were exposed for 18 hours to 10 μM L744832 (L) with or without UCN-01 (UCN or U, U937, and HL-60, 100 nM; Jurkat and Raji, 150 nM), respectively, after which the percentage of cells exhibiting annexin V positivity was determined by annexin V-FITC/PI staining and flow cytometry as described in “Materials and methods.” (B) U937 cells were exposed to 20 μM FTI-277 with or without 100 nM UCN-01 for 18 hours, after which the percentage of apoptotic cells was determined as described above. (C) U937, Jurkat, HL-60, and Raji cells were treated as in panel A, after which TUNEL staining was performed to document apoptosis. Original microscope magnification, × 300. The results of a representative experiment are shown; an additional study yielded equivalent results. (D) U937 and Jurkat cells were treated as in panel A, after which cells were washed free of drug and plated in soft agar as described in the text. After 12 days of incubation, colonies (more than 50 cells) were scored and colony formation for each condition expressed relative to untreated control cells. (E-F) Blasts from 3 patients with acute myeloid leukemia (AML; FAB classification M2) were isolated as described in “Materials and methods.” Mononuclear cells were isolated from the bone marrow (BM/MC) and peripheral blood (PB/MC) of patients with nonmalignant, nonmyeloid hematopoietic disorders or healthy volunteers, respectively, as described in “Materials and methods.” Primary rodent hepatocytes (HC) were isolated as described in “Materials and methods.” Cells were treated with 10 μM L744832 with or without 150 nM UCN-01 for 18 hours (AML, BM/MC, and PB/MC) or 10 μM L744832 with or without 100 nM UCN-01 for 24 hours (HC), respectively, after which the percentage of dead cells (E) was determined by evaluating Wright-Giemsa-stained (AML, BM/MC, and PB/MC) or Hoechst 33342-stained (HC) cytospin preparations. Alternatively, the percentage of annexin V-positive cells in AML blast samples (F, upper panels) and BM/MC and PB/MC (F, lower panels) was assessed by flow cytometry. The results of representative experiments are shown; additional studies yielded equivalent results. For panels A, B, D, and E, results represent the means ± SD for 3 separate experiments performed in triplicate.
Coadministration of UCN-01 and farnesyltransferase inhibitors dramatically induces apoptosis and loss of clonogenicity in human leukemia cells. (A) U937, Jurkat, HL-60, and Raji cells were exposed for 18 hours to 10 μM L744832 (L) with or without UCN-01 (UCN or U, U937, and HL-60, 100 nM; Jurkat and Raji, 150 nM), respectively, after which the percentage of cells exhibiting annexin V positivity was determined by annexin V-FITC/PI staining and flow cytometry as described in “Materials and methods.” (B) U937 cells were exposed to 20 μM FTI-277 with or without 100 nM UCN-01 for 18 hours, after which the percentage of apoptotic cells was determined as described above. (C) U937, Jurkat, HL-60, and Raji cells were treated as in panel A, after which TUNEL staining was performed to document apoptosis. Original microscope magnification, × 300. The results of a representative experiment are shown; an additional study yielded equivalent results. (D) U937 and Jurkat cells were treated as in panel A, after which cells were washed free of drug and plated in soft agar as described in the text. After 12 days of incubation, colonies (more than 50 cells) were scored and colony formation for each condition expressed relative to untreated control cells. (E-F) Blasts from 3 patients with acute myeloid leukemia (AML; FAB classification M2) were isolated as described in “Materials and methods.” Mononuclear cells were isolated from the bone marrow (BM/MC) and peripheral blood (PB/MC) of patients with nonmalignant, nonmyeloid hematopoietic disorders or healthy volunteers, respectively, as described in “Materials and methods.” Primary rodent hepatocytes (HC) were isolated as described in “Materials and methods.” Cells were treated with 10 μM L744832 with or without 150 nM UCN-01 for 18 hours (AML, BM/MC, and PB/MC) or 10 μM L744832 with or without 100 nM UCN-01 for 24 hours (HC), respectively, after which the percentage of dead cells (E) was determined by evaluating Wright-Giemsa-stained (AML, BM/MC, and PB/MC) or Hoechst 33342-stained (HC) cytospin preparations. Alternatively, the percentage of annexin V-positive cells in AML blast samples (F, upper panels) and BM/MC and PB/MC (F, lower panels) was assessed by flow cytometry. The results of representative experiments are shown; additional studies yielded equivalent results. For panels A, B, D, and E, results represent the means ± SD for 3 separate experiments performed in triplicate.
Furthermore, exposure of blasts from 3 patients with AML (French-American-British [FAB] classification M2) to UCN-01 ± L744832 documented the marked increase in cell death with combined drug exposure (Figure 1E), a finding graphically illustrated by flow cytometry for patient no. 3 (Figure 1F). In contrast, the UCN-01/L744832 regimen exerted only minimal toxicity toward normal human bone marrow (BM/MC) and peripheral blood mononuclear cells (PB/MC) as well as normal rodent hepatocytes (HC) (Figure 1E-F). Together, these findings indicate that combining FTIs and UCN-01 synergistically triggers apoptosis in human myeloid and lymphoid leukemia cells lines as well as in primary AML blasts but displays significantly less toxicity toward normal hematopoietic and nonhematopoietic cells.
L744832 rapidly potentiates UCN-01-mediated lethality in a dose-dependent manner
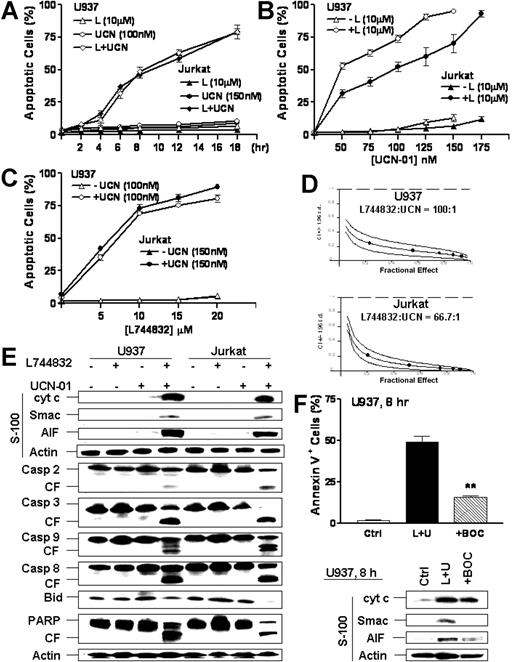
In U937 and Jurkat cells (Figure 2A), a discernible increase in apoptosis was noted as early as 6 hours and 4 hours, respectively, following combined exposure to nontoxic concentrations of L744832 and UCN-01. Apoptosis approached 80% at 18 hours, and lethality was nearly complete by 24 hours (data not shown). Coadministration of UCN-01 concentrations as low as 50 nM with a nontoxic concentration of L744832 (10 μM) resulted in a pronounced increase in cell death in both cell lines, which approached 100% for UCN-01 concentrations of 150 to 175 nM (Figure 2B). L744832 concentrations of 5 μM clearly increased the lethality of marginally toxic concentrations of UCN-01, and effects were near maximal at concentrations of 10 μM (Figure 2C).
L744832 rapidly and highly synergistically potentiates UCN-01-mediated lethality in human leukemia cells, accompanied by mitochondrial dysfunction, caspase activation, and PARP cleavage. (A) U937 and Jurkat cells were exposed to 10 μM L744832 (L) with or without UCN-01 (UCN or U, U937, 100 nM; Jurkat, 150 nM). At the indicated intervals, cells were harvested and the percentage of cells exhibiting apoptotic morphology was determined by evaluating Wright-Giemsa-stained cytospin preparations. (B) U937 and Jurkat cells were incubated for 18 hours with the indicated concentration of UCN-01 in either the presence or absence of 10 μM L744832, after which the percentage of apoptotic cells was determined as described in panel A. (C) U937 and Jurkat cells were exposed to the indicated concentration of L744832 in either the presence or absence of UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which the percentage of apoptotic cells was determined as described in panel A. (D) U937 (upper panel) and Jurkat (lower panel) cells were exposed to a range of L744832 (5-25 μM) and UCN-01 (50-250 nM) concentrations alone and in combination at a fixed ratio (eg, U937, 100:1; Jurkat, 66.7:1) for 18 hours. At the end of this period, the percentage of cells exhibiting annexin V positivity was determined for each condition. Fractional effect values were determined by comparing results with those of untreated controls and median dose effect analysis employed to characterize the nature of the interaction between L744832 and UCN-01. Combination index (CI) values less than 1.0 denote a synergistic interaction. Two additional studies yielded equivalent results. (E) U937 and Jurkat cells were treated with 10 μM L744832 in either the presence or absence of UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which cells were lysed and subjected to Western blot to assess activation of caspase-associated cascades using the indicated primary antibodies.Alternatively, cytosolic fractions (S-100) were prepared, and expression of cytochrome c, Smac/DIABLO, and AIF was monitored by Western blot. (F) U937 cells were incubated with 10 μM L744832 (L) + 100 nM UCN-01 (U) for 8 hours in either the presence or absence of 20 μM BOC-D-fmk, after which the percentage of apoptotic cells was determined by annexin V-FITC/flow cytometry (upper panel). **Significantly lower than values for cells exposed to L744832 + UCN-01 in the absence of BOC-D-fmk (P < .01). Alternatively, S-100 fractions were prepared and subjected to Western blot analysis to monitor cytosolic expression of cytochrome c, Smac/DIABLO, and AIF (lower panel). For panels A-C and F (upper panel), results represent the means ± SD for 3 separate experiments performed in triplicate. For panels E and F (lower panels), each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. The results of a representative experiment are shown; an additional study yielded equivalent results. CF indicates cleavage fragment.
L744832 rapidly and highly synergistically potentiates UCN-01-mediated lethality in human leukemia cells, accompanied by mitochondrial dysfunction, caspase activation, and PARP cleavage. (A) U937 and Jurkat cells were exposed to 10 μM L744832 (L) with or without UCN-01 (UCN or U, U937, 100 nM; Jurkat, 150 nM). At the indicated intervals, cells were harvested and the percentage of cells exhibiting apoptotic morphology was determined by evaluating Wright-Giemsa-stained cytospin preparations. (B) U937 and Jurkat cells were incubated for 18 hours with the indicated concentration of UCN-01 in either the presence or absence of 10 μM L744832, after which the percentage of apoptotic cells was determined as described in panel A. (C) U937 and Jurkat cells were exposed to the indicated concentration of L744832 in either the presence or absence of UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which the percentage of apoptotic cells was determined as described in panel A. (D) U937 (upper panel) and Jurkat (lower panel) cells were exposed to a range of L744832 (5-25 μM) and UCN-01 (50-250 nM) concentrations alone and in combination at a fixed ratio (eg, U937, 100:1; Jurkat, 66.7:1) for 18 hours. At the end of this period, the percentage of cells exhibiting annexin V positivity was determined for each condition. Fractional effect values were determined by comparing results with those of untreated controls and median dose effect analysis employed to characterize the nature of the interaction between L744832 and UCN-01. Combination index (CI) values less than 1.0 denote a synergistic interaction. Two additional studies yielded equivalent results. (E) U937 and Jurkat cells were treated with 10 μM L744832 in either the presence or absence of UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which cells were lysed and subjected to Western blot to assess activation of caspase-associated cascades using the indicated primary antibodies.Alternatively, cytosolic fractions (S-100) were prepared, and expression of cytochrome c, Smac/DIABLO, and AIF was monitored by Western blot. (F) U937 cells were incubated with 10 μM L744832 (L) + 100 nM UCN-01 (U) for 8 hours in either the presence or absence of 20 μM BOC-D-fmk, after which the percentage of apoptotic cells was determined by annexin V-FITC/flow cytometry (upper panel). **Significantly lower than values for cells exposed to L744832 + UCN-01 in the absence of BOC-D-fmk (P < .01). Alternatively, S-100 fractions were prepared and subjected to Western blot analysis to monitor cytosolic expression of cytochrome c, Smac/DIABLO, and AIF (lower panel). For panels A-C and F (upper panel), results represent the means ± SD for 3 separate experiments performed in triplicate. For panels E and F (lower panels), each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. The results of a representative experiment are shown; an additional study yielded equivalent results. CF indicates cleavage fragment.
Median dose effect analysis using apoptosis induction by annexin V/PI staining at 18 hours as an end point revealed that combined exposure of cells to L744832 (5 to 25μM) and UCN-01 (50 to 250 nM) at fixed ratios (100:1 and 66.7:1, respectively) resulted in combination index (CI) values less than 0.3, corresponding to highly synergistic interactions, in both U937 and Jurkat cells (Figure 2D).
Combined exposure of leukemic cells to UCN-01 and L744832 results in a dramatic increase in mitochondrial dysfunction, caspase activation, and PARP cleavage
Western blot analysis revealed that combined, but not individual, treatment of U937 and Jurkat cells with L744832 and UCN-01 resulted in the pronounced release of cytochrome c, Smac/DIABLO, and AIF into the cytosolic S-100 fraction, accompanied by the activation/cleavage of caspase-2, -3, -9, -8, and Bid as well as PARP degradation (Figure 2E). Similar results were obtained when loss of Δψm was monitored or when HL-60 and Raji cells were examined (data not shown). Furthermore, the broad caspase inhibitor BOC-D-fmk significantly reduced L744832/UCN-01-induced apoptosis in U937 cells (Figure 2F) but failed to attenuate cytochrome c release. However, it largely blocked Smac/DIABLO and partially blocked AIF release in response to this combination (Figure 2F). Together, these findings indicate that the L744832/UCN-01 regimen triggers caspase-independent activation of the mitochondrial pathway, manifested by cytochrome c release, accompanied by the caspase-dependent release of Smac/DIABLO and AIF.31,32
The L744832/UCN-01 regimen induces cleavage of Bcl-2, Mcl-1, and XIAP
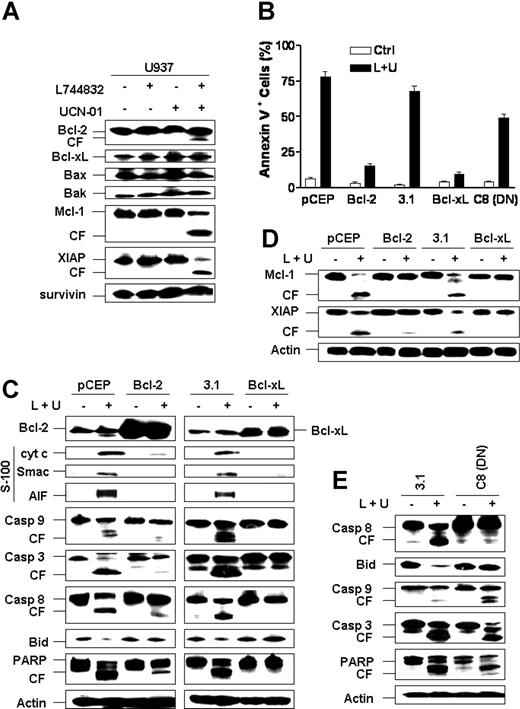
Combined treatment of U937 cells with L744832/UCN-01 did not substantially alter expression of either antiapoptotic (eg, Bcl-2 and Bcl-xL) or proapoptotic (eg, Bax and Bak) Bcl-2 family members (Figure 3A). However, a putatively proapoptotic Bcl-2 cleavage product33 was noted after combined but not individual treatment, along with cleavage and reduced expression of Mcl-1 and XIAP. Similar results were observed in Jurkat cells (data not shown). Thus, L744832/UCN-01 induced cleavage of the antiapoptotic proteins Bcl-2, Mcl-1, and XIAP to putatively proapoptotic forms.34,35
L744832/UCN-01-induced apoptosis is primarily mediated by activation of the intrinsic, mitochondrial pathway. (A) U937 cells were exposed to 10 μM L744832 in either the presence or absence of 100 nM UCN-01 for 18 hours, after which expression of Bcl-2 family molecules (eg, Bcl-2, Bcl-xL, Bax, Bak, and Mcl-1) and XIAP was evaluated by Western blot analysis. (B) U937 cells stably transfected with Bcl-2 or its empty vector (pCEP) or, alternatively, Bcl-xL, dominant-negative (DN) caspase-8 (C8), or their respective empty vector (pcDNA3.1) were treated with 10 μM L744832 (L) + 100 nM UCN-01 (U) for 18 hours, after which the percentage of apoptotic cells was determined by annexin V-FITC/flow cytometry. Results represent the means ± SD for 3 separate experiments performed in triplicate. (C-D) Bcl-2/U937, pCEP/U937, Bcl-xL/U937 and pcDNA3.1/U937 cells were treated as described in panel B, after which whole cells were lysed and subjected to Western blot analysis to monitor expression of Bcl-2 or Bcl-xL and cleavage of caspases, Bid, PARP (C), Mcl-1, and XIAP (D). Alternatively, S-100 fractions were prepared as described in “Materials and methods,” and Western blot analysis was performed to assess cytosolic distribution of cytochrome c, Smac/DIABLO, and AIF. (E) DN caspase-8/U937 and pcDNA3.1/U937 cells were treated as described in panel B, after which Western blot analysis was employed to monitor expression of caspase-8 and cleavage of caspases, Bid, and PARP. For panels A and C-E, each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin, as indicated, to ensure equivalent loading and transfer of protein. Two additional studies yielded equivalent results. CF indicates cleavage fragment.
L744832/UCN-01-induced apoptosis is primarily mediated by activation of the intrinsic, mitochondrial pathway. (A) U937 cells were exposed to 10 μM L744832 in either the presence or absence of 100 nM UCN-01 for 18 hours, after which expression of Bcl-2 family molecules (eg, Bcl-2, Bcl-xL, Bax, Bak, and Mcl-1) and XIAP was evaluated by Western blot analysis. (B) U937 cells stably transfected with Bcl-2 or its empty vector (pCEP) or, alternatively, Bcl-xL, dominant-negative (DN) caspase-8 (C8), or their respective empty vector (pcDNA3.1) were treated with 10 μM L744832 (L) + 100 nM UCN-01 (U) for 18 hours, after which the percentage of apoptotic cells was determined by annexin V-FITC/flow cytometry. Results represent the means ± SD for 3 separate experiments performed in triplicate. (C-D) Bcl-2/U937, pCEP/U937, Bcl-xL/U937 and pcDNA3.1/U937 cells were treated as described in panel B, after which whole cells were lysed and subjected to Western blot analysis to monitor expression of Bcl-2 or Bcl-xL and cleavage of caspases, Bid, PARP (C), Mcl-1, and XIAP (D). Alternatively, S-100 fractions were prepared as described in “Materials and methods,” and Western blot analysis was performed to assess cytosolic distribution of cytochrome c, Smac/DIABLO, and AIF. (E) DN caspase-8/U937 and pcDNA3.1/U937 cells were treated as described in panel B, after which Western blot analysis was employed to monitor expression of caspase-8 and cleavage of caspases, Bid, and PARP. For panels A and C-E, each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin, as indicated, to ensure equivalent loading and transfer of protein. Two additional studies yielded equivalent results. CF indicates cleavage fragment.
L744832/UCN-01-mediated apoptosis primarily proceeds via the intrinsic, mitochondrial pathway
To gain further insights into the roles of the intrinsic, mitochondrial and the extrinsic, receptor-related apoptotic pathways in L744832/UCN-01-induced apoptosis, cells ectopically expressing Bcl-2, Bcl-xL, or DN caspase-8 were employed. Whereas ectopic expression of Bcl-2 or Bcl-xL dramatically reduced L744832/UCN-01-mediated apoptosis (and loss of Δψm; data not shown), DN caspase-8 exerted only a partial protective effect (Figure 3B). Consistent with these results, ectopic expression of Bcl-2 or Bcl-xL markedly reduced release of cytochrome c, Smac/DIABLO, and AIF into the cytosolic S-100 fraction, accompanied by the marked attenuation of caspase-3 and -9 activation and PARP degradation (Figure 3C).
Interestingly, cells ectopically expressing Bcl-2 or Bcl-xL were also protected from L744832/UCN-01-mediated cleavage of procaspase-8 and Bid (Figure 3C). As anticipated, expression of DN caspase-8 effectively blocked L744832/UCN-01-mediated caspase-8 activation and Bid cleavage but was much less effective in blocking caspase-9 and -3 activation or PARP degradation (Figure 3E). Collectively, these findings are consistent with the notion that the L744832/UCN-01 regimen induces apoptosis in leukemic cells primarily by activating the intrinsic, mitochondrial apoptotic pathway but allows for the possibility that secondary activation of the extrinsic pathway may amplify this process.36 Finally, to determine whether cleavage of Bcl-2, Mcl-1, and XIAP, which have been shown to be proapoptotic,33-35 is mediated by activation of caspase cascades, U937 cells ectopically expressing Bcl-2 or Bcl-xL were employed. As shown in Figure 3C-D, overexpression of either Bcl-2 or Bcl-xL, which almost completely blocked apoptosis (Figure 3B) and activation of caspases (Figure 3C), substantially diminished cleavage of Mcl-1 and XIAP. Cleavage of Bcl-2 was also attenuated in cells ectopically expressing Bcl-2 or Bcl-xL (data not shown). These results raise the possibility that while cleavage of Bcl-2, Mcl-1, and XIAP most likely represents secondary, caspase-dependent events, the cleaved products may nevertheless potentiate cell death induced by L744832/UCN-01 combination.
L744832 leads to MEK/ERK inactivation in UCN-01-treated cells while promoting SEK/JNK and p34cdc2 activation
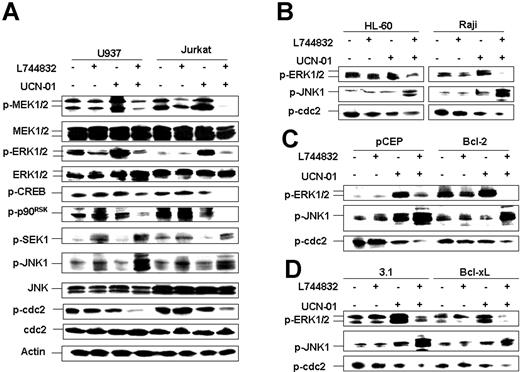
As previously reported,23 exposure to UCN-01 alone resulted in an increase in MEK1/2 and ERK1/2 phosphorylation/activation in both U937 and Jurkat cells (Figure 4A). Significantly, these effects were essentially abrogated by coadministration of L744832. Combined treatment also dramatically reduced phosphorylation of CREB and p90RSK, 2 downstream targets of the Raf/MEK/ERK pathway. Thus, L744832 mimicked the actions of MEK inhibitors in blocking UCN-01-mediated activation of the MEK/ERK pathway in UCN-01-treated leukemia cells.23,24
L744832 blocks UCN-01-induced MEK/ERK activation while promoting activation of SEK/JNK and p34cdc2 upstream of mitochondrial dysfunction. (A) U937 and Jurkat cells were exposed to 10 μM L744832 in either the presence or absence of UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which Western blot analysis was performed to monitor phosphorylation of the p42/44MAPK-related proteins (eg, MEK, ERK, CREB, and p90RSK), stress-related (SAPK/JNK) proteins (eg, SEK1 and JNK1), and cdc2/Cdk1. (B) The phosphorylation of ERK1/2, JNK1, and cdc2 was also monitored in HL-60 and Raji cells after treatment with 10 μM L744832 with or without UCN-01 (HL-60, 100 nM; Raji, 150 nM) for 18 hours. (C-D) Bcl-2/U937 and pCEP/U937 cells (C) or Bcl-xL/U937 and pcDNA3.1/U937 cells (D) were exposed to 10 μM L744832 with or without 100 nM UCN-01 for 18 hours, after which cells were lysed and subjected to Western blot analysis to assess phosphorylation statuses of ERK1/2, JNK1, and cdc2. In each panel, 30 μg protein was loaded in each lane; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. The results are representative of 3 separate experiments.
L744832 blocks UCN-01-induced MEK/ERK activation while promoting activation of SEK/JNK and p34cdc2 upstream of mitochondrial dysfunction. (A) U937 and Jurkat cells were exposed to 10 μM L744832 in either the presence or absence of UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which Western blot analysis was performed to monitor phosphorylation of the p42/44MAPK-related proteins (eg, MEK, ERK, CREB, and p90RSK), stress-related (SAPK/JNK) proteins (eg, SEK1 and JNK1), and cdc2/Cdk1. (B) The phosphorylation of ERK1/2, JNK1, and cdc2 was also monitored in HL-60 and Raji cells after treatment with 10 μM L744832 with or without UCN-01 (HL-60, 100 nM; Raji, 150 nM) for 18 hours. (C-D) Bcl-2/U937 and pCEP/U937 cells (C) or Bcl-xL/U937 and pcDNA3.1/U937 cells (D) were exposed to 10 μM L744832 with or without 100 nM UCN-01 for 18 hours, after which cells were lysed and subjected to Western blot analysis to assess phosphorylation statuses of ERK1/2, JNK1, and cdc2. In each panel, 30 μg protein was loaded in each lane; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. The results are representative of 3 separate experiments.
Reciprocal changes in the stress-related SEK/JNK pathway were observed—that is, combined treatment with L744832 and UCN-01 resulted in a pronounced increase in SEK1 and JNK phosphorylation/activation, compared with individual drug treatment. In separate studies, inhibition of ERK phosphorylation and potentiation of JNK activation by L744832 were observed at intervals as early as 2 to 4 hours following treatment, events occurring prior to discernible PARP cleavage, which was noted at 4 to 6 hours in both U937 and Jurkat cells (data not shown). On the other hand, cotreatment with UCN-01 and L744832 did not lead to alterations in phosphorylation (activation) status of p38 MAPK in either U937 and Jurkat cells (data not shown). Analogous reciprocal changes in ERK inactivation and JNK activation were observed in HL-60 and Raji cells exposed to the L744832/UCN-01 combination (Figure 4B). Lastly, similar perturbations in JNK activation and ERK inactivation were noted in U937 cells ectopically expressing Bcl-2 or Bcl-xL (Figure 4C-D) despite the abrogation of mitochondrial dysfunction (Figure 3C), indicating that these events operate upstream of UCN-01/L744832-mediated mitochondrial dysfunction.
Consistent with evidence that coadministration of MEK1/2 inhibitors with UCN-01 results in the marked activation of p34cdc2,23,24 L744832-mediated inactivation of MEK/ERK in UCN-01-treated U937 and Jurkat cells led to the pronounced dephosphorylation/activation of p34cdc2 (Figure 4A). Similar results were obtained in HL-60 and Raji cells (Figure 4B) and in U937 cells ectopically expressing Bcl-2 or Bcl-xL (Figure 4C-D), raising the possibility that antileukemic synergism between UCN-01 and L744832, as with MEK inhibitors, may also be related to unscheduled p34cdc2 activation.
Combined exposure of leukemic cells to UCN-01 and L744832 inactivates Akt and downstream signaling pathways
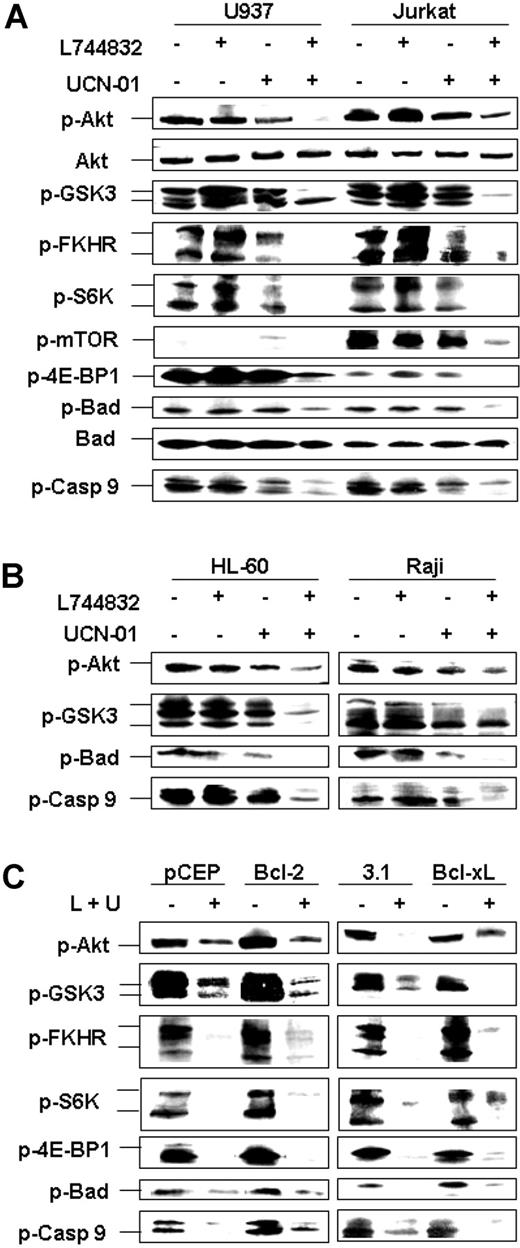
In view of evidence that both FTIs and UCN-01 can interrupt Akt/PDK1,16,37 the effects of combined treatment of leukemic cells with L744832 and UCN-01 were examined in relation to perturbations in Akt-related signaling pathways. Exposure of U937 cells to UCN-01 alone resulted in diminished levels of the phosphorylated (activated) form of Akt (Figure 5A), consistent with previous results.16 However, coadministration of L744832, which by itself had little effect, substantially reduced Akt phosphorylation. These reductions were observed within 4 hours of drug exposure in both U937 and Jurkat cells and occurred prior to PARP cleavage (data not shown). It has also been reported that FTIs inactivate p70 S6 kinase (p70S6K), a downstream target of PI3K/Akt, which may lead to inhibition of DNA synthesis and cell growth.38 However, while individual agents had little effect, coadministration of L744832 with UCN-01 essentially abrogated phospho-p70S6K expression in both cell lines. Similar responses were observed for the phosphorylated forms of 2 other Akt downstream targets, GSK-3α/β and FKHR. In addition, phosphorylation of the Akt substrates Bad and caspase-9, phenomena associated with antiapoptotic actions,39,40 was diminished only in cells exposed to both agents.
Combined exposure to UCN-01 and L744832 inactivates Akt signaling pathways independently of mitochondrial-mediated apoptosis. (A) U937 and Jurkat cells were treated with 10 μM L744832 with or without UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which Western blot analysis was performed to evaluate expression of total and/or phosphorylated Akt and its downstream targets (eg, GSK-3α/β, FKHR/FKHRL, p70/p85 S6 kinase, mTOR, 4E-BP1, Bad, and caspase-9). (B) Phosphorylation of Akt, GSK-3α/β, Bad, and caspase-9 was assessed in HL-60 and Raji cells after treatment with 10 μM L744832 ± UCN-01 (HL-60, 100 nM; Raji, 150 nM) for 18 hours. (C) Bcl-2/U937 and pCEP/U937 (left panels) or Bcl-xL/U937 and pcDNA3.1/U937 cells (right panels) were exposed to 10 μM L744832 (L) + 100 nM UCN-01 (U) for 18 hours, after which the phosphorylation status of Akt, GSK-3α/β, FKHR/FKHRL, p70/p85 S6 kinase, 4E-BP1, Bad, and caspase-9 was monitored by Western blot analysis. In each panel, each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. Two additional studies yielded equivalent results.
Combined exposure to UCN-01 and L744832 inactivates Akt signaling pathways independently of mitochondrial-mediated apoptosis. (A) U937 and Jurkat cells were treated with 10 μM L744832 with or without UCN-01 (U937, 100 nM; Jurkat, 150 nM) for 18 hours, after which Western blot analysis was performed to evaluate expression of total and/or phosphorylated Akt and its downstream targets (eg, GSK-3α/β, FKHR/FKHRL, p70/p85 S6 kinase, mTOR, 4E-BP1, Bad, and caspase-9). (B) Phosphorylation of Akt, GSK-3α/β, Bad, and caspase-9 was assessed in HL-60 and Raji cells after treatment with 10 μM L744832 ± UCN-01 (HL-60, 100 nM; Raji, 150 nM) for 18 hours. (C) Bcl-2/U937 and pCEP/U937 (left panels) or Bcl-xL/U937 and pcDNA3.1/U937 cells (right panels) were exposed to 10 μM L744832 (L) + 100 nM UCN-01 (U) for 18 hours, after which the phosphorylation status of Akt, GSK-3α/β, FKHR/FKHRL, p70/p85 S6 kinase, 4E-BP1, Bad, and caspase-9 was monitored by Western blot analysis. In each panel, each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. Two additional studies yielded equivalent results.
Different activation patterns were observed in the case of 2 additional Akt targets, mTOR and 4E-BP1.41,42 In Jurkat cells, mTOR was constitutively phosphorylated, whereas basal levels of phosphorylated mTOR were essentially undetectable in U937 cells (Figure 5A). In contrast, basal levels of phosphorylated 4E-BP1 were pronounced in U937 cells but minimal in Jurkat cells. A marked reduction in expression of phospho-4E-BP1 (in U937 and Jurkat cells) or mTOR (in Jurkat cells) was only observed in cells exposed to both UCN-01 and L744832. The UCN-01/L744832 regimen also resulted in a clear reduction in levels of phospho-Akt, -GSK-3, -Bad, and -caspase-9 in other leukemia cells (eg, HL-60 and Raji cells) (Figure 5B). Collectively, these findings indicate that combined exposure to UCN-01 and L744832 results in a pronounced reduction in levels of phosphorylated Akt and the phosphorylated forms of several of its downstream targets in multiple human leukemia cell types.
Finally, ectopic expression of Bcl-2 or Bcl-xL was either ineffective or minimally effective in attenuating Akt, GSK-3, FKHR, p70S6K, 4E-BP1, Bad, and caspase-9 phosphorylation following UCN-01/L744832 exposure (Figure 5C), arguing against the possibility that inactivation of the Akt pathway represents a secondary phenomenon.
Inducible expression of constitutively active Akt but not MEK attenuates UCN-01/L744832 lethality
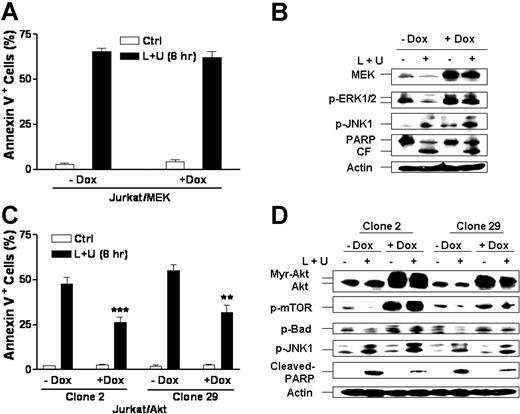
To determine the functional significance of MEK/ERK and Akt inactivation in UCN-01/L744832-mediated lethality, Jurkat cells inducibly expressing myristoylated (active) Akt or constitutively active MEK1 were employed. When Tet-on MEK1 cells were exposed for 8 hours to the combination of UCN-01 and L744832 following 24-hour preincubation (induction) with doxycycline, no reduction in apoptosis was observed (P > .05; Figure 6A) despite circumvention of ERK inactivation induced by UCN-01/L744832 treatment (Figure 6B). Consistent with its inability to attenuate apoptosis, inducible expression of constitutively active MEK1 failed to block UCN-01/L744832-mediated JNK activation or PARP cleavage, indicating that enforced activation of MEK/ERK by itself is insufficient to prevent apoptosis induced by the UCN-01/L744832 regimen, in contrast to results previously reported in the case of UCN-01/MEK inhibitors.30
Inducible expression of myristoylated Akt but not constitutively active MEK attenuates the lethality of UCN-01/L744832 regimen in human leukemia cells. (A) Jurkat cells stably transfected with a Tet-on inducible constitutively active MEK construct were exposed to 10 μM L744832 (L) + 150 nM UCN-01 (U) for 8 hours following a 24-hour induction with 2 μg/mL doxycycline (Dox), after which the percentage of apoptotic cells was determined by annexin V-FITC/flow cytometry. (B) MEK/Tet-on Jurkat cells were treated as in panel A, after which cells were lysed and subjected to Western blot analysis to monitor expression of MEK, phosphorylation of ERK1/2 and JNK1, and cleavage of PARP. (C) Jurkat cells (clone no. 2 and clone no. 29) stably transfected with a Tet-on inducible myristoylated Akt construct were incubated for 8 hours with 10 μM L744832 + 150 nM UCN-01 following a 24-hour induction with 2 μg/mL Dox, after which annexin V-FITC/flow cytometry was performed to determine the percentage of apoptotic cells. (D) Alternatively, 2 clones (no. 2 and no. 29) of Akt/Tet-on Jurkat cells were treated as described in panel C, after which Western blot analysis was employed to assess expression of myr-Akt, phosphorylation status of mTOR, Bad, and JNK1, and cleavage of PARP. For panels A and C, the results represent the means ± SD for 3 separate experiments performed in triplicate. ***P < .02 and **P < .05); significantly lower than values for cells of the same clone exposed to L744832 + UCN-01 without Dox-induction. For panels B and D, 30 μg protein was loaded in each lane; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. The results were representative of 3 separate experiments. CF indicates cleavage fragment. Two additional studies yielded equivalent results.
Inducible expression of myristoylated Akt but not constitutively active MEK attenuates the lethality of UCN-01/L744832 regimen in human leukemia cells. (A) Jurkat cells stably transfected with a Tet-on inducible constitutively active MEK construct were exposed to 10 μM L744832 (L) + 150 nM UCN-01 (U) for 8 hours following a 24-hour induction with 2 μg/mL doxycycline (Dox), after which the percentage of apoptotic cells was determined by annexin V-FITC/flow cytometry. (B) MEK/Tet-on Jurkat cells were treated as in panel A, after which cells were lysed and subjected to Western blot analysis to monitor expression of MEK, phosphorylation of ERK1/2 and JNK1, and cleavage of PARP. (C) Jurkat cells (clone no. 2 and clone no. 29) stably transfected with a Tet-on inducible myristoylated Akt construct were incubated for 8 hours with 10 μM L744832 + 150 nM UCN-01 following a 24-hour induction with 2 μg/mL Dox, after which annexin V-FITC/flow cytometry was performed to determine the percentage of apoptotic cells. (D) Alternatively, 2 clones (no. 2 and no. 29) of Akt/Tet-on Jurkat cells were treated as described in panel C, after which Western blot analysis was employed to assess expression of myr-Akt, phosphorylation status of mTOR, Bad, and JNK1, and cleavage of PARP. For panels A and C, the results represent the means ± SD for 3 separate experiments performed in triplicate. ***P < .02 and **P < .05); significantly lower than values for cells of the same clone exposed to L744832 + UCN-01 without Dox-induction. For panels B and D, 30 μg protein was loaded in each lane; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. The results were representative of 3 separate experiments. CF indicates cleavage fragment. Two additional studies yielded equivalent results.
Parallel studies were performed using cells inducibly expressing a myristoylated (active) Akt. In marked contrast to results involving the inducible expression of constitutively active MEK1, 2 separate Tet-on Akt clones (no. 2 and no. 29) displayed a partial but statistically significant reduction in UCN-01/L744832-mediated apoptosis (P < .02 or .05; Figure 6C). A parallel reduction in PARP cleavage was also observed (Figure 6D). Notably, doxycycline-induced ectopic expression of myristoylated Akt increased phosphorylation of mTOR and Bad while preventing L744832/UCN-01-mediated dephosphorylation of these proteins. Enforced expression of myristoylated Akt failed to diminish JNK phosphorylation induced by the UCN-01/L744832 combination (Figure 6D), suggesting that JNK activation might play a separate role in the lethality of this regimen. Collectively, these findings indicate that enforced activation of Akt inactivation by itself, unlike MEK1, can attenuate UCN-01/L744832-related apoptosis.
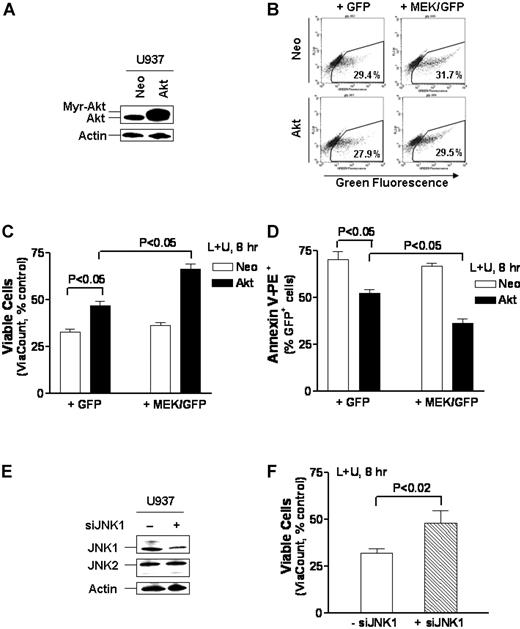
Enforced MEK/ERK activation significantly protects against UCN-01/L744832-mediated lethality only in cells ectopically expressing constitutively active Akt
In view of evidence of cooperation between the Akt and ERK pathways,43,44 the possibility existed that under conditions in which Akt is inactivated, interruption of the MEK/ERK pathway may lead to further potentiation of cell death. To test this possibility, U937 cells stably transfected with a myristoylated (active) Akt construct (Akt/U937) were transiently transfected with either a control green fluorescent protein (GFP) vector or a GFP-tagged constitutively active MEK1 construct, after which cell death was monitored in the GFP-positive cell population. Western blot analysis demonstrated ectopic expression of myristoylated Akt in Akt/U937 cells compared with empty vector controls (Neo/U937; Figure 7A), and a transient transfection efficiency of approximately 30% was obtained for both Neo/U937 and Akt/U937 cells transfected with either GFP or the constitutively active MEK1/GFP constructs (Figure 7B). Akt/U937 cells were significantly more resistant to UCN-01/L744832-mediated lethality than Neo/U937 controls (P < .05, Figure 7C-D), consistent with results involving Jurkat cells inducibly expressing active Akt (Figure 6C). Moreover, in Neo/U937 cells, transient transfection with the constitutively active MEK1/GFP construct failed to protect cells from UCN-01/L744832-mediated lethality (P > .05), consistent with results in MEK1-inducible Jurkat cells (Figure 6A). However, in marked contrast to results in Neo/U937 cells, transfection with the constitutively active MEK1/GFP construct in Akt/U937 cells led to a statistically significant increase in protection from UCN-01/L744832-mediated lethality (Figure 7C-D; P < .05). Together, these findings suggest that UCN-01/L744832-mediated down-regulation of the MEK/ERK pathway, while ineffective by itself, may promote apoptosis under conditions in which the Akt cascade is simultaneously inactivated.
Enforced MEK/ERK activation requires ectopic expression of Akt to protect cells from UCN-01/L744832-mediated apoptosis, while JNK activation plays a functional role in the lethality of this regimen. (A) U937 cells were stably transfected with constitutively active myr-Akt (Akt/U937) and its empty vector (Neo/U937), and Western blot analysis was performed to monitor expression of Akt. (B) Akt/U937 and Neo/U937 cells were transiently transfected with either MEK/EGFP or enhanced GFP (EGFP). After 24 hours, flow cytometry was employed to monitor transfection efficiency by determining the percentage of GFP-positive cells within the gated areas. Results are representative of 3 separate experiments. (C) MEK/EGFP and EGFP-transfected Akt/U937 or Neo/U937 cells, after a 24-hour recovery, were incubated for 8 hours with 10 μM L744832 + 100 nM UCN-01, after which the percentage of viable cells was determined using the Guava ViaCount assay as described in “Materials and methods.” (D) Alternatively, cells were stained by annexin V-PE and subjected to flow cytometric analysis to determine the percentage of annexin V-positive/GFP-positive cells. (E) U937 cells were transfected with JNK1 siRNA oligonucleotides as described in “Materials and methods.” After 24 hours, the levels of JNK1 and JNK2 were monitored by Western blot analysis. (F) Following a 24-hour recovery, JNK1 siRNA-transfected U937 cells were exposed to 10 μM L744832 + 100 nM UCN-01 for 8 hours, after which the percentage of viable cells was determined by ViaCount assay. For panels A and E, each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. Two additional studies yielded equivalent results. For panels C, D, and F, values represent the means ± SD for 3 separate experiments performed in triplicate.
Enforced MEK/ERK activation requires ectopic expression of Akt to protect cells from UCN-01/L744832-mediated apoptosis, while JNK activation plays a functional role in the lethality of this regimen. (A) U937 cells were stably transfected with constitutively active myr-Akt (Akt/U937) and its empty vector (Neo/U937), and Western blot analysis was performed to monitor expression of Akt. (B) Akt/U937 and Neo/U937 cells were transiently transfected with either MEK/EGFP or enhanced GFP (EGFP). After 24 hours, flow cytometry was employed to monitor transfection efficiency by determining the percentage of GFP-positive cells within the gated areas. Results are representative of 3 separate experiments. (C) MEK/EGFP and EGFP-transfected Akt/U937 or Neo/U937 cells, after a 24-hour recovery, were incubated for 8 hours with 10 μM L744832 + 100 nM UCN-01, after which the percentage of viable cells was determined using the Guava ViaCount assay as described in “Materials and methods.” (D) Alternatively, cells were stained by annexin V-PE and subjected to flow cytometric analysis to determine the percentage of annexin V-positive/GFP-positive cells. (E) U937 cells were transfected with JNK1 siRNA oligonucleotides as described in “Materials and methods.” After 24 hours, the levels of JNK1 and JNK2 were monitored by Western blot analysis. (F) Following a 24-hour recovery, JNK1 siRNA-transfected U937 cells were exposed to 10 μM L744832 + 100 nM UCN-01 for 8 hours, after which the percentage of viable cells was determined by ViaCount assay. For panels A and E, each lane was loaded with 30 μg protein; blots were subsequently stripped and reprobed for expression of β-actin to ensure equivalent loading and transfer of protein. Two additional studies yielded equivalent results. For panels C, D, and F, values represent the means ± SD for 3 separate experiments performed in triplicate.
JNK activation plays a significant functional role in UCN-01/L744832-mediated lethality
To assess the functional significance in JNK activation, U937 cells were transfected with JNK1 siRNA, leading to clear reductions in levels of JNK1 (Figure 7E). A significant increase in cell viability in cells transfected with the JNK1 siRNA was observed following UCN-01/L744832 exposure (P < .02; Figure 7F). Consistent with these results, coadministration of the JNK inhibitor SP600125 (20 μM)45 with UCN-01/L744832 resulted in partial but statistically significant reductions in the percentage of U937 cells displaying a loss of Δψm or annexin V positivity (P < .05 and .02, respectively; data not shown), indicating that enhanced activation of the stress kinase JNK plays a significant functional role in UCN-01/L744832-mediated lethality.
Discussion
The present results indicate that the FTI L744832 dramatically increases the lethality of the Chk1 inhibitor UCN-01 in diverse human leukemia cell lines as well as in primary human AML blast cells. Because Ras is frequently mutated in human cancer, efforts to target this protein have been the focus of attention.1 Additionally, FTIs potentiate the lethal effects of conventional cytotoxic agents in preclinical animal model systems.9,11 However, the ability of FTIs to enhance the lethality of other molecularly targeted agents has been little explored. The present findings indicate that in human leukemia cells, FTIs also interact synergistically with UCN-01, an agent that acts by interrupting signal transduction and cell cycle regulatory pathways.
Interactions between UCN-01 and L744832 resembled to some extent those previously described in studies involving pharmacologic MEK inhibitors such as U0126 and PD184352 (CI-1040).23,24 Although UCN-01 was originally developed as a selective PKC inhibitor, it was subsequently shown to inhibit various Cdks,46 the Chk1 kinase,15 as well as PDK1/Akt.16 Furthermore, the lethality of UCN-01 in human leukemia cells has been associated with dephosphorylation/activation of Cdk1.23 Previously, we have shown that exposure of human leukemia cells to marginally toxic concentrations of UCN-01 leads to activation of the MEK/ERK pathway and that interruption of this process (eg, by MEK inhibitors) dramatically increases cell death.23,24 It may not be coincidental that both MEK inhibitors as well as FTIs (eg, L744832) resulted in ERK1/2 inactivation and a pronounced increase in dephosphorylation of p34cdc2, inappropriate activation of which is known to be a potent apoptotic stimulus.47 It is therefore tempting to speculate that interference with multiple Ras downstream cytoprotective targets, including ERK and Akt, may lower the threshold for apoptosis induced by dysregulation of p34cdc2.
The addition of a farnesyl group to conserved amino acid residues at the Ras carboxy terminus allows the protein to be translocated to the plasma membrane and activated.1 Downstream targets of Ras include members of the Raf family (ie, Raf-1, B-Raf, and A-Raf) along with their targets (eg, MEK1/2 and ERK1/2).3 Another important substrate is the catalytic subunit of type 1 phosphatidylinositol 3-kinases (PI3Ks) and their targets PDK1 and Akt.8,37,48 Other Ras effectors include members of the Ral family49 and phospholipase C.50 The question thus arises as to which of the diverse actions of FTIs is primarily responsible for synergistic antileukemic interactions with UCN-01. Importantly, while enforced activation of Akt significantly protected cells from UCN-01/L744832-mediated apoptosis, prevention of MEK/ERK inactivation by a constitutively active construct by itself did not. This stands in marked contrast to our previous observation that enforced expression of active MEK1 significantly protects human multiple myeloma cells from the lethal effects of combined exposure to UCN-01 and the MEK inhibitor U0126.30 Such findings suggest that the mechanism by which L744832 potentiates UCN-01 lethality differs fundamentally from that of pharmacologic MEK inhibitors. In this context, both Akt and ERK have been shown to oppose induction of apoptosis through multiple mechanisms, including phosphorylation and inactivation of several proapoptotic proteins, including Bad39,51 and procaspase-9 40,52 among others. Consistent with these observations, combined exposure to UCN-01 and L744832 led to diminished Bad phosphorylation on Ser136, an event that was attenuated by enforced activation of Akt. However, combined treatment with these agents resulted in other perturbations that might have contributed to enhanced lethality, including dephosphorylation/inactivation of p34cdc2, and activation of JNK, all of which may have contributed to potentiation of apoptosis.48,53
It is particularly interesting that while enforced activation of MEK/ERK by itself, unlike Akt, failed to attenuate UCN-01/L744832-mediated apoptosis, it did significantly increase protection in cells displaying enforced Akt activation. Evidence of cross-talk between the Raf/MEK/ERK and Akt pathways has previously been described.54 Furthermore, cooperation between the PI3K/Akt and Raf/MEK/ERK pathways in suppressing apoptosis has recently been described in human leukemia cells.43 For example, MEK-mediated cell growth and prevention of apoptosis requires activation of the PI3K/Akt pathway in leukemia cells exposed to stimuli such as growth factor deprivation.44 Thus, the present findings raise the possibility that Akt and ERK, both downstream targets of Ras,3,8 may cooperate to protect leukemic cells from certain noxious stimuli (eg, UCN-01) and that combined blockade of these pathways (eg, by FTIs) may be particularly lethal to cells.
The bulk of evidence indicates that the UCN-01/L744832 regimen primarily kills leukemia cells by activating the intrinsic, mitochondrial apoptotic pathway. This assertion is based on evidence that UCN-01/L744832-mediated lethality was associated with a marked induction of mitochondrial dysfunction (eg, cytochrome c release), caspase activation, and apoptosis and that these events were substantially blocked by ectopic expression of Bcl-2 or Bcl-xL, which act in large part by preventing mitochondrial-dependent pathways.55 In contrast, protection by dominant-negative caspase-8 was considerably less pronounced. Nevertheless, combined treatment did result in enhanced activation of caspase-8, and it is likely that this process operates to amplify the apoptotic cascade, most likely by promoting Bid cleavage/activation.36 Similarly, combined exposure of cells to UCN-01 and L744832 resulted in down-regulation of Mcl-1 and XIAP, both of which are important for the survival of malignant hematopoietic cells,56,57 as well as the formation of proapoptotic cleavage products.34,35 While these phenomena are unlikely to represent primary events in the initiation of the apoptotic cascade, they may nevertheless contribute to amplification of the cell death process.
Combined treatment of cells with L744832 also resulted in enhanced activation of JNK. Decisions involving cell survival and cell death have been related to the net outputs of the cytoprotective ERK and stress-related JNK pathways.58 JNK activation may promote apoptosis by several mechanisms, including phosphorylation/inactivation of Mcl-1 59 or Bcl-2,60 as well as direct effects on mitochondrial cytochrome c release.53 Consistent with these notions, inhibition of JNK by genetic (eg, JNK1 siRNA) or pharmacologic (eg, SP600125) means significantly attenuated UCN-01/L744832-mediated JNK activation and apoptosis. Notably, cross-talk between cytoprotective (eg, MEK1/2/ERK1/2 and Akt) and stress-related (eg, JNK) pathways has been described.61,62 For example, inactivation of the PI3K/Akt pathway has been shown to be essential for JNK activation-mediated apoptosis in CC139 fibroblasts61 ; furthermore, inactivation of the MEK/ERK pathway may potentiate JNK activation in human leukemia cells.63 It is therefore plausible that inactivation of ERK and Akt by UCN-01/L744832 might amplify the lethal effects of JNK activation.
The therapeutic potential of the FTI/UCN-01 regimen will ultimately depend upon whether it exerts selective toxicity toward leukemic cells relative to their normal counterparts or other normal host tissues. In this context, we have previously reported that a regimen consisting of UCN-01 in combination with pharmacologic MEK1/2 inhibitors (eg, PD184352, U0126) preferentially killed cultured and primary AML and multiple myeloma cells while exhibiting only minimal lethality toward normal hematopoietic cells.23,30 Thus, combining UCN-01 with L744832, as in the case of agents acting at a more distal step in the Ras/Raf1/MEK1/2 pathway, results in preferential in vitro lethality toward certain malignant hematopoietic cell types. The basis for this phenomenon is not clear but may stem from observations that neoplastic cells characteristically exhibit dysregulation of checkpoint control (eg, loss of the G1 checkpoint).64,65 Consequently, such cells may be more vulnerable to additional disruptions in cell cycle regulatory pathways than their normal counterparts. Whether this hypothesis is correct and whether this phenomenon occurs under in vivo conditions remain to be determined.
In summary, the present findings indicate that FTIs such as L744832 dramatically increase the capacity of the checkpoint inhibitor UCN-01 to trigger mitochondrial dysfunction, caspase activation, and apoptosis in diverse human leukemia cell types. Furthermore, despite certain similarities, it appears likely that the mechanism of potentiation of UCN-01 lethality by FTIs, despite certain common features, differs fundamentally from that of MEK inhibitors, whose downstream targets are considerably more restricted. Specifically, it seems likely that the capacity of FTIs to disrupt Akt-related pathways, perhaps in combination with the Raf/MEK/ERK cascade, is critically involved in antileukemic synergism. However, the possibility that interactions involving L744832 may involve farnesylation of proteins other than Ras (eg, RhoB, Akt2, or centromeric proteins such as CENP-E or CENP-F)66 cannot be excluded. Given preclinical evidence of their proapoptotic effects,10 attempts to combine FTIs with conventional cytotoxic agents remain the subject of considerable interest.11,13 Additionally, recent studies indicate that FTIs may also be effective in potentiating the activity of agents that interrupt survival signaling pathways (eg, imatinib mesylate).67 The results presented here suggest that attempts to combine FTIs with an agent like UCN-01 that disrupts signaling and cell cycle pathways warrant further attention, particularly in hematologic malignancies.
Prepublished online as Blood First Edition Paper, October 19, 2004; DOI 10.1182/blood-2004-07-2767.
Supported by awards CA63753, CA100866, and CA93738 from the National Institutes of Health, award 6045-03 from the Leukemia and Lymphoma Society of America, and award DAMD-17-03-1-0209 from the Department of Defense.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal