Comment on Albrecht et al, page 542
Scott syndrome is an extremely rare inherited hemorrhagic disorder linked to the lack of exposure of procoagulant phosphatidylserine (PS) to the external leaflet of the plasma membrane of activated platelets and other hematologic lineages.1-3The loss of membrane lipid asymmetry, however, occurs in virtually all eukaryotic cells upon stimulation (including apoptosis)3and is associated with more ancestral functions than hemostasis. Hence, it is of prime interest to identify effectors of this basic process.
To date, only 3 documented cases of Scott syndrome have been reported, which is insufficient to allow the identification of positional candidate genes. It should be emphasized that none of these patients exhibited clinical symptoms other than bleeding, suggesting that the rapid PS egress for an efficient platelet hemostatic response and slower membrane remodeling occurring during apoptosis are under different control mechanisms. This does not, however, rule out that some or most of the actors can be shared. On the one hand, several studies have pointed to adenosine triphosphate (ATP)–binding cassette (ABC) transporters as possible phospholipid translocases (in particular, some that fulfill multidrug efflux function), but none has been confirmed as a true PS floppase. On the other hand, a nonspecific and energy-independent family of so-called scramblases has been proposed. But canonical phospholipid scramblase 1 (PLSCR1) is normally expressed in Scott cells, and other members or other candidate(s) remain to be characterized as effectors of bidirectional phospholipid transmembrane redistribution. Another ABC family member, ABC transporter A1 (ABCA1), involved in phagocytosis and cholesterol reverse transport and mutated in Tangier dyslipidemia, has been considered after the targeted deletion of the corresponding locus resulted in a phenotype evocative of partial Scott syndrome.4 In this issue of Blood, Albrecht and colleagues precisely report 2 types of mutations that may contribute to the Scott phenotype.
The patient was found heterozygous for a novel missense substitution in ABCA1 R1925Q, which has not been previously reported in the relatively large cohort of Tangier patients described in the literature. In vitro expression experiments revealed an impaired traffic of ABCA1 R1925Q to the plasma membrane, and sequence comparisons pointed to a conserved functional importance of this residue. The second mutation, which remains to be identified, accounts for significant reduction of ABCA1 mRNA levels and may affect a trans-acting regulatory gene. Using a retroviral expression system, complementation of the defect of PS externalization proved feasible in Epstein-Barr virus (EBV)–transformed Scott B cells. Unfortunately, no investigation of the protein expression levels was possible, most likely as a result of the lack of adequate antibody.FIG1
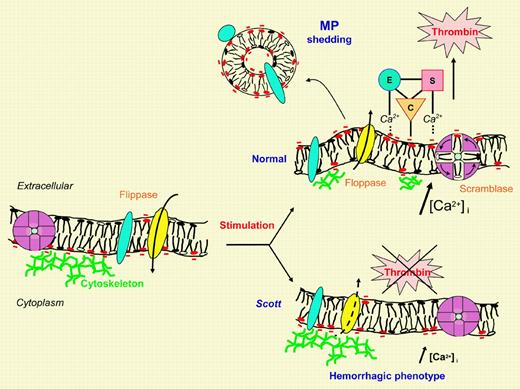
Membrane remodeling in normal and Scott cells. In the plasma membrane, dynamic maintenance or loss of asymmetric phospholipid distribution results from opposite fluxes under the control of specific transporters governing inward (flip) or outward (flop) translocation. In the resting membrane, the flippase activity of aminophospholipid translocase is prominent and subtained (left, yellow ellipse). Thus, aminophospholipids, phosphatidylserine (PS; red negative polar heads), and phosphatidylethanolamine (PE; open polar heads) are sequestered into the inner leaflet. Following stimulation, specific vectorial floppase and/or nonspecific bidirectional scramblase activities are rapidly turned on, and PS and PE are moved to the outer leaflet (upper right). Flippase activity is shut down, leading to transient mass imbalance between the two leaflets. Membrane blebbing ultimately resolves into microvesicle or microparticle (MP) shedding after cytoskeleton degradation by calcium-dependent proteolysis. Stimulated cell membrane and MP therefore expose PS, which is particularly crucial in platelet procoagulant response for the calcium-dependent assembly of the cascade of clotting enzyme complexes. The assembly of these complexes, composed of an enzyme (E) and a cofactor (C) in the activation of a substrate (S), culminates in the generation of sufficient thrombin for efficient hemostasis. In resting Scott cells, flippase activity is indeed operational; stimulation remains without effect on the swift induction of floppase and/or scramblase and consecutive membrane shedding, explaining the resulting hemorrhagic phenotype (lower right). During the slower process of apoptotic cell death, however, PS egress is normal in Scott cells. As yet, floppase candidates have remained elusive.
Membrane remodeling in normal and Scott cells. In the plasma membrane, dynamic maintenance or loss of asymmetric phospholipid distribution results from opposite fluxes under the control of specific transporters governing inward (flip) or outward (flop) translocation. In the resting membrane, the flippase activity of aminophospholipid translocase is prominent and subtained (left, yellow ellipse). Thus, aminophospholipids, phosphatidylserine (PS; red negative polar heads), and phosphatidylethanolamine (PE; open polar heads) are sequestered into the inner leaflet. Following stimulation, specific vectorial floppase and/or nonspecific bidirectional scramblase activities are rapidly turned on, and PS and PE are moved to the outer leaflet (upper right). Flippase activity is shut down, leading to transient mass imbalance between the two leaflets. Membrane blebbing ultimately resolves into microvesicle or microparticle (MP) shedding after cytoskeleton degradation by calcium-dependent proteolysis. Stimulated cell membrane and MP therefore expose PS, which is particularly crucial in platelet procoagulant response for the calcium-dependent assembly of the cascade of clotting enzyme complexes. The assembly of these complexes, composed of an enzyme (E) and a cofactor (C) in the activation of a substrate (S), culminates in the generation of sufficient thrombin for efficient hemostasis. In resting Scott cells, flippase activity is indeed operational; stimulation remains without effect on the swift induction of floppase and/or scramblase and consecutive membrane shedding, explaining the resulting hemorrhagic phenotype (lower right). During the slower process of apoptotic cell death, however, PS egress is normal in Scott cells. As yet, floppase candidates have remained elusive.
How these mutations relate to Scott syndrome may be debated, but this observation at least provides the first link between a defect in a transbilayer phospholipid transport pathway, that of ABCA1 here, and the bleeding phenotype. In light of other genetic disorders, other mutated or deleted elements may be considered, including other ABC true transporters or regulators such as ABCA1 itself, as already suggested for the latter.5
In Scott syndrome, bleeding episodes are moderate when compared with spontaneous hemorrhages in severe hemophilia, which affect a much greater number of patients. It has to be kept in mind that the Scott defect most likely can be traced back to a hematopoietic stem cell line or even earlier. Hence, the incapacity of PS externalization may turn lethal because of the prime role of PS as a recognition determinant for phagocytosis, for instance.3 This could explain the surprising rarity of Scott syndrome patients who might have survived as a result of the intervention of an opportune rescue mechanism. ▪

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal