Abstract
Progression to acute myeloid leukemia/myelodysplastic syndromes (AML/MDS) is a possible evolution of polycythemia vera (PV), but whether some patients are at increased natural risk for this complication and how much the contribution of pharmacologic cytoreduction can affect the natural course of the disease remain uncertain. The European Collaboration on Low-dose Aspirin in Polycythemia Vera (ECLAP) prospective project included 1638 patients with PV. AML/MDS was diagnosed in 22 patients after a median of 2.5 years from recruitment in the study and a median of 8.4 years from the diagnosis of PV. Variables associated with progression to AML/MDS were assessed using different models of multivariate analysis. Older age was confirmed as the main independent risk factor (hazard ratio [HR], 4.30; 95% confidence interval [95% CI], 1.16-15.94; P = .0294), whereas overall disease duration failed to reach statistical significance (more than 10 years: HR, 1.91; 95% CI, 0.64-5.69; P = .2466). Exposure to P32, busulphan, and pipobroman (HR, 5.46; 95% CI, 1.84-16.25; P = .0023), but not to hydroxyurea (HU) alone (HR, 0.86; 95% CI, 0.26-2.88; P = .8021), had an independent role in producing an excess risk for progression to AML/MDS compared with treatment with phlebotomy or interferon.
Introduction
The clinical course of polycythemia vera (PV) is characterized by significant thrombohemorrhagic complications and variable risk for disease transformation into myeloid metaplasia with myelofibrosis (MMM) or acute myeloid leukemia/myelodysplastic syndromes (AML/MDS). The incidence of AML/MDS ranges from 5% to 15% after 10 years of disease with progressive increase of risk over time.1-12
Although AML/MDS appears to be a major contributor to the death of patients with PV, there is no generally applicable way to predict which patient is likely to acquire this fatal complication. Initial splenic enlargement, marked leukocytosis, myelofibrotic features, and abnormal karyotype are reported to be associated with the risk for AML/MDS, but they could not be consistently established as useful predictive factors.13-17 The use of chlorambucil or P32 is consistently associated with increased risk for leukemic transformation.1,3,4 Although hydroxyurea (HU) and pipobroman seem to be less leukemogenic than other agents, their effect on the occurrence of AML/MDS remains a controversial issue,5-12 and clearcut evidence supporting which treatment has the best benefit-risk ratio is lacking. The relative rarity of the disease, its long-term course, the late development of malignant complications, and the unwillingness to randomize PV patients to potentially leukemogenic drugs have been major obstacles to the launching of large, well-designed, comparative trials, which are the most appropriate strategy to adequately address such open issues.
The prospective, observational database of the large cohort of the European Collaboration on Low-dose Aspirin in Polycythemia Vera (ECLAP) study, in which 1638 PV patients were monitored using the same methodology used for a clinical trial, is a unique opportunity for a comprehensive reassessment of the risk for AML/MDS associated with clinical and laboratory patients' characteristics and with cytoreductive strategies adopted in clinical practice.
Patients, materials, and methods
Patient procedures
ECLAP study procedures were planned to reflect the routine care of patients with PV without increasing the burden of clinical work at participating centers.18 During the study, the investigators were asked to register all patients with new and old diagnoses of PV made according to the criteria established by the Polycythemia Vera Study Group (PVSG) or by Pearson and Messinezy,19,20 regardless of age, therapy, or duration of disease. Patients were treated with phlebotomy, cytoreductive agents, or both according to the clinical practice in use in each center. Treatment strategies had to be in compliance with the recommendation to maintain the hematocrit value at 0.45 or less and the platelet count at 400 × 109/L or less. Data regarding clinical outcomes, treatments, and laboratory values were collected with the same methodology followed for the nested trial on low-dose aspirin and were specifically recorded at follow-up visits at 12, 24, 36, 48, and 60 months. An ad hoc committee of expert clinicians ensured the validation of causes of death and of nonfatal major events. We examined the rate of AML/MDS diagnoses and classifications that were established using the French-American-British (FAB) Cooperative Group criteria,21,22 and subsequently we analyzed possible risk factors for the occurrence of AML/MDS. According to the Declaration of Helsinki and the requirements of international good clinical practice rules, informed consent was obtained from all patients included in the study.
Statistical methods
The following variables at registration were evaluated: age, sex, disease duration, splenomegaly, packed cell volume (PCV) level, platelet count, white blood cell (WBC) count, cholesterol values, and treatments. The presence of thrombocytosis (platelet count greater than 400 × 109/L) and leukocytosis (WBC count greater than 12 × 109/L) at diagnosis of PV was also included in the analysis. Disease duration was defined as the years between the diagnosis of PV and the time of registration.
Continuous variables have been assessed as such and then categorized according to quartiles or median values. Because the findings did not materially change, we report here the results of the analysis using clinically meaningful cutoffs for age, disease duration, and hematologic parameters, or, when appropriate, by collapsing categories with similar levels of risk. Cytoreductive interventions were also categorized according to their expected leukemogenic potential: (1) no treatment, phlebotomy alone, or interferon alone or with phlebotomy; (2) HU as the only cytoreductive drug; and (3) other cytoreductive agents, alone or in combination.
Differences in proportions were ascertained by means of χ2 analysis for proportions and t test for continuous variables. Separate multivariate age- and sex-adjusted Cox proportional hazard models were fitted to evaluate each variable. Factors found to be associated with increased risk for leukemia were further analyzed by fitting a multivariate Cox proportional hazard model adjusted for all the potential predictors together. To assess possible effect modifications caused by the interaction among age, sex, disease duration, and various clinical variables, we included in the multivariate models interaction terms and carried out separate analyses for different subgroups of patients. Because the results did not materially change with the testing of possible interactions among the variables, we present here only the results for the final multivariate model. Cox proportional hazards event-free survival curves adjusted for covariates were plotted using the corrected group prognosis method.23
All P values were 2-sided. All analyses were performed using the SAS statistical package (SAS Institute, Cary, NC).
Results
Patients
Patients received diagnoses of PV between 1964 and 2001 and were registered in the study between 1997 and 2001. Median time from diagnosis to registration was 3.5 years.
Median age of the entire cohort was 62.1 years at diagnosis of PV and 67.3 years at registration. Four percent of patients were younger than 40, 24.8% were between 40 and 59, 30% were between 60 and 70, and 41.1% were older than 70 years of age. PV was diagnosed in 62% of patients within 5 years of registration, whereas it was diagnosed in 14.4% of patients more than 10 years earlier.
At the time of study closure, patients had been followed up for a median of 2.8 years (4393 person-years; maximum follow-up, 5.3 years). Twenty-one cases of AML and one case of MDS (refractory anemia with excess of blasts in transformation [RAEB-t]) with rapid progression to AML (1.3%) were diagnosed after a median of 2.5 years (range, 0.5-4.1 years) from the registration and 8.4 years (range, 2.2-19.8 years) from the diagnosis of PV. Only 3 cases of AML were preceded by a spent phase with myelofibrosis. All AML/MDS cases were fatal, and no patients survived more than 6 months after the diagnosis of AML/MDS.
The principal characteristics of the patients at registration are shown in Table 1. At the time of diagnosis of PV, patients with AML/MDS were more likely than PV patients without AML/MDS to have had WBC counts exceeding 12 × 109/L (59.1% vs 40.2%; P = .0733). At recruitment, patients with AML/MDS were more likely to have been older (71 vs 65 years; P = .0036), to have had longer disease duration (7.0 vs 3.5 years; P = .0067), to have experienced previous hemorrhagic event (22.7% vs 7.9%; P = .0116), and to have had lower total blood cholesterol level (157 vs 188 mg/dL; P = .0157). Patients with AML/MDS were more likely to have been treated with more than 1 cytoreductive drug (18.2% vs 3.3%; P = .0002) and to have experienced more frequent use of P32 (13.6% vs 2.5%; P = .0014), busulphan (18.2% vs 3.5%; P = .0003), and pipobroman (18.2% vs 6.3%; P = .0246).
Characteristics of patients
. | Patients with AML/MDS, N = 22, 1.34% . | Patients without AML/MDS, N = 1616, 98.66% . | P . |
|---|---|---|---|
| Sex (male) | 8 (36.36) | 934 (57.80) | .0434 |
| Mean age at registration, y (SD) | 70.90 (7.90) | 65.33 (12.71) | .0036 |
| Median time from diagnosis of PV to registration, y (range) | 7.0 (0.68-18.48) | 3.45 (0-39.53) | .0067 |
| Hematologic parameters at diagnosis of PV (%) | |||
| Clinically palpable splenomegaly | 11 (50.00) | 651 (40.28) | .3563 |
| Platelet count > 400 × 109/L | 14 (63.64) | 985 (60.95) | .7977 |
| WBC count > 12 × 109/L | 13 (59.09) | 650 (40.22) | .0733 |
| Clinical and laboratory parameters at registration | |||
| Splenomegaly (%) | 12 (54.55) | 720 (44.55) | .3492 |
| PCV, L/L (SD) | 0.45 (0.06) | 0.47 (0.06) | .1051 |
| Platelet count, × 109/L (SD) | 319 (177) | 400 (208) | .0695 |
| WBC count, × 109/L (SD) | 12.70 (7.42) | 10.89 (8.64) | .3490 |
| Cholesterol level, mg/dL (SD) | 157 (58) | 188 (45) | .0157 |
| Treatment at registration (%) | |||
| Phlebotomy | 13 (59.09) | 1027 (63.55) | .6660 |
| P32 | 3 (13.64) | 41 (2.54) | .0014 |
| Hydroxyurea | 10 (45.45) | 783 (48.45) | .7798 |
| Busulphan | 4 (18.18) | 57 (3.53) | .0003 |
| Chlorambucil | 0 (0.00) | 5 (0.31) | .7939 |
| Pipobroman | 4 (18.18) | 102 (6.31) | .0246 |
| Interferon | 1 (4.55) | 63 (3.90) | .8764 |
| Groups of treatment at registration (%) | |||
| No treatment, phlebotomy only, or interferon only | 5 (22.73) | 664 (41.09) | .0818 |
| Hydroxyurea as only cytoreductive drug | 6 (27.27) | 736 (45.54) | .0873 |
| Any other cytoreductive drug, alone or in combination | 11 (50.00) | 216 (13.37) | < .0001 |
| More than 1 cytoreductive drug | 4 (18.18) | 54 (3.34) | .0002 |
| Event at follow-up (%) | |||
| Thrombosis | 5 (22.73) | 226 (13.99) | .2419 |
| Hemorrhage | 4 (18.18) | 117 (7.24) | .0513 |
. | Patients with AML/MDS, N = 22, 1.34% . | Patients without AML/MDS, N = 1616, 98.66% . | P . |
|---|---|---|---|
| Sex (male) | 8 (36.36) | 934 (57.80) | .0434 |
| Mean age at registration, y (SD) | 70.90 (7.90) | 65.33 (12.71) | .0036 |
| Median time from diagnosis of PV to registration, y (range) | 7.0 (0.68-18.48) | 3.45 (0-39.53) | .0067 |
| Hematologic parameters at diagnosis of PV (%) | |||
| Clinically palpable splenomegaly | 11 (50.00) | 651 (40.28) | .3563 |
| Platelet count > 400 × 109/L | 14 (63.64) | 985 (60.95) | .7977 |
| WBC count > 12 × 109/L | 13 (59.09) | 650 (40.22) | .0733 |
| Clinical and laboratory parameters at registration | |||
| Splenomegaly (%) | 12 (54.55) | 720 (44.55) | .3492 |
| PCV, L/L (SD) | 0.45 (0.06) | 0.47 (0.06) | .1051 |
| Platelet count, × 109/L (SD) | 319 (177) | 400 (208) | .0695 |
| WBC count, × 109/L (SD) | 12.70 (7.42) | 10.89 (8.64) | .3490 |
| Cholesterol level, mg/dL (SD) | 157 (58) | 188 (45) | .0157 |
| Treatment at registration (%) | |||
| Phlebotomy | 13 (59.09) | 1027 (63.55) | .6660 |
| P32 | 3 (13.64) | 41 (2.54) | .0014 |
| Hydroxyurea | 10 (45.45) | 783 (48.45) | .7798 |
| Busulphan | 4 (18.18) | 57 (3.53) | .0003 |
| Chlorambucil | 0 (0.00) | 5 (0.31) | .7939 |
| Pipobroman | 4 (18.18) | 102 (6.31) | .0246 |
| Interferon | 1 (4.55) | 63 (3.90) | .8764 |
| Groups of treatment at registration (%) | |||
| No treatment, phlebotomy only, or interferon only | 5 (22.73) | 664 (41.09) | .0818 |
| Hydroxyurea as only cytoreductive drug | 6 (27.27) | 736 (45.54) | .0873 |
| Any other cytoreductive drug, alone or in combination | 11 (50.00) | 216 (13.37) | < .0001 |
| More than 1 cytoreductive drug | 4 (18.18) | 54 (3.34) | .0002 |
| Event at follow-up (%) | |||
| Thrombosis | 5 (22.73) | 226 (13.99) | .2419 |
| Hemorrhage | 4 (18.18) | 117 (7.24) | .0513 |
Multivariate analysis
Table 2 shows the results of multivariate analysis. Compared with patients younger than 60, those older than 70 were at higher risk for AML/MDS (HR, 4.30; 95% CI, 1.16-15.94; P = .0294), whereas the association between overall disease duration and risk for AML/MDS did not reach statistical significance. Women were at higher risk (HR, 2.93; 95% CI, 1.18-7.26; P = .0205) than men. A barely not statistically significant association was found between WBC count greater than 12 × 109/L and probability of AML/MDS (HR, 2.55; 95% CI, 0.94-6.91; P = .065). Low blood cholesterol levels at recruitment were strongly associated with evolution to AML/MDS (HR, 6.58; 95% CI, 2.08-20.86; P = .0014).
Multivariate analysis
. | Patients with AML/MDS N = 22 . | Patients without AML/MDS N = 1616 . | HR (95% CI) [P] . |
|---|---|---|---|
| Age at registration, y (%) | |||
| Younger than 60 | 3 (13.64) | 470 (29.08) | 1.00 |
| 60-69 | 5 (22.73) | 487 (30.14) | 1.86 (0.42-8.32) [.4167] |
| 70 and older | 14 (63.64) | 659 (40.78) | 4.30 (1.16-15.94) [.0294] |
| Sex (% females) | 14 (63.64) | 682 (42.20) | 2.93 (1.18-7.26) [.0205] |
| Time from diagnosis to registration, y (%) | |||
| 0-5 | 9 (40.91) | 1006 (62.25) | 1.00 |
| 6-10 | 6 (27.27) | 381 (23.58) | 1.27 (0.43-3.73) [.6700] |
| More than 10 | 7 (31.82) | 229 (14.17) | 1.91 (0.64-5.69) [.2466] |
| Presence of splenomegaly* (%) | 15 (68.18) | 934 (57.80) | 0.91 (0.35-2.40) [.8491] |
| Laboratory parameters at registration (%) | |||
| PCV, L/L | |||
| 0.45 or less | 10 (47.62) | 546 (38.72) | 1.00 |
| More than 0.45 | 11 (52.38) | 864 (61.28) | 1.22 (0.49-3.04) [.6693] |
| Platelet count, ×109/L | |||
| 400 or less | 15 (68.18) | 921 (57.60) | 1.00 |
| More than 400 | 7 (31.82) | 678 (42.40) | 0.45 (0.17-1.18) [.1027] |
| WBC count, ×109/L | |||
| 12 or less | 12 (60.00) | 1165 (73.92) | 1.00 |
| More than 12 | 8 (40.00) | 411 (26.08) | 2.55 (0.94-6.91) [.0650] |
| Cholesterol level, mg/dL | |||
| More than 150 | 6 (46.15) | 811 (82.59) | 1.00 |
| 150 or less | 7 (53.85) | 171 (17.41) | 6.58 (2.08-20.86) [.0014] |
| Treatment at registration (%) | |||
| Phlebotomy | 13 (59.09) | 1027 (63.55) | 0.91 (0.37-2.21) [.8261] |
| Hydroxyurea | 10 (45.45) | 783 (48.45) | 1.09 (0.42-2.80) [.8654] |
| Interferon | 1 (4.55) | 63 (3.90) | 1.24 (0.16-9.80) [.8397] |
| Busulphan | 4 (18.18) | 57 (3.53) | 8.64 (2.44-30.60) [.0008] |
| Pipobroman | 4 (18.18) | 102 (6.31) | 4.32 (1.27-14.68) [.0191] |
| P32 | 3 (13.64) | 41 (2.54) | 8.96 (2.13-37.58) [.0027] |
| Chlorambucil | 0 (0.00) | 5 (0.31) | NA |
| Treatment at registration, grouped (%) | |||
| No treatment, phlebotomy only, interferon only† | 5 (22.73) | 664 (41.09) | 1.00 |
| Hydroxyurea as only cytoreductive drug | 6 (27.27) | 736 (45.54) | 0.86 (0.26-2.88) [.8021] |
| Any other cytoreductive drug, alone or in combination | 11 (50.00) | 216 (13.37) | 5.46 (1.84-16.25) [.0023] |
. | Patients with AML/MDS N = 22 . | Patients without AML/MDS N = 1616 . | HR (95% CI) [P] . |
|---|---|---|---|
| Age at registration, y (%) | |||
| Younger than 60 | 3 (13.64) | 470 (29.08) | 1.00 |
| 60-69 | 5 (22.73) | 487 (30.14) | 1.86 (0.42-8.32) [.4167] |
| 70 and older | 14 (63.64) | 659 (40.78) | 4.30 (1.16-15.94) [.0294] |
| Sex (% females) | 14 (63.64) | 682 (42.20) | 2.93 (1.18-7.26) [.0205] |
| Time from diagnosis to registration, y (%) | |||
| 0-5 | 9 (40.91) | 1006 (62.25) | 1.00 |
| 6-10 | 6 (27.27) | 381 (23.58) | 1.27 (0.43-3.73) [.6700] |
| More than 10 | 7 (31.82) | 229 (14.17) | 1.91 (0.64-5.69) [.2466] |
| Presence of splenomegaly* (%) | 15 (68.18) | 934 (57.80) | 0.91 (0.35-2.40) [.8491] |
| Laboratory parameters at registration (%) | |||
| PCV, L/L | |||
| 0.45 or less | 10 (47.62) | 546 (38.72) | 1.00 |
| More than 0.45 | 11 (52.38) | 864 (61.28) | 1.22 (0.49-3.04) [.6693] |
| Platelet count, ×109/L | |||
| 400 or less | 15 (68.18) | 921 (57.60) | 1.00 |
| More than 400 | 7 (31.82) | 678 (42.40) | 0.45 (0.17-1.18) [.1027] |
| WBC count, ×109/L | |||
| 12 or less | 12 (60.00) | 1165 (73.92) | 1.00 |
| More than 12 | 8 (40.00) | 411 (26.08) | 2.55 (0.94-6.91) [.0650] |
| Cholesterol level, mg/dL | |||
| More than 150 | 6 (46.15) | 811 (82.59) | 1.00 |
| 150 or less | 7 (53.85) | 171 (17.41) | 6.58 (2.08-20.86) [.0014] |
| Treatment at registration (%) | |||
| Phlebotomy | 13 (59.09) | 1027 (63.55) | 0.91 (0.37-2.21) [.8261] |
| Hydroxyurea | 10 (45.45) | 783 (48.45) | 1.09 (0.42-2.80) [.8654] |
| Interferon | 1 (4.55) | 63 (3.90) | 1.24 (0.16-9.80) [.8397] |
| Busulphan | 4 (18.18) | 57 (3.53) | 8.64 (2.44-30.60) [.0008] |
| Pipobroman | 4 (18.18) | 102 (6.31) | 4.32 (1.27-14.68) [.0191] |
| P32 | 3 (13.64) | 41 (2.54) | 8.96 (2.13-37.58) [.0027] |
| Chlorambucil | 0 (0.00) | 5 (0.31) | NA |
| Treatment at registration, grouped (%) | |||
| No treatment, phlebotomy only, interferon only† | 5 (22.73) | 664 (41.09) | 1.00 |
| Hydroxyurea as only cytoreductive drug | 6 (27.27) | 736 (45.54) | 0.86 (0.26-2.88) [.8021] |
| Any other cytoreductive drug, alone or in combination | 11 (50.00) | 216 (13.37) | 5.46 (1.84-16.25) [.0023] |
Analysis was adjusted for sex, age, disease duration, treatment at recruitment, presence of splenomegaly, cholesterol level (2 categories), and hematologic parameters at baseline (2 categories).
At diagnosis or at registration.
Forty patients received interferon, alone or in combination with phlebotomy.
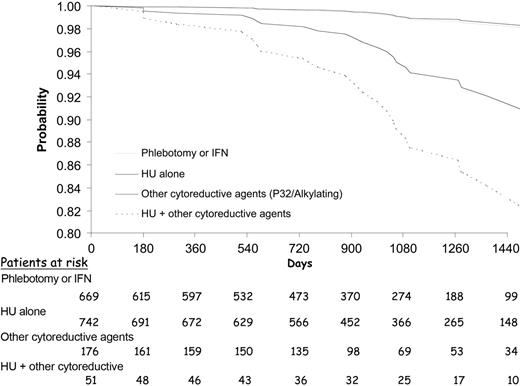
Phlebotomy, hydroxyurea, and interferon were not associated with AML/MDS, whereas pipobroman, busulfan, and P32 were significantly associated with a 4- to 8-fold increased risk for AML/MDS. Compared with patients treated with phlebotomy or interferon, patients receiving hydroxyurea as the only cytoreductive drug had no increased risk for AML/MDS, whereas those treated with pipobroman, busulfan, chlorambucil, or P32, alone or in combination with other drugs, were at significantly higher risk for AML/MDS (HR, 5.46; 95% CI, 1.84-16.25; P = .0023). When we evaluated patients receiving HU plus alkylating agents or P32, the risk for AL was significantly increased (HR, 7.58; 95% CI, 1.85-31.00; P = .0048) compared with patients treated with phlebotomy or interferon. In patients who received “only” alkylating agents or P32, the risk for AL was 4-fold higher (HR, 4.69; 95% CI, 1.42-15.46; P = .0110). Leukemia-free survival times during follow-up for these 4 groups of treatment are shown in Figure 1.
Leukemia-free survival during follow-up according to the treatment received at registration. Gray line indicates phlebotomy or interferon (IFN); thin line, HU alone; bold line, other cytoreductive agents; and broken line, HU + other cytoreductive agents.
Leukemia-free survival during follow-up according to the treatment received at registration. Gray line indicates phlebotomy or interferon (IFN); thin line, HU alone; bold line, other cytoreductive agents; and broken line, HU + other cytoreductive agents.
Discussion
In our study, we registered 1638 patients with diagnoses of PV. New patients and patients with the disorder for more than 20 years were included and then prospectively followed up for a median of 2.8 years. This is the largest and most up-to-date prospective cohort in PV, and it offers a unique opportunity to reassess the factors that influence the natural history of the disease.
One of the principal strengths of the ECLAP project is the use of the same rigorous prospective design with centralized monitoring and validation of events as that used for the nested ECLAP trial.18 The inclusion of patients with old diagnoses of PV and the large sample size allowed us to observe incident cases of AML/MDS in patients who had already been exposed to cytoreductive agents, giving the opportunity to estimate the risk for AML/MDS during different stages of the disease and to partially overcome the limitations of a 3-year follow-up study. We observed 22 patients with AML/MDS, and this figure, though limited, is not far from the figures observed in the largest trials and observational studies available in PV (Table 3).24-28
Principal randomized clinical trials and observational studies evaluating occurrence of AML/MDS in PV patients
Study/year of publication . | Strategies . | N . | Median follow-up, y . | Cases of AL . | Estimated incidence of AL, % . | Estimated annual incidence rate . |
|---|---|---|---|---|---|---|
| Randomized clinical trials | ||||||
| PVSG-01, 19811,19 | Phlebotomy | 134 | 8.6 | 2 | 1.5 | 0.17 |
| Chlorambucil | 141 | 19 | 13 | 1.5 | ||
| P32 | 156 | 15 | 9.6 | 1.1 | ||
| EORTC, 19812 | P32 | 140 | 8 | 2 | 1.4 | 0.17 |
| Busulphan | 145 | 3 | 2.0 | 0.25 | ||
| FPSG, 1997 (older than 65)4 | P32 | 242 | 16, maximum | 35 | 12 at 10 y* | 1.2 |
| P32 + HU | 219 | 21 at 10 y* | 2.1 | |||
| FSPG, 1997 (older than 65)5 | HU | 150 | 16, maximum | 13 | 5 at 10 y* | 0.5 |
| Pipobroman | 142 | Similar in both groups | ||||
| Observational prospective studies | ||||||
| PVSG-08, 19866,7 | HU | 51 | 8.6 | 5 | 9.8 | 1.1 |
| Weinfeld et al, 19948 | HU | 30 | 10 | 3 | 10 | 1 |
| Tarstarsky and Sharon, 19979 | HU | 71 | 10.9 | 4 | 5.6 | 0.5 |
| Brusamolino et al, 198410 | Pipobroman | 100 | 5 | 6 | 6 | 1.2 |
| Passamonti et al, 200011 | Pipobroman | 163 | 10 | 11 | 7 | 0.7 |
| Kiladjian et al, 200312 | Pipobroman | 164 | 11.4 | 32 | 19.5 | 1.7 |
| Passamonti et al, 2003 (younger than 50)24 | Pipobroman, 73% HU, 9% Phlebotomy, 18% | 70 | 14 | 5 | 7 | 0.5 |
| ECLAP, 2004 | Phlebotomy, 63% HU, 48% P32, 3% Interferon, 4% Alkylators, 10% | 1638 | 2.8 | 22 | 1.3 | 0.5 |
| Observational retrospective studies | ||||||
| Halnan and Russell, 196528 | No P32 | 127 | 19, maximum | 0 | 0 | 0 |
| P32 | 107 | 0 | ||||
| Najean et al, 198814 | Phlebotomy, 22% P32, 61% Cytoreductive drugs, 17% | 288 | 10, minimum | 33 | 11.4 | 1.1 |
| GISP, 199525 | Phlebotomy, 31% Alkylators, 59% Other cytoreductive drugs, 10% | 1213 | 5.3 | 28 | 2.3 | 0.43 |
| Brodman et al, 200026 | HU, 63% Alkylators, 19% P32, 5% Phlebotomy, 70% | 59 | 3.7 | 1 | 1.7 | 0.5 |
| Nielsen and Hasselbalch, 200327 | No HU | 13 | 8 | 1 | 7.7 | 0.9 |
| HU, alone or with busulphan | 29 | 7 | 24 | 3.0 |
Study/year of publication . | Strategies . | N . | Median follow-up, y . | Cases of AL . | Estimated incidence of AL, % . | Estimated annual incidence rate . |
|---|---|---|---|---|---|---|
| Randomized clinical trials | ||||||
| PVSG-01, 19811,19 | Phlebotomy | 134 | 8.6 | 2 | 1.5 | 0.17 |
| Chlorambucil | 141 | 19 | 13 | 1.5 | ||
| P32 | 156 | 15 | 9.6 | 1.1 | ||
| EORTC, 19812 | P32 | 140 | 8 | 2 | 1.4 | 0.17 |
| Busulphan | 145 | 3 | 2.0 | 0.25 | ||
| FPSG, 1997 (older than 65)4 | P32 | 242 | 16, maximum | 35 | 12 at 10 y* | 1.2 |
| P32 + HU | 219 | 21 at 10 y* | 2.1 | |||
| FSPG, 1997 (older than 65)5 | HU | 150 | 16, maximum | 13 | 5 at 10 y* | 0.5 |
| Pipobroman | 142 | Similar in both groups | ||||
| Observational prospective studies | ||||||
| PVSG-08, 19866,7 | HU | 51 | 8.6 | 5 | 9.8 | 1.1 |
| Weinfeld et al, 19948 | HU | 30 | 10 | 3 | 10 | 1 |
| Tarstarsky and Sharon, 19979 | HU | 71 | 10.9 | 4 | 5.6 | 0.5 |
| Brusamolino et al, 198410 | Pipobroman | 100 | 5 | 6 | 6 | 1.2 |
| Passamonti et al, 200011 | Pipobroman | 163 | 10 | 11 | 7 | 0.7 |
| Kiladjian et al, 200312 | Pipobroman | 164 | 11.4 | 32 | 19.5 | 1.7 |
| Passamonti et al, 2003 (younger than 50)24 | Pipobroman, 73% HU, 9% Phlebotomy, 18% | 70 | 14 | 5 | 7 | 0.5 |
| ECLAP, 2004 | Phlebotomy, 63% HU, 48% P32, 3% Interferon, 4% Alkylators, 10% | 1638 | 2.8 | 22 | 1.3 | 0.5 |
| Observational retrospective studies | ||||||
| Halnan and Russell, 196528 | No P32 | 127 | 19, maximum | 0 | 0 | 0 |
| P32 | 107 | 0 | ||||
| Najean et al, 198814 | Phlebotomy, 22% P32, 61% Cytoreductive drugs, 17% | 288 | 10, minimum | 33 | 11.4 | 1.1 |
| GISP, 199525 | Phlebotomy, 31% Alkylators, 59% Other cytoreductive drugs, 10% | 1213 | 5.3 | 28 | 2.3 | 0.43 |
| Brodman et al, 200026 | HU, 63% Alkylators, 19% P32, 5% Phlebotomy, 70% | 59 | 3.7 | 1 | 1.7 | 0.5 |
| Nielsen and Hasselbalch, 200327 | No HU | 13 | 8 | 1 | 7.7 | 0.9 |
| HU, alone or with busulphan | 29 | 7 | 24 | 3.0 |
Estimated annual incidence rate is based on number of cases per 100 persons per year.
Actuarial risk.
The relationship between advanced age and AML/MDS has been already reported.29 It is not surprising that a relationship also exists between advanced age and progression to AML/MDS in patients with PV. A more intriguing finding is the higher risk for progression to AML/MDS in women. Previous studies on AML/MDS and on progression to AML/MDS in patients with PV found a similar incidence in men and women.16,29 In further analysis (data not shown), we observed that this increased risk could not be attributed to the greater number of elderly women; within all age groups women were always at higher risk for AML/MDS AL than men, and this risk was more noticeable among patients older than 70. In addition, the proportions of use of alkylating agents and the median times of disease duration and follow-up were similar in men and women. Thus, the increased risk for leukemia in women with PV is unexplained and remains to be confirmed in further studies.
Although the group of patients with progression to AML/MDS had a longer evolution of disease than did patients without progression (8.4 vs 6.4 years from disease diagnosis to the last follow-up information), disease duration did not reach the formal statistical significance level. This result may be influenced by the fact that for subjects who were older at diagnosis—and who had the highest incidence of AML/MDS—the time from diagnosis to progression was shorter than it was in patients whose disease was diagnosed when they were younger (data not shown). This finding seems to confirm advanced age as a central independent risk factor and suggests at least 2 explanations for such increased risk for AML/MDS in older patients. The first is that a more aggressive form of PV may affect older persons. The second is that the expected lower life expectancy of older patients may allow the use of more aggressive cytoreductive strategies whose leukemogenic effect is supposed to appear in the long term. However, the increased life expectancy of Western populations observed in recent years and the possibility that malignancy may develop in elderly patients after shorter periods of exposure to drugs might explain such excess of AML/MDS.
Because the choice of therapeutic strategy was left to the judgment of patients' physicians and the only inclusion criteria regarding treatment was the maintenance of recommended levels of PCV, the ECLAP study constitutes a suitable framework for studying the association between current therapeutic strategies and development of AML/MDS. The difficulties in conducting long-term, large-scale studies, according to proper methodology (ie, randomized clinical trials), of a chronic disease such as PV, which is influenced by multiple, even unknown, factors, justify the limited number of comparative studies published to date and the substantial differences in the incidence of AL in studies with comparable times of follow-up and with evaluations of similar therapeutic strategies (Table 3). Similarly, although higher incidences of AL have been reported with the use of old strategies, such as alkylating agents and radiation, scarcely used today, the issue of leukemogenicity of drugs frequently used (such as HU and pipobroman) is still controversial.
In our study we considered patients undergoing phlebotomy or interferon therapy as the only cytoreductive agent as a reference category that could likely represent the natural risk for PV patients to progress to AML/MDS. The incidence rate of AML/MDS for this group was similar to that found in patients treated with HU as the only pharmacologic treatment (0.29 per 100 persons per year), whereas this rate was 1.8 per 100 persons per year in those receiving at least one alkylating agent or P32 at recruitment. Such findings were confirmed by the results of multivariate analysis, showing that the risk for AML/MDS in patients treated with HU alone was not higher than that of the reference category, whereas this risk was significantly higher in patients treated with pipobroman, busulphan, and P32 alone or in combination with HU. These findings are in agreement with those of randomized clinical trials suggesting an elevated incidence of AML/MDS in patients treated with alkylating agents1 or with radiation3,19 or when cytoreductive drugs were used in combination (eg, P32 or busulphan and HU) to achieve more adequate control of hematocrit values.4,28,30
Prospective observational studies have shown incidence rates of AML/MDS ranging between 0.5% and 1.0% per year in patients treated with HU6-9 and between 0.7% and 1.7% in patients treated with pipobroman.10-12 Our results show an increased risk in patients receiving pipobroman compared with those treated with HU. However, the only randomized clinical trial performed in patients younger than 65 showed similar incidences of AML/MDS in patients treated with the 2 drugs.5
The PVSG 01 trial compared the benefit-risk ratio of phlebotomy with that of chlorambucil and of P32. AML/MDS developed in 1.5% of patients who underwent phlebotomy during a median follow-up of 8.6 years (incidence rate, 0.17% cases per year).7,19 This estimate, however, pertains only to the 10% of patients who were allocated to phlebotomy, had no thrombotic events, and continued the same treatment until the 10th year of study.30 Therefore, such low occurrence of AML/MDS could not be the true incidence rate of AML/MDS in patients undergoing only phlebotomy because it reflects a select group of patients who likely had a less proliferative form of PV.31 The only other clinical trial comparing cytoreductive drugs with no intervention was conducted in essential thrombocythemia.32 After a median follow-up of 73 months, the incidence rates of AML/MDS in this study were 0.0%, 0.6%, and 5.4% per year for no pharmacologic treatment and cytoreduction with HU and HU plus busulphan, respectively.33
Because our study design prevented us from assessing the total exposition time to cytoreductive drugs, we could not rule out the possibility that HU could be leukemogenic in the long run. However, our results are reassuring and suggest that if such a risk exists, it is probably not much greater than the natural tendency of the disease to progress to AML/MDS and is definitely lower than that of pipobroman, busulphan, and P32. Our study had the power to capture the risk for AML/MDS associated with drugs used by less than 15% of the patients. Because HU was prescribed to approximately half the population and was found not to be associated with the development of AML/MDS, we could safely rule out the possibility of a large leukemogenic effect of this drug in PV patients.
We found that low levels of total blood cholesterol at recruitment were strongly associated with the development of AML/MDS. Some small studies have found low levels of cholesterol in patients affected by hematologic malignancies,34-37 and some of these reports include patients with PV.34,36,37 It has been observed that the lowest levels of blood cholesterol are observed in advanced stages of proliferative diseases, and some have suggested the use of this parameter as a marker of disease activity.33-35 Given that the average total blood cholesterol level in a 35- to 75-year-old Mediterranean population, such as Italian, is 206 mg/dL,38 the average cholesterol level of the ECLAP population (187.4 mg/dL) is surprisingly low and is indicative of the proliferative nature of the disease. In addition to patients with progression to AML/MDS, others who acquired myelofibrosis also had more noticeable hypocholesterolemia (data not shown). These findings resemble the increased risk for death from cancer observed in patients with low cholesterol levels, and they suggest that low cholesterol could be a precocious marker of neoplastic diseases in otherwise healthy persons undergoing no cholesterol-lowering treatment.39
Although the relationship between high WBC counts at diagnosis and at recruitment with the later development of AML/MDS was not statistically significant in our study, this association has been observed in some studies,12-14 and such findings suggest that high WBC counts could mark a different pathway of proliferation in some patients, indicating a subset of subjects with a distinct natural risk for progression to AML/MDS.
Relationships between the progression to AML/MDS and markers of cellular proliferation, such as low blood cholesterol levels, and the administration of alkylating drugs, alone or in combination with other drugs, should not be viewed as independent phenomena because they may indicate that some PV patients may have a more aggressive disease with a higher proliferation rate, making them more likely to be resistant to cytoreductive treatments and, in the end, to progress to AML/MDS. If so, these patients are also more likely to be exposed to recurrent changes in therapy, often toward more aggressive options, for improved control of the disease. Because such work-up bias cannot be eliminated in observational studies and because the effect of unmeasured and unknown confounders cannot be adjusted for in multivariate analyses, new randomized trials are needed for correct evaluation of the efficacy and safety of cytoreductive treatments in PV to determine a final answer to the debated issue of the supposed leukemogenic effect of frequently used drugs such as HU.
Appendix
Principal ECLAP Investigators (recruiting 40 or more patients in the study): Austria, Vienna: H Gisslinger. Italy, Bergamo: T Barbui, G Finazzi, S Pusterla, A Falanga, M Galli. Bologna: S Tura, C Finelli, G Marino. Monza: E Pogliani, I R Miccolis, A La Rocca, E Elli. Padova: M L Randi. Reggio Emilia: L Gugliotta, F Ilariucci. Venezia: T Chisesi, A Polacco. Venezia-Mestre: T Chisesi, G Capnist. Sweden, Göteborg: J Kutti, H Wadenvik. Other ECLAP Investigators: Austria, Innsbruck: G Gastl, Ch Ludescher, M Fiegl. Linz: D Lutz, M Girschikfsky, G Michlmayr, E Rechberger. Wr Neustadt: H Niessner, E Ivansich. France, Paris: J D Rain, C Chommienne-Thomas. Germany, Mannheim: R Hehlmann, G Engelich. Ulm: E Kohne, A Kramer. Greece, Thessaloniki: J I Christakis, M Papaioannou, G Gerotziafas, J Korantzis, P Marinaki, A Kioumi, I Katodritou. Ireland, Dublin: D McCarthy, R O'Donnell. Israel, Afula: M Bennett. Ashkelon: G Lugassy. Kfar Saba: M Ellis. Tel-Aviv: A Eldor,† E Naparstek, R Marilus. Italy, Ancona: P Leoni, S Rupoli, A R Scortechini, V Agostini. Avellino: E Volpe, F Palmieri, A Volpe, G Storti, A Ciampa. Bari: F Dammacco, V M Lauta, G Ranieri, R Rizzi. Brescia: G Rossi, C Almici, A Capucci, F Zanetti. Castrovillari CS: F Laghi, G Micciulli. Catania: R Giustolisi, RR Cacciola, E Cacciola. Catanzaro: A Peta, D Magro. Como: G Frigerio, F Alberio, A Beretta. Cuneo: M Bonferroni, Raviolo. Firenze: P L Rossi Ferrini, A Grossi, A Fabbri. Genova: M Gobbi, I Pierri, M Cavaliere. Latina: S Nardelli, A Centra. Messina: C Musolino, G Bellomo, O Tringali, G Spatari. Milano: P Foa, G Gerli, M C Carraro, A Zanella, A Iurlo, F Barraco. Modena: G Torelli, M Marietta. Montebelluna TV: A Puglisi, G Sardeo. Napoli: B Rotoli, V Martinelli, R Ciancia, A Abbadessa, O Villani, R Cimino, A Fasanaro. Parma: V Rizzoli, C Caramatti, Gaeta. Pavia: M Lazzarino, F Passamonti, M Lazzola, L Malabarba. Pescara: D Natale, G. Davì, S Pulini. Roma: R Landolfi, E De Candia, S Amadori, F Buccisano, F Mandelli, E Montefusco, M C Petti, A Spadea, M Carotenuto,† A Morelli, M Nobile. Sassari: M Longinotti, S M Pardini. Siena: F Lauria, A Buccalossi, S Gentili. Taranto: P Mazza, M Cervellera, A Maggi. Teramo: A Di Francesco, E Pasqualoni. Vicenza: F Rodeghiero, M Ruggeri. Udine: M Baccarani, D Russo. Spain, Barakaldo: B Arrizabalaga. Barcelona: A Remacha. Burgos: B Pérez De Mendiguren. La Laguna (Santa Cruz de Tenerife): L Hernández-Nieto, M T Hernández-García, G González-Brito, P Machado. León: G Garcia. Madrid: A Villegas, A Peña, A González Fernández. Valencia: F Carbonell, A Del Arco. Sweden, Borås: H Bäck. Danderyd: L Stenke. Eksjö: S Hansen. Huddinge: J Samuelsson. Hudiksvall: L Nezadal. Kristianstad: G Larsson. Kungälv: G Strömblad. Luleå: B Lauri. Motala: B O Ryden. Norrköping: T End. Örebro: O Linder. Örnsköldsvik: B G Lundholm. Säffle: O Lannemyr. Skövde: J Väärt. Sundsvall: M Strandberg. Uddevalla: B Andréasson, D Stockelberg. Värnamo: L Engquist. Västerås: F Pasquariello. Switzerland, Basel: A Tichelli. Oldenburg: B Otremba, H F Hinrichs. Schweiz: W Weber-Stadelmann. The Netherlands, Heerlen: M M F Fickers. Rotterdam: J J Michiels. United Kingdom, Belfast: M F McMullin. Birmingham: D Bareford. Bournemouth: D G Oscier, N Bowey. Cambridge: T A R Green. London: M Messinezy. South Yorks: P C Taylor. Steering committee: R Landolfi, T Barbui, G de Gaetano, R Marchioli, Y Najean, C Patrono, TC Pearson. Scientific and organizing secretariat: R Marchioli, S Atashkar, V Caruso, A Di Blasio, G Finazzi, H Gisslinger, E Mari, D Tamayo, G Tognoni. Data management and analysis: G Borrelli, B Ferri, R M Marfisi, M Olivieri, A Polidoro, R Spoltore. Event adjudicating committee: R Landolfi, G Levantesi, R Di Mascio, G Finazzi, G Miceli, G Sperti. Writing committee: V Caruso, G Finazzi, R Landolfi, R Marchioli, J Kutti, H Gisslinger, C Patrono, R Marilus, A Villegas, G Tognoni, T Barbui.
Prepublished online as Blood First Edition Paper, December 7, 2004; DOI 10.1182/blood-2004-09-3426.
A complete list of the ECLAP Investigators appears in the Appendix.
The ECLAP Project was supported by the European Union, BIOMED 2 Program (contract ERBBMH4CT961433).
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal